Modern Chemistry
... the answer as 0.571429. a. Is the setup for calculating density correct? b. How many significant figures should the answer contain? 4. It was shown in the text that in a value such as 4000 g, the precision of the number is uncertain. The zeros may or may not be significant. a. Suppose that the mass ...
... the answer as 0.571429. a. Is the setup for calculating density correct? b. How many significant figures should the answer contain? 4. It was shown in the text that in a value such as 4000 g, the precision of the number is uncertain. The zeros may or may not be significant. a. Suppose that the mass ...
TiO2-graphene oxide nanocomposite as advanced photocatalytic
... In our previous work [8], we reported on nonstoichiometric TiO2-graphene oxide nanocomposite, which was prepared by thermal hydrolysis of suspension with graphene oxide nanosheets and titania peroxo-complex. It should be mentioned that we are able to produce pure graphene nanosheets in large quantit ...
... In our previous work [8], we reported on nonstoichiometric TiO2-graphene oxide nanocomposite, which was prepared by thermal hydrolysis of suspension with graphene oxide nanosheets and titania peroxo-complex. It should be mentioned that we are able to produce pure graphene nanosheets in large quantit ...
Solving Problems: A Chemistry Handbook
... f. weight of an elephant A hypothesis is a possible explanation for what has been observed. Based on the observations of ozone thinning and CFC buildup in the atmosphere, the chemists Mario Molina and F. Sherwood Rowland hypothesized that CFCs break down in the atmosphere due to the Sun’s ultraviole ...
... f. weight of an elephant A hypothesis is a possible explanation for what has been observed. Based on the observations of ozone thinning and CFC buildup in the atmosphere, the chemists Mario Molina and F. Sherwood Rowland hypothesized that CFCs break down in the atmosphere due to the Sun’s ultraviole ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... • Consider the reaction that occurs in an automobile air bag: • 2 NaN3(s) 2Na(s) + 3N2(g) • Since there are more products than reactants, the sodium azide has decomposed into sodium metal and nitrogen gas. Stoichiometry © 2009, Prentice-Hall, Inc. ...
... • Consider the reaction that occurs in an automobile air bag: • 2 NaN3(s) 2Na(s) + 3N2(g) • Since there are more products than reactants, the sodium azide has decomposed into sodium metal and nitrogen gas. Stoichiometry © 2009, Prentice-Hall, Inc. ...
CHEM 1412. Chapter 15. Chemical Equilibrium (Homework)
... 11. Equilibrium is established for the reaction 2X(s) + Y(g) 2Z(g) at 500K, Kc = 100. Determine the concentration of Z in equilibrium with 0.2 mol X and 0.50 M Y at 500K. A. 3.2 M B. 3.5 M C. 4.5 M D. 7.1 M E. None of these. ...
... 11. Equilibrium is established for the reaction 2X(s) + Y(g) 2Z(g) at 500K, Kc = 100. Determine the concentration of Z in equilibrium with 0.2 mol X and 0.50 M Y at 500K. A. 3.2 M B. 3.5 M C. 4.5 M D. 7.1 M E. None of these. ...
Syllabus and Regulations for 2-year, 4
... Theoretical-50 Marks and Group-B: Practical- 25/30/35/40/50-Marks) each. The examiners shall forward assessment in respect of every candidate to the Principal / Controller of Examination / Coordinator P. G. Courses (as the case may be) for tabulation of the results. 3.(a) The entire course of 1000 m ...
... Theoretical-50 Marks and Group-B: Practical- 25/30/35/40/50-Marks) each. The examiners shall forward assessment in respect of every candidate to the Principal / Controller of Examination / Coordinator P. G. Courses (as the case may be) for tabulation of the results. 3.(a) The entire course of 1000 m ...
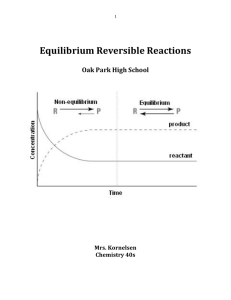
Chemical Equilibrium
... Once equilibrium is established, the reaction is over, right? Not exactly. An experimenter has some ability to affect the equilibrium. Chemical equilibria can be shifted by changing the conditions that the system experiences. We say that we “stress” the equilibrium. When we stress the equilibrium, th ...
... Once equilibrium is established, the reaction is over, right? Not exactly. An experimenter has some ability to affect the equilibrium. Chemical equilibria can be shifted by changing the conditions that the system experiences. We say that we “stress” the equilibrium. When we stress the equilibrium, th ...
Stoichiometery
... A more typical problem 2 H2 + O2 → 2 H2O What masses of oxygen and hydrogen are required to create 5.0 g H2O? 5.0 g H2O * 1 mol H2O * 1 mol O2 * 32.0 g O2 = 4.44 g O2 18.016 g H2O 2 mol H2O 1 mol O2 5.0 g H2O * 1 mol H2O * 2 mol H2 * 2.016 g H2 = 0.56 g H2 18.016 g H2O 2 mol H2O 1 mol H2 ...
... A more typical problem 2 H2 + O2 → 2 H2O What masses of oxygen and hydrogen are required to create 5.0 g H2O? 5.0 g H2O * 1 mol H2O * 1 mol O2 * 32.0 g O2 = 4.44 g O2 18.016 g H2O 2 mol H2O 1 mol O2 5.0 g H2O * 1 mol H2O * 2 mol H2 * 2.016 g H2 = 0.56 g H2 18.016 g H2O 2 mol H2O 1 mol H2 ...
Inorganic Chemistry
... before Chapters 5 or 6. Chapter 6 contains material dealing with intermolecular forces and polarity of molecules because of the importance of these topics when interpreting properties of substances and their chemical behavior. In view of the importance of the topic, especially in industrial chemistr ...
... before Chapters 5 or 6. Chapter 6 contains material dealing with intermolecular forces and polarity of molecules because of the importance of these topics when interpreting properties of substances and their chemical behavior. In view of the importance of the topic, especially in industrial chemistr ...
FREE Sample Here
... protein that speeds up chemical reactions without being changed or depleted protein that functions as a hormone protein that inhibits chemical reactions by being changed or depleted fibrous protein that is part of certain tissues in the body ...
... protein that speeds up chemical reactions without being changed or depleted protein that functions as a hormone protein that inhibits chemical reactions by being changed or depleted fibrous protein that is part of certain tissues in the body ...
Chapter 5: Calculations and the Chemical Equation
... 1. If a reactant or product is a solid, (s) and liquid, (l) is placed after the formula. 2. If a reactant or product is a gas, (g) is placed after it. 3. If a reactant or product is in water solution, (aq) is placed after it. A Recipe for Chemical Change A chemical equation is similar to a cookbook ...
... 1. If a reactant or product is a solid, (s) and liquid, (l) is placed after the formula. 2. If a reactant or product is a gas, (g) is placed after it. 3. If a reactant or product is in water solution, (aq) is placed after it. A Recipe for Chemical Change A chemical equation is similar to a cookbook ...
ANNEX (Manuscrits posteriors a la Comissió de Doctorat de Juliol del...
... proceed and avoid unreacted di-iodo starting material a high ratio (2.5:1) of the Grignard reagent per iodine in [2]- has to be used. When the ratio is lowered to 1.25:1 a large proportion of unreacted starting material is recovered. This suggest that other reactions consuming CH3MgBr or CH3CH2MgBr ...
... proceed and avoid unreacted di-iodo starting material a high ratio (2.5:1) of the Grignard reagent per iodine in [2]- has to be used. When the ratio is lowered to 1.25:1 a large proportion of unreacted starting material is recovered. This suggest that other reactions consuming CH3MgBr or CH3CH2MgBr ...
Chemistry In action
... and a step-by-step approach that students can follow. Over the years, more than three million students have learned chemistry using a text by Morris Hein. In addition to Foundations of College Chemistry, Fourteenth Edition, he is co-author of Introduction to General, Organic, and Biochemistry, Tenth ...
... and a step-by-step approach that students can follow. Over the years, more than three million students have learned chemistry using a text by Morris Hein. In addition to Foundations of College Chemistry, Fourteenth Edition, he is co-author of Introduction to General, Organic, and Biochemistry, Tenth ...
Stoichiometry worksheet KEY
... c) How many grams of H2O are produced when 52.0 g of C2H2 burn? 36.0 g H2O d) How many grams of O2 are required to completely burn 52.0 g of C2H2? 160 g O2 e) Use the answers from questions b, c, and d above to show that this equation obeys the law of conservation of mass. Mass of reactants = mass o ...
... c) How many grams of H2O are produced when 52.0 g of C2H2 burn? 36.0 g H2O d) How many grams of O2 are required to completely burn 52.0 g of C2H2? 160 g O2 e) Use the answers from questions b, c, and d above to show that this equation obeys the law of conservation of mass. Mass of reactants = mass o ...
Chemistry Honours - SCS Autonomous College
... First law: Concept of heat, q, work, w, internal energy, U, and statement of first law; enthalpy, H, relation between heat capacities, calculations of q, w, U and H for reversible, irreversible and free expansion of gases (ideal and van der Waals) under isothermal and adiabatic conditions. Thermoche ...
... First law: Concept of heat, q, work, w, internal energy, U, and statement of first law; enthalpy, H, relation between heat capacities, calculations of q, w, U and H for reversible, irreversible and free expansion of gases (ideal and van der Waals) under isothermal and adiabatic conditions. Thermoche ...
Document
... increased to 2.0 liters, while the temperature was kept at 300 K. This will result in: A) an increase in Kp. B) a decrease in Kp. C) a shift in the equilibrium position to the right. D) a shift in the equilibrium position to the left. E) none of these ...
... increased to 2.0 liters, while the temperature was kept at 300 K. This will result in: A) an increase in Kp. B) a decrease in Kp. C) a shift in the equilibrium position to the right. D) a shift in the equilibrium position to the left. E) none of these ...
Specification and sample assessment material - Edexcel
... The best way to develop practical and investigative skills is to embed practical activities in your teaching of theory. The development of knowledge and skills can happen together, leading to secure acquisition of knowledge and skills. There are some practicals in the specification content, which st ...
... The best way to develop practical and investigative skills is to embed practical activities in your teaching of theory. The development of knowledge and skills can happen together, leading to secure acquisition of knowledge and skills. There are some practicals in the specification content, which st ...
Chapter 1 – Reaction Kinetics Answer Key
... water will occur as water is formed in a reaction that occurs in aqueous solution. This is, of course, nonsense! As the entire reaction occurs in the solvent water, there will simply be a ...
... water will occur as water is formed in a reaction that occurs in aqueous solution. This is, of course, nonsense! As the entire reaction occurs in the solvent water, there will simply be a ...
General and Inorganic Chemistry – Laboratory Techniques
... substance is a homogeneous material consisting of one particular kind of matter. A mixture is a material that can be separated by physical means into two or more substances. A substance is a kind of matter that cannot be separated into other kinds of matter by any physical process. Substances can be ...
... substance is a homogeneous material consisting of one particular kind of matter. A mixture is a material that can be separated by physical means into two or more substances. A substance is a kind of matter that cannot be separated into other kinds of matter by any physical process. Substances can be ...
Lecture 1 and 2 Volumetric analysis Zuhair Khammas
... alkalimetry, while for base is called acidimetry . (A) Alkalimetry ...
... alkalimetry, while for base is called acidimetry . (A) Alkalimetry ...
BS Chemistry - Government College University Faisalabad
... General characteristics of the following group of p-block elements with reference to the aspects given against each Carbon and Silicon: Group anomalies. Allotropic forms of carbon, fullerenes and their applications. Production of pure silicon for solar energy and silicon chips, silicates and silicon ...
... General characteristics of the following group of p-block elements with reference to the aspects given against each Carbon and Silicon: Group anomalies. Allotropic forms of carbon, fullerenes and their applications. Production of pure silicon for solar energy and silicon chips, silicates and silicon ...
Chemistry 30 June 2001 Grade 12 Diploma Examination
... greater than it is during cellular respiration because the production of H2O(g) releases more energy than does the production of H2O(l) less than it is during cellular respiration because the production of H2O(g) releases less energy than does the production of H2O(l) the same as it is in the body b ...
... greater than it is during cellular respiration because the production of H2O(g) releases more energy than does the production of H2O(l) less than it is during cellular respiration because the production of H2O(g) releases less energy than does the production of H2O(l) the same as it is in the body b ...
Review of N and Metal co-Doped TiO for Water Purification under
... nitrogen [7], sulfur [8], fluorine [9], iodine [10] and boron [11] into the TiO2 bulk to successfully cause the red shift of the absorption onset of TiO2 to the visible region and enhance the photocatalytic activity. Among these elements, nitrogen was thought as the most effective substitution doping ...
... nitrogen [7], sulfur [8], fluorine [9], iodine [10] and boron [11] into the TiO2 bulk to successfully cause the red shift of the absorption onset of TiO2 to the visible region and enhance the photocatalytic activity. Among these elements, nitrogen was thought as the most effective substitution doping ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.