pdfCfE Higher - Unit 3 - Pupil Booklet 2 MB
... Road and rail tankers that carry chemicals are constructed to withstand impact in accidents. Plants have their own fire fighting teams on site. Plants are designed with safety in mind. Chemicals are hazardous so the accident rate will never be zero but the aim is to learn from mistakes and reduce th ...
... Road and rail tankers that carry chemicals are constructed to withstand impact in accidents. Plants have their own fire fighting teams on site. Plants are designed with safety in mind. Chemicals are hazardous so the accident rate will never be zero but the aim is to learn from mistakes and reduce th ...
Acids and Bases - Personal.kent.edu
... pH Measure of acid/base strength. pH = 7 is neutral. pH < 7 is acidic. pH > 7 is basic. pH is defined as: pH = -log10 ([H+ ]) Buffers A buffer solution is a mixture of a weak acid and its conjugate weak base. Buffers are important because they prevent drastic pH changes from occurring. The pH of a b ...
... pH Measure of acid/base strength. pH = 7 is neutral. pH < 7 is acidic. pH > 7 is basic. pH is defined as: pH = -log10 ([H+ ]) Buffers A buffer solution is a mixture of a weak acid and its conjugate weak base. Buffers are important because they prevent drastic pH changes from occurring. The pH of a b ...
Model-based Analysis of Non-specific Binding for
... fluorescent labeled targets, which is inevitable when a complex mixture of DNA/RNA fragments is hybridized to millions of probes simultaneously. Thus, the development and the evaluation of algorithms to predict the amount of NSB are important topics for the improvement of microarray analysis. In thi ...
... fluorescent labeled targets, which is inevitable when a complex mixture of DNA/RNA fragments is hybridized to millions of probes simultaneously. Thus, the development and the evaluation of algorithms to predict the amount of NSB are important topics for the improvement of microarray analysis. In thi ...
ws-8-14-2
... _____ 4. (T/F) For the reaction aA bB, the rate remains constant over time. Reactant A is therefore a first order reactant. _____ 5. (T/F) Zero order reactions often have their rate controlled (limited) by a factor other than reactant concentrations, such as a catalyst or adsorption surface. 6. Th ...
... _____ 4. (T/F) For the reaction aA bB, the rate remains constant over time. Reactant A is therefore a first order reactant. _____ 5. (T/F) Zero order reactions often have their rate controlled (limited) by a factor other than reactant concentrations, such as a catalyst or adsorption surface. 6. Th ...
Name __KEY____________ Per. ______ Polarity and
... maximum possible amount of another reactant or product. This is called _____stoichiometry_____ (density/ writing a chemical formula/ stoichiometry). Sometimes not all of both reactants are completely used up in a chemical reaction. The reactant that will be completely used up is called the ___ limit ...
... maximum possible amount of another reactant or product. This is called _____stoichiometry_____ (density/ writing a chemical formula/ stoichiometry). Sometimes not all of both reactants are completely used up in a chemical reaction. The reactant that will be completely used up is called the ___ limit ...
Chapter 18 review
... ____ 23. In a first-order reaction, how does the rate change if the concentration of the reactant decreases to one-third its original value? a. The rate decreases by a factor of one-ninth. b. The rate decreases by a factor of one-third. c. The rate decreases by a factor of one-half. d. The rate stay ...
... ____ 23. In a first-order reaction, how does the rate change if the concentration of the reactant decreases to one-third its original value? a. The rate decreases by a factor of one-ninth. b. The rate decreases by a factor of one-third. c. The rate decreases by a factor of one-half. d. The rate stay ...
std 8 9 reviewanswers
... 9. A catalyst can speed up the rate of a given chemical reaction by A increasing the equilibrium constant in favor of products. B lowering the activation energy required for the reaction to occur. C raising the temperature at which the reaction occurs. D increasing the pressure of reactants, thus fa ...
... 9. A catalyst can speed up the rate of a given chemical reaction by A increasing the equilibrium constant in favor of products. B lowering the activation energy required for the reaction to occur. C raising the temperature at which the reaction occurs. D increasing the pressure of reactants, thus fa ...
LECTURE 6 - GENESIS OF MINERAL1
... Equilibrium: - The condition of minimum energy for a system. Phase: A physically and chemically homogeneous, mechanically separable part of a physicalchemical system. Degree of freedom (also known as variance): A variable, which if changed, does not result in a change in the equilibrium assemblage o ...
... Equilibrium: - The condition of minimum energy for a system. Phase: A physically and chemically homogeneous, mechanically separable part of a physicalchemical system. Degree of freedom (also known as variance): A variable, which if changed, does not result in a change in the equilibrium assemblage o ...
the importance of electron transfer mechanism in
... from 0-100 sccm at He pressure of 0.7 Torr. The plots of the four J levels are shown in Fig. 2 for the reaction with 02 and they are indistinguishable within experimental accuracy. Rate constants determined from these kinds of plots are (2.7 + 0.2) 10-12 cm3s (9.7 + 0.8) 10-12 cm3s and (0.40 + 0.01) ...
... from 0-100 sccm at He pressure of 0.7 Torr. The plots of the four J levels are shown in Fig. 2 for the reaction with 02 and they are indistinguishable within experimental accuracy. Rate constants determined from these kinds of plots are (2.7 + 0.2) 10-12 cm3s (9.7 + 0.8) 10-12 cm3s and (0.40 + 0.01) ...
Kinetics of the fading of phenolphthalein in alkaline solution
... to Beer's law, the spectral absorbance of the solution is directly proportional to [P2-1, or [P2-] = constant X (ahsorhance), so ln[P2-] = In (constant) In (ahsorhance). Therefore, a plot of in (absorbance) versus time should also be a straight line with slope of - k l , if the reaction is pseudo fi ...
... to Beer's law, the spectral absorbance of the solution is directly proportional to [P2-1, or [P2-] = constant X (ahsorhance), so ln[P2-] = In (constant) In (ahsorhance). Therefore, a plot of in (absorbance) versus time should also be a straight line with slope of - k l , if the reaction is pseudo fi ...
Chemical Equilibrium - The Gurukul Institute
... 2. One mole of H2, 2 mol of l2 and 3 mol of Hl are injected in a 1 litre flask. What will be the concentration of H2, l2 and Hl at equilibrium? Kc for the equilibrium, H2(g) + l2(g) ⇌ 2Hl is 45.9. 3. 15 g sample of BaO2 is heated to 794o C in a closed evacuated vessel of 5 litres capacity. How many ...
... 2. One mole of H2, 2 mol of l2 and 3 mol of Hl are injected in a 1 litre flask. What will be the concentration of H2, l2 and Hl at equilibrium? Kc for the equilibrium, H2(g) + l2(g) ⇌ 2Hl is 45.9. 3. 15 g sample of BaO2 is heated to 794o C in a closed evacuated vessel of 5 litres capacity. How many ...
Types of Reactions and Solution Chemistry
... (conductors of electricity) are placed in a solution and connected to a battery, the cations will migrate through the solution to the negatively charged electrode and the anions will migrate towards the positively charged electrode. If a light bulb is inserted into the circuit, the bulb will light u ...
... (conductors of electricity) are placed in a solution and connected to a battery, the cations will migrate through the solution to the negatively charged electrode and the anions will migrate towards the positively charged electrode. If a light bulb is inserted into the circuit, the bulb will light u ...
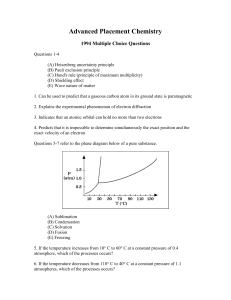
Advanced Placement Chemistry
... 11. Utilized as a coating to protect Fe from corrosion 12. Is added to silicon to enhance its properties as a semiconductor 13. Utilized as a shield from sources of radiation Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select ...
... 11. Utilized as a coating to protect Fe from corrosion 12. Is added to silicon to enhance its properties as a semiconductor 13. Utilized as a shield from sources of radiation Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select ...