Section 4.6: Double Displacement Reactions
... 7. Silver ions are the only metal ions that can be precipitated from a solution containing the C2H3O2− ions. Therefore, a solution such as NaC2H3O2(aq) can be used to precipitate silver ions from a mixture of dissolved metal ions. 8. Answers may vary. Sample answer: Most of the limescale that forms ...
... 7. Silver ions are the only metal ions that can be precipitated from a solution containing the C2H3O2− ions. Therefore, a solution such as NaC2H3O2(aq) can be used to precipitate silver ions from a mixture of dissolved metal ions. 8. Answers may vary. Sample answer: Most of the limescale that forms ...
Chemical Equilibrium
... Hydrogen and oxygen gas will react to form steam (gaseous water). Hydrogen and oxygen gases were mixed in a 2 L flask at 450 C. After equilibrium was established, it was determined that there were 3 moles of water, 1.2 moles of hydrogen and 0.8 moles of oxygen in the flask. What is the equilibrium c ...
... Hydrogen and oxygen gas will react to form steam (gaseous water). Hydrogen and oxygen gases were mixed in a 2 L flask at 450 C. After equilibrium was established, it was determined that there were 3 moles of water, 1.2 moles of hydrogen and 0.8 moles of oxygen in the flask. What is the equilibrium c ...
Catalyst Notes - University of Idaho
... Surface reaction: Generally, species are highly mobile on surfaces. Adsorbed species ‘hop’ to adjacent sites by overcoming a small energy barrier. A good catalyst for a species would be one where these energy barriers are small. A poor catalyst may be such a strong absorber for a species that the sp ...
... Surface reaction: Generally, species are highly mobile on surfaces. Adsorbed species ‘hop’ to adjacent sites by overcoming a small energy barrier. A good catalyst for a species would be one where these energy barriers are small. A poor catalyst may be such a strong absorber for a species that the sp ...
Chemistry II Exams and Keys Corrected 2016 Season
... 17. Analysis of a brass sample (sample 1) shows that it contains 5 grams of zinc and 20 grams of copper. Another brass sample (sample 2) contains 10 grams of zinc and 10 grams of copper. How will it be possible to prepare 10 grams of a brass sample containing 30% of zinc using these two brass sample ...
... 17. Analysis of a brass sample (sample 1) shows that it contains 5 grams of zinc and 20 grams of copper. Another brass sample (sample 2) contains 10 grams of zinc and 10 grams of copper. How will it be possible to prepare 10 grams of a brass sample containing 30% of zinc using these two brass sample ...
Unit 2:
... acid by a strong base. Standard school laboratory equipment and a supply of standardized 1.00 molar HCl and standardized 1.00 molar NaOH are available. (a) What equipment would be needed? (b) What measurements should be taken? (c) Without performing calculations, describe how the resulting data shou ...
... acid by a strong base. Standard school laboratory equipment and a supply of standardized 1.00 molar HCl and standardized 1.00 molar NaOH are available. (a) What equipment would be needed? (b) What measurements should be taken? (c) Without performing calculations, describe how the resulting data shou ...
unit 4: chemical reaction rates
... carbon, namely 12. In order to avoid confusion, it is necessary to specify the elementary units, which may be a molecule, an atom, an electron, an ion, etc. For example, a mole of hydrogen can mean either a mole hydrogen atoms (H) or hydrogen molecules (H2). The symbol n is used to represent the amo ...
... carbon, namely 12. In order to avoid confusion, it is necessary to specify the elementary units, which may be a molecule, an atom, an electron, an ion, etc. For example, a mole of hydrogen can mean either a mole hydrogen atoms (H) or hydrogen molecules (H2). The symbol n is used to represent the amo ...
Estimate the strength of given sodium carbonate solution
... A primary standard solution must follow these conditions: Compound should be free from impurity. Compound should be completely soluble in dissolving solvent. Compound should not be hygroscopic. Solution formed should be stable and should not decompose with light / time. There should not be ...
... A primary standard solution must follow these conditions: Compound should be free from impurity. Compound should be completely soluble in dissolving solvent. Compound should not be hygroscopic. Solution formed should be stable and should not decompose with light / time. There should not be ...
dx cx dx and x - Cameron University
... 3.5 Treat mixtures of gases using the concept of partial pressure and the ideal gas law 4.1 Evaluate the derivative d/dx of f(x) = ax, f(x) = ln x, and f(x) = axn at least 4.2 Apply the chain rule to differentiate a function 4.3 Given a multiple variable expression, determine the partial derivatives ...
... 3.5 Treat mixtures of gases using the concept of partial pressure and the ideal gas law 4.1 Evaluate the derivative d/dx of f(x) = ax, f(x) = ln x, and f(x) = axn at least 4.2 Apply the chain rule to differentiate a function 4.3 Given a multiple variable expression, determine the partial derivatives ...
precipitation rxn_level_packet
... Which of the following reactions is (are) oxidation-reduction reactions? a) Zn(OH)2(s) + H2SO4(aq) ZnSO4(aq) + 2 H2O(l) b) Ca(s) + 2 H2O(l) Ca(OH)2(s) + H2(g) ...
... Which of the following reactions is (are) oxidation-reduction reactions? a) Zn(OH)2(s) + H2SO4(aq) ZnSO4(aq) + 2 H2O(l) b) Ca(s) + 2 H2O(l) Ca(OH)2(s) + H2(g) ...
Original powerpoint (~1.9 MB)
... We’ve seen in reference to Le Chatalier’s Principle that if more than one reaction can take place in a container, then the reactions might not be able to be treated independently. Other equilibrium processes may affect the solubility of the solid and lead to miscalculated Ksp ...
... We’ve seen in reference to Le Chatalier’s Principle that if more than one reaction can take place in a container, then the reactions might not be able to be treated independently. Other equilibrium processes may affect the solubility of the solid and lead to miscalculated Ksp ...
AP syllabus
... 6. Using the Henderson-Hasselbalch equation to select components for a buffer of a given pH 7. Solving problems involving the ionization of polyprotic acids 8. Using a pH meter to determine an ionization curve and ionization constants 9. Calculating the pH at various points of a variety of titratio ...
... 6. Using the Henderson-Hasselbalch equation to select components for a buffer of a given pH 7. Solving problems involving the ionization of polyprotic acids 8. Using a pH meter to determine an ionization curve and ionization constants 9. Calculating the pH at various points of a variety of titratio ...
Contents
... example, the shells of electronic devices are usually made of plastic. The plastic was synthesized from a chemical compound which itself had been produced from oil. The silicon at the heart of most electronic devices is produced by converting silicon dioxide (the mineral quartz) to the element silic ...
... example, the shells of electronic devices are usually made of plastic. The plastic was synthesized from a chemical compound which itself had been produced from oil. The silicon at the heart of most electronic devices is produced by converting silicon dioxide (the mineral quartz) to the element silic ...
Chemistry Chapter 12
... of the substances in the compound -can use this mole relationship to go from one substance to another -for this calculation it makes no difference if you are working with reactants or products -when the phrase "an excess of" is used in the wording of the problem, this simply means that not all of th ...
... of the substances in the compound -can use this mole relationship to go from one substance to another -for this calculation it makes no difference if you are working with reactants or products -when the phrase "an excess of" is used in the wording of the problem, this simply means that not all of th ...
Chemical reactions cause chemical changes. They involve the
... To React or Not to React! Answer Key Instructions: Read the article. Answer all the questions on a separate sheet of paper. You may write on this article. Chemical reactions cause chemical changes. They involve the breaking and making of chemical bonds. All chemical reactions involve a change in sub ...
... To React or Not to React! Answer Key Instructions: Read the article. Answer all the questions on a separate sheet of paper. You may write on this article. Chemical reactions cause chemical changes. They involve the breaking and making of chemical bonds. All chemical reactions involve a change in sub ...
Physical Chemistry 20130410 week 2 Wednesday April 10 2013
... If k1/k2 >>1 then [C]>>[D] so C is favored kinetically. If K1/K2<<1 then [D]>>[D] so D is favored thermodynamically. See handout for 1,3 butadiene. HBr is a very strong acid. The intermediate of a reaction is always at the bottom of the dip. 1,4 addition is favored thermodynamically because it has l ...
... If k1/k2 >>1 then [C]>>[D] so C is favored kinetically. If K1/K2<<1 then [D]>>[D] so D is favored thermodynamically. See handout for 1,3 butadiene. HBr is a very strong acid. The intermediate of a reaction is always at the bottom of the dip. 1,4 addition is favored thermodynamically because it has l ...
buffers - sbhschemistry
... (HNO2) which has been combined with 0.1molL-1 sodium nitrite (NaNO2). Ka (HNO2) = 5 x 10-4 ...
... (HNO2) which has been combined with 0.1molL-1 sodium nitrite (NaNO2). Ka (HNO2) = 5 x 10-4 ...
CBSE-12th/2011/CHEMISTRY
... larger in size. so lp repulsion is less significant. Also, S-S bond is stronger than O-O bond & S=S is less strong(less than 2 S-S bonds). This is also affected by the fact that O forms strong bonds with mostly other elements than itself. Ans.13 (i)In aqueous solution, Cu+ ion undergoes oxidation to ...
... larger in size. so lp repulsion is less significant. Also, S-S bond is stronger than O-O bond & S=S is less strong(less than 2 S-S bonds). This is also affected by the fact that O forms strong bonds with mostly other elements than itself. Ans.13 (i)In aqueous solution, Cu+ ion undergoes oxidation to ...
unit-4-notes-1_enthalpy-and-entropy
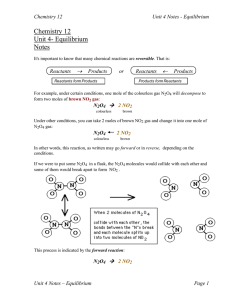
... 2. Letting material into or out of the system will affect rates so a system at equilibrium is a closed system. 3. Again, consider the equilibrium reaction: N2O4 2 NO2 In the example that we did to construct the graphs, we had started with pure N2O4 and no NO2. The forward reaction rate was high at t ...
... 2. Letting material into or out of the system will affect rates so a system at equilibrium is a closed system. 3. Again, consider the equilibrium reaction: N2O4 2 NO2 In the example that we did to construct the graphs, we had started with pure N2O4 and no NO2. The forward reaction rate was high at t ...