Chapter 18: Chemical Equilibrium
... You have learned that some chemical systems have little tendency to react and others go readily to completion. In between these two extremes are the majority of reactions that reach a state of equilibrium with varying amounts of reactants unconsumed. If the reactants are not consumed, then not all t ...
... You have learned that some chemical systems have little tendency to react and others go readily to completion. In between these two extremes are the majority of reactions that reach a state of equilibrium with varying amounts of reactants unconsumed. If the reactants are not consumed, then not all t ...
Chem 5336_Potentiometry
... EB is related to a1 and a2, but a2 is constant (0.1 M HCl) These ion activities control the membrane potentials, E1 and E2 and so control EB, at 25 oC EB = E1 – E2 = 0.0592 log (a1/a2) Ecell = const + 0.0592 log (a1) Ecell = const - 0.0592 pH In practice, pH meter incorporates these equations And r ...
... EB is related to a1 and a2, but a2 is constant (0.1 M HCl) These ion activities control the membrane potentials, E1 and E2 and so control EB, at 25 oC EB = E1 – E2 = 0.0592 log (a1/a2) Ecell = const + 0.0592 log (a1) Ecell = const - 0.0592 pH In practice, pH meter incorporates these equations And r ...
Chapter 18 pdf
... You have learned that some chemical systems have little tendency to react and others go readily to completion. In between these two extremes are the majority of reactions that reach a state of equilibrium with varying amounts of reactants unconsumed. If the reactants are not consumed, then not all t ...
... You have learned that some chemical systems have little tendency to react and others go readily to completion. In between these two extremes are the majority of reactions that reach a state of equilibrium with varying amounts of reactants unconsumed. If the reactants are not consumed, then not all t ...
PPT
... • A balanced chemical equation is one in which the number of atoms of each element in the reactants is equal to the number of atoms of that same element in the products. • A reaction can be balanced by applying the law of conservation of matter. • Coefficients (in red below) are written to the left ...
... • A balanced chemical equation is one in which the number of atoms of each element in the reactants is equal to the number of atoms of that same element in the products. • A reaction can be balanced by applying the law of conservation of matter. • Coefficients (in red below) are written to the left ...
Ministry of Education and Science of the Ukraine
... the solution concentration: mass and volume percents, molar fraction, molarity, molality. Electrolytic dissociation, electrolytes. Strong and weak electrolytes. The degree of dissociation and dissociation constant. Ostwald’s dilution law. 5. Acid-base equilibrium in biological liquids. General prope ...
... the solution concentration: mass and volume percents, molar fraction, molarity, molality. Electrolytic dissociation, electrolytes. Strong and weak electrolytes. The degree of dissociation and dissociation constant. Ostwald’s dilution law. 5. Acid-base equilibrium in biological liquids. General prope ...
physical chemistry notes
... with alkalis, metal oxides, metal carbonates or metals such as magnesium. The molar ratio will be the same, but the reaction rate might be different. This is because, as the hydrogen ions from the weak acid are used up, the weak acid dissociates further to replace these hydrogen ions, so that eventu ...
... with alkalis, metal oxides, metal carbonates or metals such as magnesium. The molar ratio will be the same, but the reaction rate might be different. This is because, as the hydrogen ions from the weak acid are used up, the weak acid dissociates further to replace these hydrogen ions, so that eventu ...
Reaction Rate Reading Packet
... For the same mass, many small particles have a greater total surface area than one large particle. For example, steel wool has a larger surface area than a block of steel of the same mass. This allows oxygen molecules to collide with many more iron atoms per unit of time. The more surface contact be ...
... For the same mass, many small particles have a greater total surface area than one large particle. For example, steel wool has a larger surface area than a block of steel of the same mass. This allows oxygen molecules to collide with many more iron atoms per unit of time. The more surface contact be ...
ksp - lozon.ca
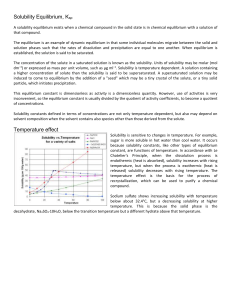
... The concentration of the solute in a saturated solution is known as the solubility. Units of solubility may be molar (mol dm−3) or expressed as mass per unit volume, such as μg ml−1. Solubility is temperature dependent. A solution containing a higher concentration of solute than the solubility is sa ...
... The concentration of the solute in a saturated solution is known as the solubility. Units of solubility may be molar (mol dm−3) or expressed as mass per unit volume, such as μg ml−1. Solubility is temperature dependent. A solution containing a higher concentration of solute than the solubility is sa ...
Expt. 5: Binary Phase Diagram CHEM 366 V-1 Binary Solid
... particular phase or phases in which a pure substance or mixture exists under a given set of conditions, e.g., temperature, pressure and composition, is obviously of utmost importance in our use of or interaction with the myriad of material substances in the world. We take it for granted that the mix ...
... particular phase or phases in which a pure substance or mixture exists under a given set of conditions, e.g., temperature, pressure and composition, is obviously of utmost importance in our use of or interaction with the myriad of material substances in the world. We take it for granted that the mix ...
Construction of Detailed Chemical Reaction Models
... Where Ae is the pre-exponential factor, E an empirically determined activation energy, and a and b, the exponents of the reactant concentrations that are able to represent the concentration dependence over a range of conditions also empirically determined. Unfortunately, these oversimplified mechani ...
... Where Ae is the pre-exponential factor, E an empirically determined activation energy, and a and b, the exponents of the reactant concentrations that are able to represent the concentration dependence over a range of conditions also empirically determined. Unfortunately, these oversimplified mechani ...
Technical Data Sheet (E
... 1. Thoroughly clean and degrease parts with an immersion in a hot EPI alkaline soak cleaner such as E-Kleen SR 102 or E-Kleen SR 102-E. E-Kleen SR 149-L is recommended for spray cleaning. Heavy duty cleaners and rust removers can be used without affecting the grain of the E-Phos 660 coating. 2. Bott ...
... 1. Thoroughly clean and degrease parts with an immersion in a hot EPI alkaline soak cleaner such as E-Kleen SR 102 or E-Kleen SR 102-E. E-Kleen SR 149-L is recommended for spray cleaning. Heavy duty cleaners and rust removers can be used without affecting the grain of the E-Phos 660 coating. 2. Bott ...
LABORATORY MANUAL CHEMISTRY 121
... dissociates into chloride ion and trans-[Co(en)2Cl2]+. This cobalt complex ion then reacts in acid solution to yield a mixture of the cis and trans forms of [Co(en)2(H2O)Cl]2+. The progress of this reaction can be followed visually because the products are red while the reactant is green. The purpos ...
... dissociates into chloride ion and trans-[Co(en)2Cl2]+. This cobalt complex ion then reacts in acid solution to yield a mixture of the cis and trans forms of [Co(en)2(H2O)Cl]2+. The progress of this reaction can be followed visually because the products are red while the reactant is green. The purpos ...
On the formation of radiation fogs under heavily polluted
... curves very closely as they grow. With certain types of atmospheric clouds this would not be the case as the droplets are actually out of equilibrium because of kinetic limitations in water vapor condensation (Nenes et al., 2001). Considering gases such as HNO3 , with mixing ratios orders of magnitu ...
... curves very closely as they grow. With certain types of atmospheric clouds this would not be the case as the droplets are actually out of equilibrium because of kinetic limitations in water vapor condensation (Nenes et al., 2001). Considering gases such as HNO3 , with mixing ratios orders of magnitu ...
No Slide Title
... • The standard rate constant k0: It is the measure of the kinetic facility of a redox couple. A system with a large k0 will achieve equilibrium on a short time scale, but a system with small k0 will be sluggish • Values of k0 reported in the literature for electrochemical reactions vary from about 1 ...
... • The standard rate constant k0: It is the measure of the kinetic facility of a redox couple. A system with a large k0 will achieve equilibrium on a short time scale, but a system with small k0 will be sluggish • Values of k0 reported in the literature for electrochemical reactions vary from about 1 ...
Solubility
... • We can also determine the concentration of an ion necessary for precipitation to begin. • Assume that precipitation begins when Qsp = Ksp • Example: If a solution contains 0.0020 mol CrO42per liter, what concentration of Ag+ ion must be added as AgNO3 before Ag2CrO4 begins to precipitate. (Neglect ...
... • We can also determine the concentration of an ion necessary for precipitation to begin. • Assume that precipitation begins when Qsp = Ksp • Example: If a solution contains 0.0020 mol CrO42per liter, what concentration of Ag+ ion must be added as AgNO3 before Ag2CrO4 begins to precipitate. (Neglect ...
File
... Smaller nonmetals have greater nuclear charge because the outer electrons are closer to the nucleus. Thus the tendency to attract electrons will increase. Metalloids have some chemical and physical properties of metals and other properties of nonmetals. In the periodic table, the metalloids lie alon ...
... Smaller nonmetals have greater nuclear charge because the outer electrons are closer to the nucleus. Thus the tendency to attract electrons will increase. Metalloids have some chemical and physical properties of metals and other properties of nonmetals. In the periodic table, the metalloids lie alon ...
MASSACHUSETTS INSTITUTE OF TECHNOLOGY
... Solving Kinetic Equations Numerically Using CHEMKIN The Sample Transient Calculation shipped with CHEMKIN is the for the spontaneous adiabatic combustion of a stoichiometric H2/air mixture, which according to their model takes less than a millisecond. A different H2/air kinetic model is given in GRI ...
... Solving Kinetic Equations Numerically Using CHEMKIN The Sample Transient Calculation shipped with CHEMKIN is the for the spontaneous adiabatic combustion of a stoichiometric H2/air mixture, which according to their model takes less than a millisecond. A different H2/air kinetic model is given in GRI ...
Kinetics and Equilibrium ___ 1. In a chemical reaction the use of a
... H1+ ions; (2) concentration of OH1– ions; (3) pressure; (4) temperature. ___ 17. Which change may occur in a reaction system when a catalyst is added? (1) The equilibrium point is reached more rapidly; (2) The potential energy of the reactants increases; (3) The potential energy of the products decr ...
... H1+ ions; (2) concentration of OH1– ions; (3) pressure; (4) temperature. ___ 17. Which change may occur in a reaction system when a catalyst is added? (1) The equilibrium point is reached more rapidly; (2) The potential energy of the reactants increases; (3) The potential energy of the products decr ...
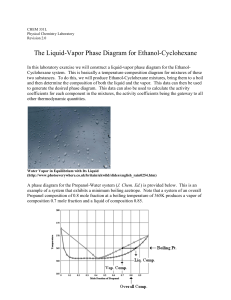
The Liquid-Vapor Phase Diagram for Ethanol
... between the Claissen Head and the distilling flask. To expedite reaching a steady state with mixtures, flush out the distillate collected in the Claissen Head several times by inserting a disposable pipet with a squeeze bulb attached and forcing Air through it. After a steady temperature has been re ...
... between the Claissen Head and the distilling flask. To expedite reaching a steady state with mixtures, flush out the distillate collected in the Claissen Head several times by inserting a disposable pipet with a squeeze bulb attached and forcing Air through it. After a steady temperature has been re ...