Chapter 3 Part 2 Review
... photosynthesis in the forest. One morning, it wakes up with dew on its leaves. The sunshine immediately begins to evaporate the dew. Suddenly, a storm rolls in and a giant wind knocks the tree to the ground. A limb is struck by lighting and partially burns. Over the next year, the remaining parts of ...
... photosynthesis in the forest. One morning, it wakes up with dew on its leaves. The sunshine immediately begins to evaporate the dew. Suddenly, a storm rolls in and a giant wind knocks the tree to the ground. A limb is struck by lighting and partially burns. Over the next year, the remaining parts of ...
Mid Term Exam Topics 1-5 solution - OCW
... For water, the density of the solid is smaller than for the liquid so the slope is negative. But this is not the case for carbon dioxide which has a positive slope. c) From 218 to 1 atm, water exists as a liquid. At 1 atm, it transforms in solid and remains as a solid until pressure reaches a value ...
... For water, the density of the solid is smaller than for the liquid so the slope is negative. But this is not the case for carbon dioxide which has a positive slope. c) From 218 to 1 atm, water exists as a liquid. At 1 atm, it transforms in solid and remains as a solid until pressure reaches a value ...
Physical and Chemical change: Introduction
... During a chemical change, the particles themselves are changed in some way. In the example of copper (II) chloride that was used earlier, the CuCl2 molecules were split up into their component atoms. The number of particles will change because each CuCl2 molecule breaks down into one copper atom (Cu ...
... During a chemical change, the particles themselves are changed in some way. In the example of copper (II) chloride that was used earlier, the CuCl2 molecules were split up into their component atoms. The number of particles will change because each CuCl2 molecule breaks down into one copper atom (Cu ...
In organic chemistry, we studied a lot about the essential elements
... and regulate the rate of metabolism in various tissues. However, recently researches has shown that Prostaglandins is responsible for lower blood pressure, influence platelet aggregation during blood clotting, lower extent of gastric secretions, pain and swelling accompanied inflammation. Nucleic a ...
... and regulate the rate of metabolism in various tissues. However, recently researches has shown that Prostaglandins is responsible for lower blood pressure, influence platelet aggregation during blood clotting, lower extent of gastric secretions, pain and swelling accompanied inflammation. Nucleic a ...
Introduction(s)
... Binary aqueous acid compounds (recognized because hydrogen is the first element that is combined with an ide ion) are named by using the pattern: hydro-(root word of negative element)-ic acid ...
... Binary aqueous acid compounds (recognized because hydrogen is the first element that is combined with an ide ion) are named by using the pattern: hydro-(root word of negative element)-ic acid ...
Memorization?
... Binary aqueous acid compounds (recognized because hydrogen is the first element that is combined with an ide ion) are named by using the pattern: hydro-(root word of negative element)-ic acid ...
... Binary aqueous acid compounds (recognized because hydrogen is the first element that is combined with an ide ion) are named by using the pattern: hydro-(root word of negative element)-ic acid ...
Honors Chemistry- Chapter 16 Homework Packet Reaction Energy
... starts at a temperature of 45°C, what will the final temperature of the copper metal be? (Cp (Cu) = 0.385 J/g°C). ...
... starts at a temperature of 45°C, what will the final temperature of the copper metal be? (Cp (Cu) = 0.385 J/g°C). ...
urbano, mariajose
... Holism: The principle that a higher level of order cannot be meaningfully explained by examining component parts in isolation. • An organism is a living whole greater than the sum of its parts. • For example, a cell dismantled to its chemical ingredients is no longer a cell. 4. Describe seven emerge ...
... Holism: The principle that a higher level of order cannot be meaningfully explained by examining component parts in isolation. • An organism is a living whole greater than the sum of its parts. • For example, a cell dismantled to its chemical ingredients is no longer a cell. 4. Describe seven emerge ...
IPC Semester Exam Review – Chemistry Topics
... Questions will include multiple-choice and matching. You will need a calculator and a pencil for the Scantron form. A periodic table and conversion chart will be provided. The Nature of Science Identify each of the following examples as PURE or APPLIED sciences. 1. Development of the computer ch ...
... Questions will include multiple-choice and matching. You will need a calculator and a pencil for the Scantron form. A periodic table and conversion chart will be provided. The Nature of Science Identify each of the following examples as PURE or APPLIED sciences. 1. Development of the computer ch ...
Department of Chemistry, IIT-Delhi CY110N Tutorial
... system, What is the entropy change for the combined system? Is the process reversible or irreversible? Why? 8. One mol of hydrogen occupies a volume of 0.1 m3 at 300K and one mol of argon also occupies the same volume but at 400K. While isolated from their surroundings, each undergoes a free expansi ...
... system, What is the entropy change for the combined system? Is the process reversible or irreversible? Why? 8. One mol of hydrogen occupies a volume of 0.1 m3 at 300K and one mol of argon also occupies the same volume but at 400K. While isolated from their surroundings, each undergoes a free expansi ...
Chapter 8 Thermochemistry: Chemical Energy
... Thermal Energy: The kinetic energy of molecular motion and is measured by finding the temperature of an object Heat: The amount of thermal energy transferred from one object to another as the result of a temperature difference between the two ...
... Thermal Energy: The kinetic energy of molecular motion and is measured by finding the temperature of an object Heat: The amount of thermal energy transferred from one object to another as the result of a temperature difference between the two ...
Revision exam - Dynamic Science
... c) Carbon dioxide does not support combustion. d) Carbon dioxide is not used by green plants and therefore accumulates in closed environments. ...
... c) Carbon dioxide does not support combustion. d) Carbon dioxide is not used by green plants and therefore accumulates in closed environments. ...
Chapter 2: Matter
... nucleus of an atoms is made of _________ &___________. The _______________ of an atom are in constant motion around the nucleus. ...
... nucleus of an atoms is made of _________ &___________. The _______________ of an atom are in constant motion around the nucleus. ...
Complete the following equations
... The concentration of Mg2+ in seawater is about 0.055 M. (a) How many liters of seawater will produce 1.00 kg of magnesium. (b) How many kilograms of calcium oxide, CaO, must be added to the seawater sample of part (a) in order to precipitate all of Mg2+ as magnesium hydroxide. (c) Write a balanced e ...
... The concentration of Mg2+ in seawater is about 0.055 M. (a) How many liters of seawater will produce 1.00 kg of magnesium. (b) How many kilograms of calcium oxide, CaO, must be added to the seawater sample of part (a) in order to precipitate all of Mg2+ as magnesium hydroxide. (c) Write a balanced e ...
CHEM 101 Final (Term 151)
... 34. Which one of the following statements is TRUE? A) The magnetic quantum number (ml) describes the orientation of an orbital. B) The principal quantum number (n) describes the shape of an orbital. C) The principal quantum number (n) describes the orientation of an orbital. D) The angular momentum ...
... 34. Which one of the following statements is TRUE? A) The magnetic quantum number (ml) describes the orientation of an orbital. B) The principal quantum number (n) describes the shape of an orbital. C) The principal quantum number (n) describes the orientation of an orbital. D) The angular momentum ...
Chemistry Standards Review
... 57. Describe the dissolving process at the molecular level by using the concept of random molecular motion. 58. What three factors affect the dissolving process? 59. When 5.20 g of salt is added to 5000 g of water, what is the concentration in parts per million (ppm)? 60. How many grams of KOH would ...
... 57. Describe the dissolving process at the molecular level by using the concept of random molecular motion. 58. What three factors affect the dissolving process? 59. When 5.20 g of salt is added to 5000 g of water, what is the concentration in parts per million (ppm)? 60. How many grams of KOH would ...
Chem 430 - Fall 1994
... K at constant pressure given the following equation for the temperature dependence of the molar heat capacity of N2(g) and that the HVAP and Tb are 5.586 kJ/mol and 77.35K? Cpo = 26.9835 + 0.0059622 T - 3.377 x 10-7 T2 (J K-1mol-1) ...
... K at constant pressure given the following equation for the temperature dependence of the molar heat capacity of N2(g) and that the HVAP and Tb are 5.586 kJ/mol and 77.35K? Cpo = 26.9835 + 0.0059622 T - 3.377 x 10-7 T2 (J K-1mol-1) ...
Matter 1. ______ is anything that has ______ and takes up ______
... 7. Classification of Matter – matter can be classified by its physical and chemical properties. a _______________ _________________– a characteristic of a substance that can be observed without changing the substance into another substance. Examples: physical state (solid, liquid, gas) electrical an ...
... 7. Classification of Matter – matter can be classified by its physical and chemical properties. a _______________ _________________– a characteristic of a substance that can be observed without changing the substance into another substance. Examples: physical state (solid, liquid, gas) electrical an ...
Full answers
... • Explain, in terms of chemical bonding and intermolecular forces, the following trend in melting points: CH4 < I2 < NaCl < silica (SiO2) There are only dispersion forces between the molecules in CH4 and I2. The I atom is a large, many-electron atom so its electron cloud is more easily polarised tha ...
... • Explain, in terms of chemical bonding and intermolecular forces, the following trend in melting points: CH4 < I2 < NaCl < silica (SiO2) There are only dispersion forces between the molecules in CH4 and I2. The I atom is a large, many-electron atom so its electron cloud is more easily polarised tha ...
Chemistry Standards Review
... (B) The energy of the reactants (C) The energy of the products (D) The minimum energy required to start a chemical reaction 37. Which of the following statements describes the effect of catalysts? (A) They increase the reaction rate by lowering the activation energy (B) They increase the reaction ra ...
... (B) The energy of the reactants (C) The energy of the products (D) The minimum energy required to start a chemical reaction 37. Which of the following statements describes the effect of catalysts? (A) They increase the reaction rate by lowering the activation energy (B) They increase the reaction ra ...
Additional questions
... Concentrated sulfuric acid contains very little water, only 5.0% by mass. It has a density of 1.84 g/mL. What is the molarity of this acid? ...
... Concentrated sulfuric acid contains very little water, only 5.0% by mass. It has a density of 1.84 g/mL. What is the molarity of this acid? ...
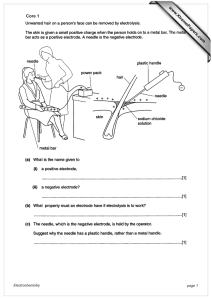
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.