Preparatory Problems of the 40th IChO - IChO-2016
... Compound A is a stable salt of metal H. It contains 11.97 % N, 3.45 % H and 41.03 % O (mass fractions), besides the metal. The following chart describes some reactions starting from A and H (∆ signifies heating). Above the arrows the necessary reactants are displayed. All substances tagged with a le ...
... Compound A is a stable salt of metal H. It contains 11.97 % N, 3.45 % H and 41.03 % O (mass fractions), besides the metal. The following chart describes some reactions starting from A and H (∆ signifies heating). Above the arrows the necessary reactants are displayed. All substances tagged with a le ...
14.1 Dynamic Equilibrium, Keq , and the Mass Action Expression
... Dr. Fred Omega Garces Chemistry 201 Miramar College ...
... Dr. Fred Omega Garces Chemistry 201 Miramar College ...
Study Guide for Content Mastery - Student Edition
... a specific situation. There are many kinds of graphs. One of the most common is the bar graph. ...
... a specific situation. There are many kinds of graphs. One of the most common is the bar graph. ...
GCSE Chemistry Specification Specification for exams from 2014 2014
... Scientific investigations often seek to identify links between two or more variables. These links may be: – causal, in that a change in one variable causes a change in another − due to association, in that changes in one variable and a second variable are linked by a third variable − due to chance o ...
... Scientific investigations often seek to identify links between two or more variables. These links may be: – causal, in that a change in one variable causes a change in another − due to association, in that changes in one variable and a second variable are linked by a third variable − due to chance o ...
CHEMICAL EQUILIBRIUM
... 8. Even though the individual sets of equilibrium concentrations are quite different for the different situations, the equilibrium constant which depends on the ratio of the concentrations, remains the same. 9. Each set of equilibrium concentrations is called an ______________________________. 10. ...
... 8. Even though the individual sets of equilibrium concentrations are quite different for the different situations, the equilibrium constant which depends on the ratio of the concentrations, remains the same. 9. Each set of equilibrium concentrations is called an ______________________________. 10. ...
GEOCHEMICAL AND BIOGEOCHEMICAL
... quickly becomes a chore. After calculating a few Eh–pH diagrams, what does one learn by manually producing more plots? For many students, trees quickly come to obscure a beautiful forest. The computer can take over the mechanics of basic tasks, once they have been mastered, freeing the student to ab ...
... quickly becomes a chore. After calculating a few Eh–pH diagrams, what does one learn by manually producing more plots? For many students, trees quickly come to obscure a beautiful forest. The computer can take over the mechanics of basic tasks, once they have been mastered, freeing the student to ab ...
Chapter One Hemilabile Ligands in Transition
... conversion of 4 to 5 could only be accomplished photochemically. Solutions of 5 with an inert gas purge converted to the expected monocarbonyl 3. The 1H NMR data of 3 was shown to be consistent with two equivalent structures (3 and 3’) in equilibrium involving a five-coordinate intermediate (Scheme ...
... conversion of 4 to 5 could only be accomplished photochemically. Solutions of 5 with an inert gas purge converted to the expected monocarbonyl 3. The 1H NMR data of 3 was shown to be consistent with two equivalent structures (3 and 3’) in equilibrium involving a five-coordinate intermediate (Scheme ...
LABORATORY MANUAL FOR CHEMISTRY 102
... where [A], [B], [C], ... represent molarities of all chemical species that affect the rate, and x, y, z, ... are the experimentally determined exponents for each species. (The overall order of the reaction is equal to the sum of x + y + z +... .) The term k is known as the rate constant for the reac ...
... where [A], [B], [C], ... represent molarities of all chemical species that affect the rate, and x, y, z, ... are the experimentally determined exponents for each species. (The overall order of the reaction is equal to the sum of x + y + z +... .) The term k is known as the rate constant for the reac ...
9/10/10 1 Chemistry 121: Atomic and Molecular Chemistry
... The Mole and Molar Mass: Chemists measure atoms and molecules in moles. • In the SI system the mole (mol) is the amount of a substance that contains as many elementary entities (atoms, molecules, or other particles) as there are atoms in exactly 12 g (or 0.012 kg) of the carbon-12 isotope. The actu ...
... The Mole and Molar Mass: Chemists measure atoms and molecules in moles. • In the SI system the mole (mol) is the amount of a substance that contains as many elementary entities (atoms, molecules, or other particles) as there are atoms in exactly 12 g (or 0.012 kg) of the carbon-12 isotope. The actu ...
20. Chemical Equilibrium
... The formula for water is not included in the examples above because dissolving is not a chemical change and, therefore, water is not consider a reactant. When substances dissolve in water they do not chemically react with it. They merely come part in the water. Later in this chapter you will study r ...
... The formula for water is not included in the examples above because dissolving is not a chemical change and, therefore, water is not consider a reactant. When substances dissolve in water they do not chemically react with it. They merely come part in the water. Later in this chapter you will study r ...
SCH4U TEXT BOOK
... s you wander through the supermarket, some advertising claims catch your eye. “Certified organic” and “all natural” are stamped on the labels of some foods. Other labels claim that the foods are “chemical free.” As a chemistry student, you are aware that these labels may be misleading. Are all “chem ...
... s you wander through the supermarket, some advertising claims catch your eye. “Certified organic” and “all natural” are stamped on the labels of some foods. Other labels claim that the foods are “chemical free.” As a chemistry student, you are aware that these labels may be misleading. Are all “chem ...
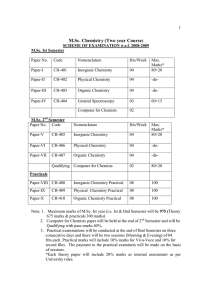
M.Sc. Chemistry (Two year Course)
... Entropy changes in reversible and irreversible processes; variation of entropy with temperature , pressure and volume, entropy concept as a measure of unavailable energy and criteria for the spontaneity of reaction; free energy functions and their significance, criteria for spontaneity of a process; ...
... Entropy changes in reversible and irreversible processes; variation of entropy with temperature , pressure and volume, entropy concept as a measure of unavailable energy and criteria for the spontaneity of reaction; free energy functions and their significance, criteria for spontaneity of a process; ...
Mole Concept - Shailendra Kumar Chemistry
... (a) If the haemoglobin molecule contain one atom of iron. What is its molecular weight ? (b) If the haemoglobin molecule contains four atom of iron. What is its molecular weight ? ...
... (a) If the haemoglobin molecule contain one atom of iron. What is its molecular weight ? (b) If the haemoglobin molecule contains four atom of iron. What is its molecular weight ? ...
5 Steps
... constantly evolving and so this guide has evolved. In this edition, we have updated the book to match the new AP Chemistry exam, especially the changes in the free-response section. In the new exam, questions about laboratory experiments will be treated differently than in previous years. We have re ...
... constantly evolving and so this guide has evolved. In this edition, we have updated the book to match the new AP Chemistry exam, especially the changes in the free-response section. In the new exam, questions about laboratory experiments will be treated differently than in previous years. We have re ...
Physiological and phylogenetic studies of thermophilic
... (94.7 – 97.3% homology). Detailed results on various growth parameters were investigated for both moderate thermophilic strains, under various growth conditions (chemolithotrophic, heterotrophic, mixotrophic), concerning hydrogen uptake rate, biomass formation, sulfate production and utilization rat ...
... (94.7 – 97.3% homology). Detailed results on various growth parameters were investigated for both moderate thermophilic strains, under various growth conditions (chemolithotrophic, heterotrophic, mixotrophic), concerning hydrogen uptake rate, biomass formation, sulfate production and utilization rat ...
General chemistry laboratory activities, Lorentz
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
content - Thesis Scientist
... sulphate, respectively are connected in series. A steady current of 1.5 ampere was passed through them until 1.45 g of silver were deposited at the cathode of cell B. How long did the current flow? What mass of copper and what mass of zinc were deposited in the concerned cells? (Atomic masses of Ag ...
... sulphate, respectively are connected in series. A steady current of 1.5 ampere was passed through them until 1.45 g of silver were deposited at the cathode of cell B. How long did the current flow? What mass of copper and what mass of zinc were deposited in the concerned cells? (Atomic masses of Ag ...
General and Inorganic Chemistry
... 2. II Writing chemical equations ...................................................................................................... 14 1. II.1 Qualitative relationships ............................................................................................... 14 2. II.2 Quantitative relatio ...
... 2. II Writing chemical equations ...................................................................................................... 14 1. II.1 Qualitative relationships ............................................................................................... 14 2. II.2 Quantitative relatio ...
Coordination Chemistry Reviews Iron–dinitrogen coordination
... debated, but from the electron density data it is expected to be either C, N, or O [20–29]. The FeMo cofactor is accepted to be the site of N2 binding and reduction based on a wide variety of evidence [13]. In fact, recent site-directed mutagenesis studies have suggested that two of the “belt” iron ...
... debated, but from the electron density data it is expected to be either C, N, or O [20–29]. The FeMo cofactor is accepted to be the site of N2 binding and reduction based on a wide variety of evidence [13]. In fact, recent site-directed mutagenesis studies have suggested that two of the “belt” iron ...
5 Steps to a 5 AP Chemistry, 2008-2009 Edition
... constantly evolving and so this guide has evolved. In this edition, we have updated the book to match the new AP Chemistry exam, especially the changes in the free-response section. In the new exam, questions about laboratory experiments will be treated differently than in previous years. We have re ...
... constantly evolving and so this guide has evolved. In this edition, we have updated the book to match the new AP Chemistry exam, especially the changes in the free-response section. In the new exam, questions about laboratory experiments will be treated differently than in previous years. We have re ...
Calculations with Chemical Formulas and Chemical Reactions
... substance. The average atomic weight of an element is generally found at the bottom of the element block (Figure 7.1). The chemical formula of the substance clearly indicates how many atoms or i ...
... substance. The average atomic weight of an element is generally found at the bottom of the element block (Figure 7.1). The chemical formula of the substance clearly indicates how many atoms or i ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.