Assumptions in equilibrium analysis and
... The work analysis considers a soil specimen in the triaxial cell and treats the specimen as a mass with no distinction as to its degree of saturation or composition. The analysis thus considers only the applied total stresses and does not consider ‘effective’ interparticle stresses or fluid pressur ...
... The work analysis considers a soil specimen in the triaxial cell and treats the specimen as a mass with no distinction as to its degree of saturation or composition. The analysis thus considers only the applied total stresses and does not consider ‘effective’ interparticle stresses or fluid pressur ...
Thermodynamics of ideal gases
... gas may dissociate or even ionize when heated, and thereby change its value of k, but we shall for simplicity assume that k is in fact constant with k = 3 for monatomic, k = 5 for diatomic, and k = 6 for multiatomic gases. For mixtures of gases the number of degrees of freedom is the molar average o ...
... gas may dissociate or even ionize when heated, and thereby change its value of k, but we shall for simplicity assume that k is in fact constant with k = 3 for monatomic, k = 5 for diatomic, and k = 6 for multiatomic gases. For mixtures of gases the number of degrees of freedom is the molar average o ...
Second Law of Thermodynamics
... Note that the Second Law does not address anything specifically about the entropy of the system, but only that of the universe (system + surroundings). A system process is defined as reversible if a system, after having experienced several transformations, can be returned to its original state witho ...
... Note that the Second Law does not address anything specifically about the entropy of the system, but only that of the universe (system + surroundings). A system process is defined as reversible if a system, after having experienced several transformations, can be returned to its original state witho ...
CHAP4
... following (also assuming isobaric conditions): a) the change in internal energy b) the change in enthalpy c) the change in entropy 2. Calculate the change in entropy of 2 g of ice initially at -10 C which is converted to steam at 100 C due to heating. [ans: 17.3 J K-1] 3. A 200 g sample of dry air ...
... following (also assuming isobaric conditions): a) the change in internal energy b) the change in enthalpy c) the change in entropy 2. Calculate the change in entropy of 2 g of ice initially at -10 C which is converted to steam at 100 C due to heating. [ans: 17.3 J K-1] 3. A 200 g sample of dry air ...
Entropy, free energy and equilibrium
... original values without any change An irreversible process is one where the system and surroundings cannot be restored A reversible process produces the maximum possible work ...
... original values without any change An irreversible process is one where the system and surroundings cannot be restored A reversible process produces the maximum possible work ...
EQATION OF STATE IN FORM WHICH RELATES MOL FRACTION
... to know to calculate the mole fraction in a given system is its temperature, total pressure, and molarity of the substance for which we are calculating mole fraction. Note that since no parameters for other substances in the system are required for equations (4) and (5) we can easily transform from ...
... to know to calculate the mole fraction in a given system is its temperature, total pressure, and molarity of the substance for which we are calculating mole fraction. Note that since no parameters for other substances in the system are required for equations (4) and (5) we can easily transform from ...
PPT
... • First, we’ll figure out where vapor, liquid and ice are in equilibrium, and the energy changes associated with the transitions – Can calculate changes such as heat released so we can treat the energy changes in our parcel – Can also figure out if the change is favored (e.g., if the transition is a ...
... • First, we’ll figure out where vapor, liquid and ice are in equilibrium, and the energy changes associated with the transitions – Can calculate changes such as heat released so we can treat the energy changes in our parcel – Can also figure out if the change is favored (e.g., if the transition is a ...
Example 2 - The Graduate School | UNC Charlotte
... Why focus on conformational flexibility? A critical link between structure, stability and biological function is conformational flexibility, which is characterized by an ensemble of accessible states. Conformational flexibility regulates specificity in mechanical response as manifested in dynamics. ...
... Why focus on conformational flexibility? A critical link between structure, stability and biological function is conformational flexibility, which is characterized by an ensemble of accessible states. Conformational flexibility regulates specificity in mechanical response as manifested in dynamics. ...
documentstyle[12pt]{article}
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
Minimum energetic cost to maintain a target nonequilibrium state
... to be static parameters fixing the thermal transition rates and modify the steady-state distribution by introducing additional “control” transitions with transition rates {Mkl }, as in Fig. 2. As was outlined in our analysis of the two-state system at the beginning of this article, we assume that fo ...
... to be static parameters fixing the thermal transition rates and modify the steady-state distribution by introducing additional “control” transitions with transition rates {Mkl }, as in Fig. 2. As was outlined in our analysis of the two-state system at the beginning of this article, we assume that fo ...
Chapter 1
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
Equilibrium
... land-forming processes were responsible for the shape of the Earth’s surface, and the idea of a ‘balance of nature’ prevailed. This was expressed first through the graded profile of rivers for time-bound studies, and subsequently by the definition of other types of equilibrium such as dynamic, quasi ...
... land-forming processes were responsible for the shape of the Earth’s surface, and the idea of a ‘balance of nature’ prevailed. This was expressed first through the graded profile of rivers for time-bound studies, and subsequently by the definition of other types of equilibrium such as dynamic, quasi ...
Lecture 3: FIRST LAW OF THERMODYNAMICS
... The water cools to 0◦ . The ice warms to 0◦ . The ice melts. Finally, the sum of these energies is put back into the system to warm the water back up from 0◦ to its final temperature, or refreeze some ice. This is an example of using an equivalent process to make a thermodynamic problem mathematical ...
... The water cools to 0◦ . The ice warms to 0◦ . The ice melts. Finally, the sum of these energies is put back into the system to warm the water back up from 0◦ to its final temperature, or refreeze some ice. This is an example of using an equivalent process to make a thermodynamic problem mathematical ...
Statistical mechanics
... contains mathematical tools for dealing with large populations, to the study of the thermodynamic behavior of systems composed of a large number of particles. Statistical mechanics provides a framework for relating the microscopic properties of individual atoms and molecules to the macroscopic bulk ...
... contains mathematical tools for dealing with large populations, to the study of the thermodynamic behavior of systems composed of a large number of particles. Statistical mechanics provides a framework for relating the microscopic properties of individual atoms and molecules to the macroscopic bulk ...
The “Second Law” of Probability: Entropy Growth in the Central Limit
... Is it possible for a thermodynamic system to move from state A to state B without any net energy being put into the system from outside? A single experimental quantity, dubbed entropy, made it possible to decide the direction of thermodynamic changes. ...
... Is it possible for a thermodynamic system to move from state A to state B without any net energy being put into the system from outside? A single experimental quantity, dubbed entropy, made it possible to decide the direction of thermodynamic changes. ...
The first law of thermodynamics
... This is an important concept in physics (wherein the net work being zero does not necessarily mean that no work was done ). In what other units have we explored this concept? ...
... This is an important concept in physics (wherein the net work being zero does not necessarily mean that no work was done ). In what other units have we explored this concept? ...
CHGN 351 A FALL 1999 PHYSICAL CHEMISTRY I: Quantum
... Physical chemistry is a demanding subject due both to its exceptional breadth and to its rigor. It is, however, these same two qualities which makes it so useful in scientific or engineering endeavors. The building of knowledge is both horizontal (varied principles) and vertical (principles building ...
... Physical chemistry is a demanding subject due both to its exceptional breadth and to its rigor. It is, however, these same two qualities which makes it so useful in scientific or engineering endeavors. The building of knowledge is both horizontal (varied principles) and vertical (principles building ...
Entropy
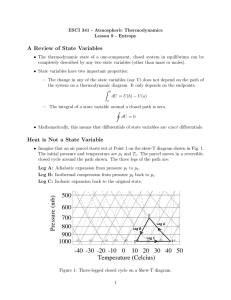
... • The thermodynamic state of a one-component, closed system in equilibrium can be completely described by any two state variables (other than mass or moles). • State variables have two important properties: – The change in any of the state variables (say U) does not depend on the path of the system ...
... • The thermodynamic state of a one-component, closed system in equilibrium can be completely described by any two state variables (other than mass or moles). • State variables have two important properties: – The change in any of the state variables (say U) does not depend on the path of the system ...











![documentstyle[12pt]{article}](http://s1.studyres.com/store/data/010234315_1-392ad57a1bf5b2aaeca94206588a5307-300x300.png)