Ex - Bosna Sema
... the substance which is totally consumed when the chemical reaction is complete. The amount of product formed is limited by this reagent since the reaction cannot proceed further without it. The other reagents may be present in excess of the quantities required to react with the limiting reagent. 4 c ...
... the substance which is totally consumed when the chemical reaction is complete. The amount of product formed is limited by this reagent since the reaction cannot proceed further without it. The other reagents may be present in excess of the quantities required to react with the limiting reagent. 4 c ...
Full-Text PDF
... and mesopores have been synthesized by delaminating the layered zeolite precursors MCM-22 and ferrierite [14]. However, none of these techniques yields a regular distribution of mesopores, let alone an ideal channel system of mesopores structurally connected with the regular micropores of the zeolit ...
... and mesopores have been synthesized by delaminating the layered zeolite precursors MCM-22 and ferrierite [14]. However, none of these techniques yields a regular distribution of mesopores, let alone an ideal channel system of mesopores structurally connected with the regular micropores of the zeolit ...
2.6 M - Thierry Karsenti
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
Rhenium- and molybdenum-catalyzed dehydration reactions
... supply of fossil feedstocks, combined with a strong increase in both the global population and their prosperity, makes our current way of life unsustainable. If we want to maintain our prosperity and enable others to share in it, it is inevitable that we find a sustainable alternative for fossil fee ...
... supply of fossil feedstocks, combined with a strong increase in both the global population and their prosperity, makes our current way of life unsustainable. If we want to maintain our prosperity and enable others to share in it, it is inevitable that we find a sustainable alternative for fossil fee ...
Activation of Nitrous Oxide and Selective Epoxidation of Alkenes
... toward transition metals.2 However, there is incentive to use N2O as an oxygen donor because it contains 36 wt % oxygen, and the byproduct of an oxidation reaction would be N2. In practice, there are only a few catalytic systems that have been shown to be efficient for the activation of N2O for sele ...
... toward transition metals.2 However, there is incentive to use N2O as an oxygen donor because it contains 36 wt % oxygen, and the byproduct of an oxidation reaction would be N2. In practice, there are only a few catalytic systems that have been shown to be efficient for the activation of N2O for sele ...
Gases - chemmybear.com
... Represented above are five identical balloons, each filled to the same volume at 25C and 1.0 atmosphere pressure with the pure gases indicated. (a) Which balloon contains the greatest mass of gas? Explain. (b) Compare the average kinetic energies of the gas molecules in the balloons. Explain. (c) W ...
... Represented above are five identical balloons, each filled to the same volume at 25C and 1.0 atmosphere pressure with the pure gases indicated. (a) Which balloon contains the greatest mass of gas? Explain. (b) Compare the average kinetic energies of the gas molecules in the balloons. Explain. (c) W ...
18-19 SpontEnt
... Nature tends to move spontaneously from a state of lower probability to one of higher probability »!G.N. Lewis (Nobel Laureate) ...
... Nature tends to move spontaneously from a state of lower probability to one of higher probability »!G.N. Lewis (Nobel Laureate) ...
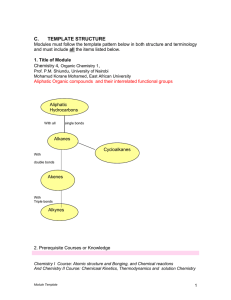
Organic Molecules
... they are bonded. The condensed structural formula below shows that in 2-methylpropane, there is a branched chain attached to the second carbon atom of the main chain. You can check this by looking at the structural formula in . ...
... they are bonded. The condensed structural formula below shows that in 2-methylpropane, there is a branched chain attached to the second carbon atom of the main chain. You can check this by looking at the structural formula in . ...
Production of Materials by Jason Yu #2
... Ethylene is a high demand hydrocarbon used to make polymers; however the fractional distillation of petroleum does not produce enough to meet our needs Because very little ethylene is found in natural gas or crude oil, it must be produced from other hydrocarbons by a process known as ‘cracking’ Frac ...
... Ethylene is a high demand hydrocarbon used to make polymers; however the fractional distillation of petroleum does not produce enough to meet our needs Because very little ethylene is found in natural gas or crude oil, it must be produced from other hydrocarbons by a process known as ‘cracking’ Frac ...
ANSWERS Problem Set 5a – Chemical Reactions
... Subscripts are underlined, Coefficients are boxed 3 H2SO4+ 2 Al Al2(SO4)3 + 3 H2 11) Write the word equation for the following balanced reactions: a. 2 Mg (s) + O2 (g) 2 MgO (s) solid magnesium reacts with oxygen gas to produce solid magnesium oxide in a synthesis reaction. b. HCl (aq) + NaOH (a ...
... Subscripts are underlined, Coefficients are boxed 3 H2SO4+ 2 Al Al2(SO4)3 + 3 H2 11) Write the word equation for the following balanced reactions: a. 2 Mg (s) + O2 (g) 2 MgO (s) solid magnesium reacts with oxygen gas to produce solid magnesium oxide in a synthesis reaction. b. HCl (aq) + NaOH (a ...
chapter 8
... important to specify the pressure at which the reaction occurs or to specify that the pressure must be above normal. Many reactions are speeded up and can take place at lower temperatures in the presence of a catalyst. A catalyst is a substance that changes the rate of a chemical reaction but can be ...
... important to specify the pressure at which the reaction occurs or to specify that the pressure must be above normal. Many reactions are speeded up and can take place at lower temperatures in the presence of a catalyst. A catalyst is a substance that changes the rate of a chemical reaction but can be ...
Entropy (Part I)
... is less; therefore, the entropy has increased. D. expand to fill the container only if it is an exothermic process and the net result has a greater probability. ...
... is less; therefore, the entropy has increased. D. expand to fill the container only if it is an exothermic process and the net result has a greater probability. ...
chapter 8 - Denton ISD
... important to specify the pressure at which the reaction occurs or to specify that the pressure must be above normal. Many reactions are speeded up and can take place at lower temperatures in the presence of a catalyst. A catalyst is a substance that changes the rate of a chemical reaction but can be ...
... important to specify the pressure at which the reaction occurs or to specify that the pressure must be above normal. Many reactions are speeded up and can take place at lower temperatures in the presence of a catalyst. A catalyst is a substance that changes the rate of a chemical reaction but can be ...
Hydrogen Storage in Magnesium Clusters
... techniques, so the desorption temperature of MgH2 cannot be lowered below that of the bulk value of 573 K. Although some alloying or doping techniques are able to affect the desorption temperature, this is accompanied by a lower hydrogen storage capacity due to the added weight.10,26-28 Furthermore, ...
... techniques, so the desorption temperature of MgH2 cannot be lowered below that of the bulk value of 573 K. Although some alloying or doping techniques are able to affect the desorption temperature, this is accompanied by a lower hydrogen storage capacity due to the added weight.10,26-28 Furthermore, ...
a) How many moles of water are created when 108 moles of oxygen
... work as is explained in the lessons. You are required to have this package completed BEFORE you write your unit test. Do your best and ask questions if you don’t understand anything! ...
... work as is explained in the lessons. You are required to have this package completed BEFORE you write your unit test. Do your best and ask questions if you don’t understand anything! ...
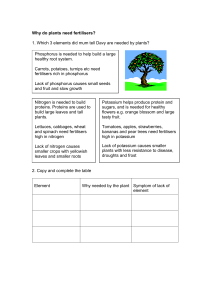
Fertilisers
... a. Why is nitrogen so unreactive? b. Write a word equation to show the reaction of oxygen and nitrogen. c. What colour was the nitrogen dioxide gas that formed? d. What colour did the indicator go when the water was added to the flask? e. What have you learned about the pH of the solution made when ...
... a. Why is nitrogen so unreactive? b. Write a word equation to show the reaction of oxygen and nitrogen. c. What colour was the nitrogen dioxide gas that formed? d. What colour did the indicator go when the water was added to the flask? e. What have you learned about the pH of the solution made when ...
Stoichiometry File
... model for the combustion of gasoline. The use of octane to represent all of the hydrocarbons in gasoline is mainly for simplicity. If we chose to, it would not be very difficult to write similar combustion equations for each hydrocarbon that is actually present. But the assumption of complete combus ...
... model for the combustion of gasoline. The use of octane to represent all of the hydrocarbons in gasoline is mainly for simplicity. If we chose to, it would not be very difficult to write similar combustion equations for each hydrocarbon that is actually present. But the assumption of complete combus ...
Chemistry Worksheets
... Why are homogeneous mixtures more difficult to separate than heterogeneous mixtures? ...
... Why are homogeneous mixtures more difficult to separate than heterogeneous mixtures? ...
sec chemistry may 2011 marking scheme
... • Unreactive / does not react with food • Removes O2 / slows the bacterial growth / slows the decay of the food / keeps food fresh / food preservation. • Slows the oxidation of food / retains the flavour of the food ...
... • Unreactive / does not react with food • Removes O2 / slows the bacterial growth / slows the decay of the food / keeps food fresh / food preservation. • Slows the oxidation of food / retains the flavour of the food ...
Analyze
... Think about It This is a very exothermic reaction that occurs very fast and is therefore explosive. 5.74. Collect and Organize We are given the balanced chemical equation for the decomposition of TNT. The enthalpy change from Problem 5.73 for the explosion of ammonium nitrate with fuel oil is –7198 ...
... Think about It This is a very exothermic reaction that occurs very fast and is therefore explosive. 5.74. Collect and Organize We are given the balanced chemical equation for the decomposition of TNT. The enthalpy change from Problem 5.73 for the explosion of ammonium nitrate with fuel oil is –7198 ...
- sartep.com
... 1. __________ A piece of calcite has a mass of 35.6 grams and a volume of 12.9 cm3. On which of the following liquids will calcite float? (A) carbon tetrachloride (density = 1.60 g/cm3) (B) methylene bromide (density = 2.50 g/cm3) (C) neither of the above substances (D) both of the above substances ...
... 1. __________ A piece of calcite has a mass of 35.6 grams and a volume of 12.9 cm3. On which of the following liquids will calcite float? (A) carbon tetrachloride (density = 1.60 g/cm3) (B) methylene bromide (density = 2.50 g/cm3) (C) neither of the above substances (D) both of the above substances ...
Chemical Equations
... Note, there appear to be more oxygen atoms, fewer hydrogen atoms at the end that at the beginning! ...
... Note, there appear to be more oxygen atoms, fewer hydrogen atoms at the end that at the beginning! ...
Unit- 5.pmd
... Another important factor featuring adsorption is the heat of adsorption. During adsorption, there is always a decrease in residual forces of the surface, i.e., there is decrease in surface energy which appears as heat. Adsorption, therefore, is invariably an exothermic process. In other words, ∆H of ...
... Another important factor featuring adsorption is the heat of adsorption. During adsorption, there is always a decrease in residual forces of the surface, i.e., there is decrease in surface energy which appears as heat. Adsorption, therefore, is invariably an exothermic process. In other words, ∆H of ...
At equilibrium
... • Know the definition of Le Chatelier’s Principle, and understand its application to the prediction of the direction of change in a chemical reaction at equilibrium, following changes in pressure, volume, temperature and amount of reactants and products. • Understand the definition of the reaction q ...
... • Know the definition of Le Chatelier’s Principle, and understand its application to the prediction of the direction of change in a chemical reaction at equilibrium, following changes in pressure, volume, temperature and amount of reactants and products. • Understand the definition of the reaction q ...
Carbon dioxide capture and utilization in petrochemical industry
... Carbon dioxide capture and sequestration (CCS) can significantly reduce emissions from CO2 sources as well as certain industry types such as ethanol and natural gas processing plants. CCS methodologies for industrial and energy-related sources comprise three steps: CO2 capture, CO2 transportation an ...
... Carbon dioxide capture and sequestration (CCS) can significantly reduce emissions from CO2 sources as well as certain industry types such as ethanol and natural gas processing plants. CCS methodologies for industrial and energy-related sources comprise three steps: CO2 capture, CO2 transportation an ...
Catalytic reforming

Catalytic reforming is a chemical process used to convert petroleum refinery naphthas distilled from crude oil (typically having low octane ratings) into high-octane liquid products called reformates, which are premium blending stocks for high-octane gasoline. The process converts low-octane linear hydrocarbons (paraffins) into branched alkanes (isoparaffins) and cyclic naphthenes, which are then partially dehydrogenated to produce high-octane aromatic hydrocarbons. The dehydrogenation also produces significant amounts of byproduct hydrogen gas, which is fed into other refinery processes such as hydrocracking. A side reaction is hydrogenolysis, which produces light hydrocarbons of lower value, such as methane, ethane, propane and butanes.In addition to a gasoline blending stock, reformate is the main source of aromatic bulk chemicals such as benzene, toluene, xylene and ethylbenzene which have diverse uses, most importantly as raw materials for conversion into plastics. However, the benzene content of reformate makes it carcinogenic, which has led to governmental regulations effectively requiring further processing to reduce its benzene content.This process is quite different from and not to be confused with the catalytic steam reforming process used industrially to produce products such as hydrogen, ammonia, and methanol from natural gas, naphtha or other petroleum-derived feedstocks. Nor is this process to be confused with various other catalytic reforming processes that use methanol or biomass-derived feedstocks to produce hydrogen for fuel cells or other uses.