6. Macroscopic equilibrium states and state variables (Hiroshi
... cannot exchange energy with another system and whose volumes and mole numbers are both kept constant. We then extended the notion to the systems which can exchange energy with another system through a thermal contact where the volumes and the mole numbers of the systems in contact are still kept con ...
... cannot exchange energy with another system and whose volumes and mole numbers are both kept constant. We then extended the notion to the systems which can exchange energy with another system through a thermal contact where the volumes and the mole numbers of the systems in contact are still kept con ...
+ p
... Technically we have gained because h(t) is an integral: not good in the denominator. The real point is that we get to choose p. Instead of choosing the optimal p which yields the intractable formula for information, we choose a non-optimal p with which we can work. ...
... Technically we have gained because h(t) is an integral: not good in the denominator. The real point is that we get to choose p. Instead of choosing the optimal p which yields the intractable formula for information, we choose a non-optimal p with which we can work. ...
revision - metc instructors collab site
... dependent only on thermodynamic temperature and is energy stored in the molecules States that the total energy stored in a body, or system, is termed enthalpy (H) Defines total stored energy the sum of internal energy and the product of pressure(P) and volume (V), i.e. H = U + PV Defines potential e ...
... dependent only on thermodynamic temperature and is energy stored in the molecules States that the total energy stored in a body, or system, is termed enthalpy (H) Defines total stored energy the sum of internal energy and the product of pressure(P) and volume (V), i.e. H = U + PV Defines potential e ...
simulating fritz haber`s ammonia synthesis with thermodynamic
... given a printed step-by-step guide to HSC Chemistry concerning this exercise. So the students were familiar with the program when they started doing the ammonia simulation. The students did not have too many difficulties with the HSC Chemistry simulations. Some of the students need the HSC Chemistry ...
... given a printed step-by-step guide to HSC Chemistry concerning this exercise. So the students were familiar with the program when they started doing the ammonia simulation. The students did not have too many difficulties with the HSC Chemistry simulations. Some of the students need the HSC Chemistry ...
Chap-4
... In the p-V diagram below , isotherms are distinguished by differences in temperature and the adiabats by differences in potential temperature q. There is another way of distinguishing differences between adiabats. In passing from one of the adiabats (q1 or q2) to another along an isotherm (this is a ...
... In the p-V diagram below , isotherms are distinguished by differences in temperature and the adiabats by differences in potential temperature q. There is another way of distinguishing differences between adiabats. In passing from one of the adiabats (q1 or q2) to another along an isotherm (this is a ...
Basic Thermodynamics - CERN Accelerator School
... A thermodynamic system is in thermodynamic equilibrium when all its state variables remain constant with time: there is no net flow of matter or energy, no phase changes, and no unbalanced potentials (or driving forces) within the system. A system that is in thermodynamic equilibrium experiences no ...
... A thermodynamic system is in thermodynamic equilibrium when all its state variables remain constant with time: there is no net flow of matter or energy, no phase changes, and no unbalanced potentials (or driving forces) within the system. A system that is in thermodynamic equilibrium experiences no ...
Lecture 2
... Thermal radiation emitted by matter in thermal equilibrium Black-Body radiation comes from a body that is in local thermodynamic equilibrium (LTE) with its surroundings. Thus all populations (atomic, ionic, molecular) are described entirely by the local temperature, and given by SahaBoltzmann statis ...
... Thermal radiation emitted by matter in thermal equilibrium Black-Body radiation comes from a body that is in local thermodynamic equilibrium (LTE) with its surroundings. Thus all populations (atomic, ionic, molecular) are described entirely by the local temperature, and given by SahaBoltzmann statis ...
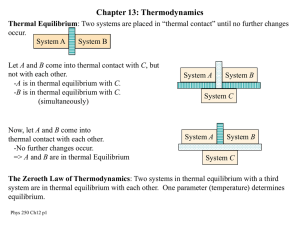
p250c13
... The Second Law of Thermodynamics •Heat cannot, by itself, pass from a cooler to a warmer body •It is impossible for any system to undergo a cyclic process whose sole result is the absorption of heat from a single reservoir at a single temperature and the performance of an equivalent amount of work. ...
... The Second Law of Thermodynamics •Heat cannot, by itself, pass from a cooler to a warmer body •It is impossible for any system to undergo a cyclic process whose sole result is the absorption of heat from a single reservoir at a single temperature and the performance of an equivalent amount of work. ...
The second law of thermodynamics
... the conditions in which this system is in equilibrium ?. We know that in equilibrium, the state of the system is fully specified by the functions of state, (E, V, N ). Thus, if the system is not in equilibrium, we need to add (at least) one more variable, call it α. To each choice of α, there corres ...
... the conditions in which this system is in equilibrium ?. We know that in equilibrium, the state of the system is fully specified by the functions of state, (E, V, N ). Thus, if the system is not in equilibrium, we need to add (at least) one more variable, call it α. To each choice of α, there corres ...
Engineering Building Room 2303 Mail Code Phone: 818-677
... 5. Kinetic energy terms are usually negligible unless the Mach number is large; potential energy terms are usually negligible unless the process specifically is intended to use potential energy changes (e.g., a hydroelectric plant. 6. The simplest open system is a steady-state, steady-flow system in ...
... 5. Kinetic energy terms are usually negligible unless the Mach number is large; potential energy terms are usually negligible unless the process specifically is intended to use potential energy changes (e.g., a hydroelectric plant. 6. The simplest open system is a steady-state, steady-flow system in ...
MS PowerPoint - Catalysis Eprints database
... The first law of thermodynamics deals with the law of conservation of energy. According to this law, energy can be neither created nor destroyed even though it can converted from one form to the other The first law can be stated in several other ways. It has been accepted that the perpetual motion o ...
... The first law of thermodynamics deals with the law of conservation of energy. According to this law, energy can be neither created nor destroyed even though it can converted from one form to the other The first law can be stated in several other ways. It has been accepted that the perpetual motion o ...
Document
... these cases , q = ∆u and w =∆u , respectively . But if q ≠ 0 and w ≠ 0 , is there a definite maximum amount of work which the system can do during its change of state ? the answer to this question requires an examination of the nature of process . The examination which will be made in this chapt ...
... these cases , q = ∆u and w =∆u , respectively . But if q ≠ 0 and w ≠ 0 , is there a definite maximum amount of work which the system can do during its change of state ? the answer to this question requires an examination of the nature of process . The examination which will be made in this chapt ...
Structure of Thrmodynamics
... A finite rate of change (or a finite rate of a rate of change) cannot be stopped by means of infinitesimal alteration in the circumstances. (J.W. Gibbs, Collected Works, Yale University. Press, Vol. 1 p.56,1948) ...
... A finite rate of change (or a finite rate of a rate of change) cannot be stopped by means of infinitesimal alteration in the circumstances. (J.W. Gibbs, Collected Works, Yale University. Press, Vol. 1 p.56,1948) ...
Thermodynamics
... 15.7 Entropy and the 2nd Law of Thermodynamics Example – A sample of 50 kg of water at 20oC is mixed with 50 kg at 24oC. The final temperature is 22oC. Estimate the change in Entropy. ...
... 15.7 Entropy and the 2nd Law of Thermodynamics Example – A sample of 50 kg of water at 20oC is mixed with 50 kg at 24oC. The final temperature is 22oC. Estimate the change in Entropy. ...
PHYSICAL CHEMISTRY I (01:160:327) SYLLABUS AND GENERAL
... statistical thermodynamics, and kinetics, which you should be able to apply in future studies and in your career in science or a related field. be able to apply your knowledge of thermodynamics/kinetics to physical transformations, chemical reactions, phase and chemical equilibria, and solutions. ha ...
... statistical thermodynamics, and kinetics, which you should be able to apply in future studies and in your career in science or a related field. be able to apply your knowledge of thermodynamics/kinetics to physical transformations, chemical reactions, phase and chemical equilibria, and solutions. ha ...
EGU2016-10322 - CO Meeting Organizer
... An interesting point in the Adam and Gibbs framework is that it assumes that viscous flow occurs through the cooperative re-arrangement of molecular sub-regions in the melt. From high temperature 29 Si NMR and Raman spectroscopy data, it actually is known that viscous flow occurs because of the coop ...
... An interesting point in the Adam and Gibbs framework is that it assumes that viscous flow occurs through the cooperative re-arrangement of molecular sub-regions in the melt. From high temperature 29 Si NMR and Raman spectroscopy data, it actually is known that viscous flow occurs because of the coop ...