AS Chemistry Teacher Handbook
... The guide is not intended as a comprehensive reference, but as support for professional teachers to develop stimulating and exciting courses tailored to the needs and skills of their own learners in their particular institutions. The guide offers assistance to teachers with regard to the depth of co ...
... The guide is not intended as a comprehensive reference, but as support for professional teachers to develop stimulating and exciting courses tailored to the needs and skills of their own learners in their particular institutions. The guide offers assistance to teachers with regard to the depth of co ...
Equilibrium
... The value of the equilibrium constant for any reaction can be determined by experiment. As detailed in the above section, the equilibrium position for a given reaction does not depend on the starting concentrations, so the equilibrium constant has the same value regardless of the initial amounts of ...
... The value of the equilibrium constant for any reaction can be determined by experiment. As detailed in the above section, the equilibrium position for a given reaction does not depend on the starting concentrations, so the equilibrium constant has the same value regardless of the initial amounts of ...
08272012BC Science Chem 12 Chapter 1 Answer Key
... water will occur as water is formed in a reaction that occurs in aqueous solution. This is, of course, nonsense! As the entire reaction occurs in the solvent water, there will simply be a small amount of water formed, replacing the hydrogen and oxygen atoms (actually hydrogen and hydroxide ions) rea ...
... water will occur as water is formed in a reaction that occurs in aqueous solution. This is, of course, nonsense! As the entire reaction occurs in the solvent water, there will simply be a small amount of water formed, replacing the hydrogen and oxygen atoms (actually hydrogen and hydroxide ions) rea ...
Chapter 3 - Educator
... Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO2 molecules. The number of NO2 molecules on the right equals the number of NO molecules on the left as the balanced equation requires. Counting the atoms, we find eight ...
... Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO2 molecules. The number of NO2 molecules on the right equals the number of NO molecules on the left as the balanced equation requires. Counting the atoms, we find eight ...
PDF File
... interactions, Shan et al. developed “thermodynamic fingerprint analysis” (TFA),1 a quantitative approach that determines whether the same or different metal ions give rescue at different positions (11). TFA and related analyses (11, 12) provided functional evidence for a network of transition state ...
... interactions, Shan et al. developed “thermodynamic fingerprint analysis” (TFA),1 a quantitative approach that determines whether the same or different metal ions give rescue at different positions (11). TFA and related analyses (11, 12) provided functional evidence for a network of transition state ...
Chemistry II - Mr. Dougan`s Wonderful World of Chemistry
... rather than in the molecular from NaOH. Sodium chloride is soluble in water and thus exists as ions, while water is a weak electrolyte and must be written in molecular form. Since the sodium and chloride ions do not change their form in this reaction, you will be determining the heat of formation fo ...
... rather than in the molecular from NaOH. Sodium chloride is soluble in water and thus exists as ions, while water is a weak electrolyte and must be written in molecular form. Since the sodium and chloride ions do not change their form in this reaction, you will be determining the heat of formation fo ...
A Biosensor Based on Immobilization of Horseradish Peroxidase in Chitosan Matrix Cross-linked with Glyoxal for Amperometric Determination of Hydrogen Peroxide
... and robust amperometric hydrogen peroxide biosensor based on HRP immobilized by chitosan film cross-linked with glyoxal. We selected the “sandwich” configuration: ferrocene-chitosan: HRP: chitosan-glyoxal. The chitosan film cross-linked with glyoxal permitted high enzyme loadings due to the cross-li ...
... and robust amperometric hydrogen peroxide biosensor based on HRP immobilized by chitosan film cross-linked with glyoxal. We selected the “sandwich” configuration: ferrocene-chitosan: HRP: chitosan-glyoxal. The chitosan film cross-linked with glyoxal permitted high enzyme loadings due to the cross-li ...
Problem Authors - PianetaChimica
... e) Compound C was first used as rocket fuel during World War II. Today, it is used as a lowpower propellant in spacecrafts. In the presence of certain catalysts such as carbon nanofibers or molybdenum nitride supported on alumina, one of the decomposition reactions of C involves production of ammoni ...
... e) Compound C was first used as rocket fuel during World War II. Today, it is used as a lowpower propellant in spacecrafts. In the presence of certain catalysts such as carbon nanofibers or molybdenum nitride supported on alumina, one of the decomposition reactions of C involves production of ammoni ...
Name:
... b) Dispersion forces: The C5H12 (hexane) molecule has covalent bonds between atoms. c) Dipole-induced dipole: The H2O molecule has a bent shape, so it has a molecular dipole. This dipole induces a dipole in the O2 molecules, resulting in their mutual attraction. d) Ion-dipole interactions: NaCl diss ...
... b) Dispersion forces: The C5H12 (hexane) molecule has covalent bonds between atoms. c) Dipole-induced dipole: The H2O molecule has a bent shape, so it has a molecular dipole. This dipole induces a dipole in the O2 molecules, resulting in their mutual attraction. d) Ion-dipole interactions: NaCl diss ...
2003 AP Chemistry Form B Scoring Guidelines - AP Central
... (e) How would the calculated concentration of the KMnO4 solution be affected if 40 mL of water was added to the oxalic acid initially instead of 30 mL? Explain your reasoning. There would be no effect on the concentration of the KMnO4 solution. We are only interested in the moles of oxalic acid. Sin ...
... (e) How would the calculated concentration of the KMnO4 solution be affected if 40 mL of water was added to the oxalic acid initially instead of 30 mL? Explain your reasoning. There would be no effect on the concentration of the KMnO4 solution. We are only interested in the moles of oxalic acid. Sin ...
Grossmont College Chemistry 120 Laboratory Manual 6th Edition
... data, but do not clutter the calculation section with arithmetic details. Likewise, think through and answer important questions that are intended to give you an understanding of the principles in which the experimental procedure is based as you perform the experiment. Scientists learn much by discu ...
... data, but do not clutter the calculation section with arithmetic details. Likewise, think through and answer important questions that are intended to give you an understanding of the principles in which the experimental procedure is based as you perform the experiment. Scientists learn much by discu ...
Topical KCSE Mock-Chemistry Answers(15 Schools)
... (iii) Magnesium react with oxygen and nitrogen hence greater of fraction of air is used. (iv) (a) Blue litmus changed to red as remained red. The solution was acid due to phosphoric (b) Red litmus changed to blue as blue remained blue due to formation of basic magnesium hydroxide ammonia solution. ( ...
... (iii) Magnesium react with oxygen and nitrogen hence greater of fraction of air is used. (iv) (a) Blue litmus changed to red as remained red. The solution was acid due to phosphoric (b) Red litmus changed to blue as blue remained blue due to formation of basic magnesium hydroxide ammonia solution. ( ...
MULTIPLY CHOICE QUESTIONS ON MEDICAL CHEMISTRY
... 2.32. Decomposition of HI (hydrogen iodide) on the surface of gold is the reaction of: А. second order B. higher order C. first order D. zero order E. ...
... 2.32. Decomposition of HI (hydrogen iodide) on the surface of gold is the reaction of: А. second order B. higher order C. first order D. zero order E. ...
Isotopes of Volatile Organic Compounds: An Emerging Approach for
... potential applications of this information for increasing our understanding of their atmospheric budgets and chemistry. For example, the kinetic isotope effect of atmospheric photochemical loss processes of nonmethane hydrocarbons (NMHCs) has been exploited to estimate their photochemical age from a ...
... potential applications of this information for increasing our understanding of their atmospheric budgets and chemistry. For example, the kinetic isotope effect of atmospheric photochemical loss processes of nonmethane hydrocarbons (NMHCs) has been exploited to estimate their photochemical age from a ...
The shock tube as wave reactor for kinetic studies and material
... After Schott and Kinsey [5] demonstrated that the course of an exothermic reaction like H2 1 O2 ! products, highly diluted in argon, could be satisfactorily resolved using a shock tube, several other investigators used the technique to study many other reactions of varying complexity, covering a wid ...
... After Schott and Kinsey [5] demonstrated that the course of an exothermic reaction like H2 1 O2 ! products, highly diluted in argon, could be satisfactorily resolved using a shock tube, several other investigators used the technique to study many other reactions of varying complexity, covering a wid ...
Synthetic Organic Chemistry - Name
... 1.0 Principle of organolithium compounds 1.1 Preparations of organolithium compounds 1.1.1 Oxidative- Addition reaction 1.1.2 Metal-metal exchange (Transmetallation) 1.1.3 Carbanion-halide exchange 1.1.4 Metal hydrogen exchange reactions (Metallation) 1.1.5 Metal hydride addition to alkenes 1.1.6 Ha ...
... 1.0 Principle of organolithium compounds 1.1 Preparations of organolithium compounds 1.1.1 Oxidative- Addition reaction 1.1.2 Metal-metal exchange (Transmetallation) 1.1.3 Carbanion-halide exchange 1.1.4 Metal hydrogen exchange reactions (Metallation) 1.1.5 Metal hydride addition to alkenes 1.1.6 Ha ...
kcse chemistry questions
... Dilute nitric acid reacts with copper according to the equation; 3CU(s) + 8H + (aq) + 2N-3 3Cu2+ (aq) + 2NO (g) + 4H2O (l) a) What is the oxidation number of nitrogen in? i) 2N-3 ii) NO b) With respect to nitrogen, explain whether the above reaction is an oxidation or reduction process. (1mk) Explai ...
... Dilute nitric acid reacts with copper according to the equation; 3CU(s) + 8H + (aq) + 2N-3 3Cu2+ (aq) + 2NO (g) + 4H2O (l) a) What is the oxidation number of nitrogen in? i) 2N-3 ii) NO b) With respect to nitrogen, explain whether the above reaction is an oxidation or reduction process. (1mk) Explai ...
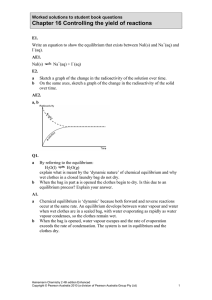
Chapter 16 Controlling the yield of reactions
... 0.220 mol of H2 and 0.110 mol of I2. a Calculate the concentration of HI in this mixture. b Another mixture was prepared by placing 4.0 mol of HI in a 2.0 L vessel at 330°C. At equilibrium 0.44 mol of H2 and 0.44 mol of I2 were present. Calculate the value of the equilibrium constant at this tempera ...
... 0.220 mol of H2 and 0.110 mol of I2. a Calculate the concentration of HI in this mixture. b Another mixture was prepared by placing 4.0 mol of HI in a 2.0 L vessel at 330°C. At equilibrium 0.44 mol of H2 and 0.44 mol of I2 were present. Calculate the value of the equilibrium constant at this tempera ...
Document
... 1. How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? 2. How many moles of KClO3 are needed to produce 5.45 moles of KCl? 3. If 10.4 moles of KCl were produced, how many moles of O2 were also produced? 4. How many moles of KCl can be produced by letting 7.5 moles of KClO3 de ...
... 1. How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? 2. How many moles of KClO3 are needed to produce 5.45 moles of KCl? 3. If 10.4 moles of KCl were produced, how many moles of O2 were also produced? 4. How many moles of KCl can be produced by letting 7.5 moles of KClO3 de ...
BSC with Chemistry CBCS Syllabus 2016-17
... 2. Determination of enthalpy of neutralization of hydrochloric acid with sodium hydroxide. 3. Determination of enthalpy of ionization of acetic acid. 4. Determination of integral enthalpy of solution of salts (KNO 3, NH4Cl). 5. Determination of enthalpy of hydration of copper sulphate. II. Ionic Equ ...
... 2. Determination of enthalpy of neutralization of hydrochloric acid with sodium hydroxide. 3. Determination of enthalpy of ionization of acetic acid. 4. Determination of integral enthalpy of solution of salts (KNO 3, NH4Cl). 5. Determination of enthalpy of hydration of copper sulphate. II. Ionic Equ ...
Rhenium- and molybdenum-catalyzed dehydration reactions
... If we want to maintain our prosperity and enable others to share in it, it is inevitable that we find a sustainable alternative for fossil feedstocks. For energy supply, many resources are available, among which solar, wind, and hydropower. For transportation fuels alternatives are also available, s ...
... If we want to maintain our prosperity and enable others to share in it, it is inevitable that we find a sustainable alternative for fossil feedstocks. For energy supply, many resources are available, among which solar, wind, and hydropower. For transportation fuels alternatives are also available, s ...
chem 102 class notes - Louisiana Tech University
... Any chemical reaction could be considered as a forward and backward reactions ) as described previously. If the rates of backward and occurring at the same time( forward reactions chemical reactions are comparable both reactants and products can coexist leading to a condition called chemical equilib ...
... Any chemical reaction could be considered as a forward and backward reactions ) as described previously. If the rates of backward and occurring at the same time( forward reactions chemical reactions are comparable both reactants and products can coexist leading to a condition called chemical equilib ...
An Analogy for an Equilibrium Reaction
... products react with each other to reform the reactants. The reaction of the reactants to form the products is called the forward reaction. The reaction of the products to form the reactants is called the reverse reaction. At some point in a chemical reaction, the rate of the forward reaction will eq ...
... products react with each other to reform the reactants. The reaction of the reactants to form the products is called the forward reaction. The reaction of the products to form the reactants is called the reverse reaction. At some point in a chemical reaction, the rate of the forward reaction will eq ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.