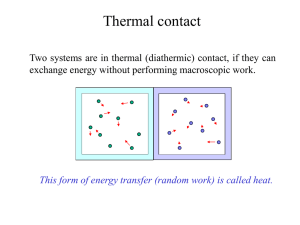
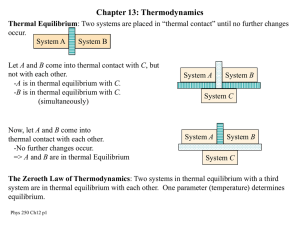
Heat and Thermodynamics
... system with time, it is said to give us the direction of "time's arrow " . If snapshots of a system at two different times shows one state which is more disordered, then it could be implied that this state came later in time. For an isolated system, the natural course of events takes the system to a ...
... system with time, it is said to give us the direction of "time's arrow " . If snapshots of a system at two different times shows one state which is more disordered, then it could be implied that this state came later in time. For an isolated system, the natural course of events takes the system to a ...
Entropy, Carnot Engine and Thermoelectric Effect
... Mechanical Equivalent of Heat : The amount of dissipated mechanical energy is directly proportional to the amount of heat produced. WQ W = JQ J= Mechanical Equivalent of heat = 4.18*107 ergs/cal Thermodynamical Variables : Quantities which determine the state of system are known as thermodynamical ...
... Mechanical Equivalent of Heat : The amount of dissipated mechanical energy is directly proportional to the amount of heat produced. WQ W = JQ J= Mechanical Equivalent of heat = 4.18*107 ergs/cal Thermodynamical Variables : Quantities which determine the state of system are known as thermodynamical ...
Measuring Temperature
... Heat depends on temperature, but also on the mass of the object, and its heat capacity. Even though Lake Ontario is at a colder temperature than your cup of coffee, it contains a lot more heat. The reason is that Lake Ontario is so much bigger (more massive) than your morning beverage. ...
... Heat depends on temperature, but also on the mass of the object, and its heat capacity. Even though Lake Ontario is at a colder temperature than your cup of coffee, it contains a lot more heat. The reason is that Lake Ontario is so much bigger (more massive) than your morning beverage. ...
T - Massey University
... The two forms of energy that influence this internal energy are heat, either transferred to the system from a source at higher temperature or lost to a sink at lower temperature and work, which can increase the internal energy if work is done on the system by its surroundings, or decrease the intern ...
... The two forms of energy that influence this internal energy are heat, either transferred to the system from a source at higher temperature or lost to a sink at lower temperature and work, which can increase the internal energy if work is done on the system by its surroundings, or decrease the intern ...
15-7 Entropy and the Second Law of Thermodynamics
... Entropy is sometimes referred to as time’s arrow – time proceeds in the direction of increasing entropy. Imagine watching a science fiction movie in which a spacecraft in deep space explodes into a million pieces. Then you play the film backwards, and see the million pieces magically come together t ...
... Entropy is sometimes referred to as time’s arrow – time proceeds in the direction of increasing entropy. Imagine watching a science fiction movie in which a spacecraft in deep space explodes into a million pieces. Then you play the film backwards, and see the million pieces magically come together t ...
Using the “Clicker”
... In general, efficiency is the ratio of the work done divided by the heat needed to do the work. e ...
... In general, efficiency is the ratio of the work done divided by the heat needed to do the work. e ...