Plasma Treatment for Environment Protection
... keeping our economy running, also has a significant share in both to the air and water pollution. The production of various commodities, as well as livestock and other agricultural activities is an additional source of pollution with a very large variety of pollutants. The pollution from these sourc ...
... keeping our economy running, also has a significant share in both to the air and water pollution. The production of various commodities, as well as livestock and other agricultural activities is an additional source of pollution with a very large variety of pollutants. The pollution from these sourc ...
CHM 423 Coordination Chemistry
... Coordination chemistry is the foundation of modern inorganic and bioinorganic chemistry, both of which have contributed immensely to the development of the chemical industry and medicine. The knowledge of coordination chemistry has provided insight into the mode of actions (kinetics and mechanisms) ...
... Coordination chemistry is the foundation of modern inorganic and bioinorganic chemistry, both of which have contributed immensely to the development of the chemical industry and medicine. The knowledge of coordination chemistry has provided insight into the mode of actions (kinetics and mechanisms) ...
Recent Developments on the Mechanism and Kinetics
... concentration of the catalyst and oleic acid, and temperature on the reaction rate were investigated. A related esterification mechanism was presented and described as follows: in presence of Sn2+( SnCl2•2H2O) catalyst, the carbonyl of the fatty acid is polarized to activate of substrate, which make ...
... concentration of the catalyst and oleic acid, and temperature on the reaction rate were investigated. A related esterification mechanism was presented and described as follows: in presence of Sn2+( SnCl2•2H2O) catalyst, the carbonyl of the fatty acid is polarized to activate of substrate, which make ...
chapter 21
... Strategy: We are given information as to how the concentrations of X2, Y, and Z affect the rate of the reaction and are asked to determine the rate law. We assume that the rate law takes the form rate k[X2]x[Y]y[Z]z How do we use the information to determine x, y, and z? Solution: Since the reacti ...
... Strategy: We are given information as to how the concentrations of X2, Y, and Z affect the rate of the reaction and are asked to determine the rate law. We assume that the rate law takes the form rate k[X2]x[Y]y[Z]z How do we use the information to determine x, y, and z? Solution: Since the reacti ...
odd - WWW2
... carbides react with water to produce the appropriate hydrocarbon. Covalent carbides are formed by nonmetals, specifically boron and silicon, more electronegative than carbon. These carbides are very hard and have high melting points. The metallic carbides are interstitial carbides, in that the carbo ...
... carbides react with water to produce the appropriate hydrocarbon. Covalent carbides are formed by nonmetals, specifically boron and silicon, more electronegative than carbon. These carbides are very hard and have high melting points. The metallic carbides are interstitial carbides, in that the carbo ...
CHAPTER 14 CHEMICAL KINETICS
... Strategy: The relationship between the concentration of a reactant at different times in a first-order reaction is given by Equations (14.3) and (14.4) of the text. We are asked to determine the time required for 95% of the phosphine to decompose. If we initially have 100% of the compound and 95% ha ...
... Strategy: The relationship between the concentration of a reactant at different times in a first-order reaction is given by Equations (14.3) and (14.4) of the text. We are asked to determine the time required for 95% of the phosphine to decompose. If we initially have 100% of the compound and 95% ha ...
Chapter 12
... You are given moles of the reactant propane, and moles of the product carbon dioxide must be found. The balanced chemical equation must be written. Conversion from moles of C3H8 to moles of CO2 is required. The correct mole ratio has moles of unknown substance in the numerator and moles of known sub ...
... You are given moles of the reactant propane, and moles of the product carbon dioxide must be found. The balanced chemical equation must be written. Conversion from moles of C3H8 to moles of CO2 is required. The correct mole ratio has moles of unknown substance in the numerator and moles of known sub ...
Catalytic oxidation of ammonia to nitrogen
... acidification of the environment. NOx and SOx are converted in the atmosphere to give nitric and sulphuric acid. However emission of ammonia causes acidification of the environment in an indirect way. Reaction of ammonia with acidic aerosols in the atmosphere, such as aerosols of sulphuric acid or n ...
... acidification of the environment. NOx and SOx are converted in the atmosphere to give nitric and sulphuric acid. However emission of ammonia causes acidification of the environment in an indirect way. Reaction of ammonia with acidic aerosols in the atmosphere, such as aerosols of sulphuric acid or n ...
Version A
... (1) Test Questions are Copyright © 1984-2002 by College Entrance Examination Board, Princeton, NJ. All rights reserved. For face-to-face teaching purposes, classroom teachers are permitted to reproduce the questions. Web or Mass distribution prohibited. (2) AP® is a registered trademark of the Colle ...
... (1) Test Questions are Copyright © 1984-2002 by College Entrance Examination Board, Princeton, NJ. All rights reserved. For face-to-face teaching purposes, classroom teachers are permitted to reproduce the questions. Web or Mass distribution prohibited. (2) AP® is a registered trademark of the Colle ...
Mole Concept
... contains as many elementary entities (atoms, molecules, or other particles) as there are atoms in exactly ...
... contains as many elementary entities (atoms, molecules, or other particles) as there are atoms in exactly ...
Abdullah F. Eid
... The octahedra are arranged in four M3O13 groups. Each group is formed by three edges sharing octahedra and having a common oxygen atom which is also shared with the central tetrahedron XO4 . Among a wide variety of HPAs, the Keggin’s are the most stable and more easily available; these are the most ...
... The octahedra are arranged in four M3O13 groups. Each group is formed by three edges sharing octahedra and having a common oxygen atom which is also shared with the central tetrahedron XO4 . Among a wide variety of HPAs, the Keggin’s are the most stable and more easily available; these are the most ...
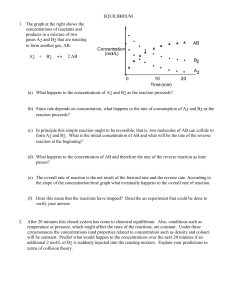
15.0 EquilibriumIHS2014
... The rate of reaction of the reactants decreases as the number of reactant molecules decrease. The rate at which the product turns back to reactants increases as the number of product molecules increases. These two rates become equal at some point, after which the quantity of each will not change. ...
... The rate of reaction of the reactants decreases as the number of reactant molecules decrease. The rate at which the product turns back to reactants increases as the number of product molecules increases. These two rates become equal at some point, after which the quantity of each will not change. ...
EQUILIBRIUM - SCH4U1-CCVI
... (a) Line up five clean test tubes all of the same diameter, and label them. Add 5.0 mL of 0.002 mol/L potassium thiocyanate solution to each of these five test tubes. To test tube (1) add 5.0 mL of 0.2 mol/L iron (III) nitrate solution. This tube will be used as the standard. (b) Measure 10.0 mL of ...
... (a) Line up five clean test tubes all of the same diameter, and label them. Add 5.0 mL of 0.002 mol/L potassium thiocyanate solution to each of these five test tubes. To test tube (1) add 5.0 mL of 0.2 mol/L iron (III) nitrate solution. This tube will be used as the standard. (b) Measure 10.0 mL of ...
Chemistry Science Notebook: Student Edition
... addresses how students understand difficult concepts and content in school. Glencoe/McGrawHill has developed the Science Notebook for science students based upon that research. Evidence indicates that students need to know how to take notes, use graphic organizers, learn vocabulary, and develop thei ...
... addresses how students understand difficult concepts and content in school. Glencoe/McGrawHill has developed the Science Notebook for science students based upon that research. Evidence indicates that students need to know how to take notes, use graphic organizers, learn vocabulary, and develop thei ...
PREPARATION, STRUCTURAL STUDIES AND CHEMICAL
... Preparation methods for hypervalent iodine reagents can be generally described as an oxidation of organic iodide by appropriate oxidizer resulting in a hypervalent iodine compound with, if necessary, an optional following step of ligand-exchange reaction. There are many useful oxidizing agents for t ...
... Preparation methods for hypervalent iodine reagents can be generally described as an oxidation of organic iodide by appropriate oxidizer resulting in a hypervalent iodine compound with, if necessary, an optional following step of ligand-exchange reaction. There are many useful oxidizing agents for t ...
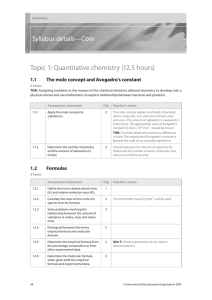
2016-2018 Syllabus - Cambridge International Examinations
... grade band. They give a general indication of the required standard at each specified grade. The descriptions should be interpreted in relation to the content outlined in the syllabus; they are not designed to define that content. The grade awarded will depend in practice upon the extent to which th ...
... grade band. They give a general indication of the required standard at each specified grade. The descriptions should be interpreted in relation to the content outlined in the syllabus; they are not designed to define that content. The grade awarded will depend in practice upon the extent to which th ...
PHOSPHORUS AND SULFUR COSMOCHEMISTRY
... phosphorus compounds. Schreibersite corrosion was studied by a variety of techniques, including NMR, MS, XRD, and EPR. Reduced phosphorus in schreibersite corrodes through release of phosphite radicals which react with other radicals to form the phosphorus compounds observed. These radicals are also ...
... phosphorus compounds. Schreibersite corrosion was studied by a variety of techniques, including NMR, MS, XRD, and EPR. Reduced phosphorus in schreibersite corrodes through release of phosphite radicals which react with other radicals to form the phosphorus compounds observed. These radicals are also ...
Brief Contents - Educhimica.it
... 5. a. Because water is going from a liquid phase to a gas phase, the change is a physical change. b. Because of the food, chemical changes are occurring in the digestive system. This is a chemical change. c. Because alcohol is going from a liquid phase to a solid phase, the change is a physical chan ...
... 5. a. Because water is going from a liquid phase to a gas phase, the change is a physical change. b. Because of the food, chemical changes are occurring in the digestive system. This is a chemical change. c. Because alcohol is going from a liquid phase to a solid phase, the change is a physical chan ...
Chapter 5 Geochemical Weathering
... Weathering of landscapes involves an array of mechanical and geochemical agents that conspire to alter primary geological formations to sediments and solutes. Geochemical weathering is driven by water. In soils, water is the limiting factor for the activity of aerobic bacteria that degrade organics ...
... Weathering of landscapes involves an array of mechanical and geochemical agents that conspire to alter primary geological formations to sediments and solutes. Geochemical weathering is driven by water. In soils, water is the limiting factor for the activity of aerobic bacteria that degrade organics ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.