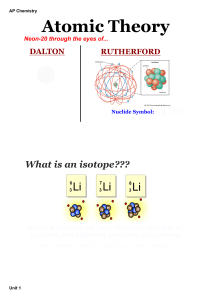
How many grams of oxygen are made if 3.75 moles of KClO 3
... 26. When steam (water vapor) and carbon react, carbon monoxide and hydrogen are produced. What weights of carbon monoxide will be formed by the action of 250000 g of steam on carbon? Write the chemical equation first. ...
... 26. When steam (water vapor) and carbon react, carbon monoxide and hydrogen are produced. What weights of carbon monoxide will be formed by the action of 250000 g of steam on carbon? Write the chemical equation first. ...
CHEMISTRY 102B Name Hour Exam II March 19, 2015 Signature
... 1.0 mol of helium gas at 25 °C. 1.0 mol of helium gas at 125 °C. 1.0 mol of oxygen gas at 25 °C. 1.0 mol of oxygen gas at 125 °C. Choices a and b have equal collision rates, and these are higher than those in c and d. ...
... 1.0 mol of helium gas at 25 °C. 1.0 mol of helium gas at 125 °C. 1.0 mol of oxygen gas at 25 °C. 1.0 mol of oxygen gas at 125 °C. Choices a and b have equal collision rates, and these are higher than those in c and d. ...
Honors Chemistry Final Review
... Classify each of the following bonds as ionic, polar-covalent, nonpolar covalent or metallic bonds. a. Na – Cl b. C – Cl c. C – C d. C – O e. In Na f. Na – OH g. Which pair of ions forms the most ionic bond: K to Cl or Fr to F? ...
... Classify each of the following bonds as ionic, polar-covalent, nonpolar covalent or metallic bonds. a. Na – Cl b. C – Cl c. C – C d. C – O e. In Na f. Na – OH g. Which pair of ions forms the most ionic bond: K to Cl or Fr to F? ...
Part I Power generation in fuel cells
... outside the cell rather than forming an integral part of its construction. A fundamental but important example of a fuel cell is the hydrogen/oxygen cell. If the electrolyte is an acid, the reduction reaction at the cathode is O2 + 4H+ + 4e- 2H2O At the anode the reaction is 2H2 4H+ + 4eOnce pro ...
... outside the cell rather than forming an integral part of its construction. A fundamental but important example of a fuel cell is the hydrogen/oxygen cell. If the electrolyte is an acid, the reduction reaction at the cathode is O2 + 4H+ + 4e- 2H2O At the anode the reaction is 2H2 4H+ + 4eOnce pro ...
ICSE Board Class X Chemistry Board Paper – 2015
... Second, on diluting sodium aluminate with water and cooling to 50°C, sodium aluminate is hydrolysed to give aluminium hydroxide as precipitate. Here, the impurities dissolve in sodium hydroxide. (3) Graphite: Thick rods of graphite are suspended into the fused electrolyte. They act as an anode where ...
... Second, on diluting sodium aluminate with water and cooling to 50°C, sodium aluminate is hydrolysed to give aluminium hydroxide as precipitate. Here, the impurities dissolve in sodium hydroxide. (3) Graphite: Thick rods of graphite are suspended into the fused electrolyte. They act as an anode where ...
ic199p5a
... (b) Explain why KNO3(s) is highly soluble in water despite this observation regarding the temperature change on solution. If it is readily soluble, the G for solution is likely to be negative, which means the increase in entropy on dissolution (the TS term) is sufficient to overcome the positive ...
... (b) Explain why KNO3(s) is highly soluble in water despite this observation regarding the temperature change on solution. If it is readily soluble, the G for solution is likely to be negative, which means the increase in entropy on dissolution (the TS term) is sufficient to overcome the positive ...
study guide and review for first semester final
... 10. Use data obtained from quantitative analysis for the composition of a compound or from percentage composition to determine the compound’s empirical formula. Ex. A compound with 0.90 g Ca and 1.6 g Cl has what empirical formula? (CaCl2) Ex. A white powder used in paints, enamels and ceramics has ...
... 10. Use data obtained from quantitative analysis for the composition of a compound or from percentage composition to determine the compound’s empirical formula. Ex. A compound with 0.90 g Ca and 1.6 g Cl has what empirical formula? (CaCl2) Ex. A white powder used in paints, enamels and ceramics has ...
Chemistry Final Exam Review 2006-2007
... triple point, critical point, the melting point /freezing point line and the boiling point/condensation point line. Also label the 3 sections as solid , liquid, and gas. 5. Know how to read a heating and cooling curve. What do the plateaus tells you? What do the slopes tell you? Where is the KE of t ...
... triple point, critical point, the melting point /freezing point line and the boiling point/condensation point line. Also label the 3 sections as solid , liquid, and gas. 5. Know how to read a heating and cooling curve. What do the plateaus tells you? What do the slopes tell you? Where is the KE of t ...
(Tungsten)–Nickel (Cobalt) Alloys and Intermetallic Compounds
... For the cathodic codeposition of metals, the correspondence of their crystalline lattices and the difference in their standard electrode potentials are of great importance. It seems significant from a practical point of view to study the electrodeposition of alloys whose components have crystalline ...
... For the cathodic codeposition of metals, the correspondence of their crystalline lattices and the difference in their standard electrode potentials are of great importance. It seems significant from a practical point of view to study the electrodeposition of alloys whose components have crystalline ...
Electrochem 1 - GCG-42
... Electron transfer reactions are oxidation-reduction or redox reactions. Therefore, this field is often called ELECTROCHEMISTRY. ...
... Electron transfer reactions are oxidation-reduction or redox reactions. Therefore, this field is often called ELECTROCHEMISTRY. ...
HONORS: UNIT 2B: Antacids Below are the class objectives
... reaction using the reference tables Write and balance ionic & net ionic equations** Investigate variables that affect the rate of a reaction (nature of reactants, concentration, surface area, and temperature Explain that nature of reactants can refer to their complexity and the number of bonds that ...
... reaction using the reference tables Write and balance ionic & net ionic equations** Investigate variables that affect the rate of a reaction (nature of reactants, concentration, surface area, and temperature Explain that nature of reactants can refer to their complexity and the number of bonds that ...
Stuff Matters Handout
... Matter is everything around you. Matter is anything made of atoms and molecules. Matter is anything that has mass and takes up space. If you are new to the idea of mass, it is the amount of stuff in an object. Matter is sometimes related to light and electromagnetic radiation. Even though matter can ...
... Matter is everything around you. Matter is anything made of atoms and molecules. Matter is anything that has mass and takes up space. If you are new to the idea of mass, it is the amount of stuff in an object. Matter is sometimes related to light and electromagnetic radiation. Even though matter can ...
1 Q. If ΔrH is positive, what can you say about the reaction? 2 Q If
... down (and so its container would get cold) because it is absorbing energy from its surroundings. ...
... down (and so its container would get cold) because it is absorbing energy from its surroundings. ...
Dear Chemistry Student, I am excited that you have chosen to
... molecular shape, bond angles and formal charges for each and resonance where necessary. a. NH3 b. SO42- c. CCl4 d. NO3-1 e. SF6 f. XeF4 g. H2S h. CO2 i. O2 j. BF3 k. CClF3 l. XeF2 m. CO32- n. NH4+1 o. PF5 Week 4: The Mole/Stoich/LR/ER/%yield/%comp/EF/MF 9. Answer the questions below using the equati ...
... molecular shape, bond angles and formal charges for each and resonance where necessary. a. NH3 b. SO42- c. CCl4 d. NO3-1 e. SF6 f. XeF4 g. H2S h. CO2 i. O2 j. BF3 k. CClF3 l. XeF2 m. CO32- n. NH4+1 o. PF5 Week 4: The Mole/Stoich/LR/ER/%yield/%comp/EF/MF 9. Answer the questions below using the equati ...
Introduction
... The ammonia-water system has many applications in industrial processes and environmental systems. For this reason, its behavior has been the interest subject. Ammonia in aqueous phase and it is difficult to describe in an weak electrolyte. To overcome this difficulty, typically, the pH of the soluti ...
... The ammonia-water system has many applications in industrial processes and environmental systems. For this reason, its behavior has been the interest subject. Ammonia in aqueous phase and it is difficult to describe in an weak electrolyte. To overcome this difficulty, typically, the pH of the soluti ...
Solution - ZOMUedu
... ■ 1 M = 1 mol solute/ liter solution ○ Dilution = adding more solvent to a known solution ■ The moles of solute stay the same ■ M1V1 = M2V2 ○ Stock solution = a solution of known concentration that is used to make more dilute solutions Precipitation ○ Precipitation = when aqueous solutions of ionic ...
... ■ 1 M = 1 mol solute/ liter solution ○ Dilution = adding more solvent to a known solution ■ The moles of solute stay the same ■ M1V1 = M2V2 ○ Stock solution = a solution of known concentration that is used to make more dilute solutions Precipitation ○ Precipitation = when aqueous solutions of ionic ...
Chemistry Final Exam Review
... • activated complex, activation energy, heat of reaction (∆H) - exothermic vs. endothermic • enthalpy (∆H) - exothermic vs. endothermic ...
... • activated complex, activation energy, heat of reaction (∆H) - exothermic vs. endothermic • enthalpy (∆H) - exothermic vs. endothermic ...
Spring 2001 - TAMU Chemistry
... Directions: (1) Put your name, S.I.D. number and signature on the free response part of the exam where indicated. (2) Each multiple choice question is actually 2 questions on your scanning sheet. If you are sure of an answer, put the same answer down for both questions for 5 pts. If you cannot decid ...
... Directions: (1) Put your name, S.I.D. number and signature on the free response part of the exam where indicated. (2) Each multiple choice question is actually 2 questions on your scanning sheet. If you are sure of an answer, put the same answer down for both questions for 5 pts. If you cannot decid ...
Quantities, Units, Symbols and Nomenclature used in
... The term ‘number of moles’ is to be avoided in favour of the ‘amount of substance in moles’. In the same manner, the size of an object can be described in terms of its ‘length in metres’, rather than its ‘number of metres’. Graph Axes and Table Headings Labelled as quantity / unit, eg c / mol L–1. O ...
... The term ‘number of moles’ is to be avoided in favour of the ‘amount of substance in moles’. In the same manner, the size of an object can be described in terms of its ‘length in metres’, rather than its ‘number of metres’. Graph Axes and Table Headings Labelled as quantity / unit, eg c / mol L–1. O ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.