16.050 Thermal Energy
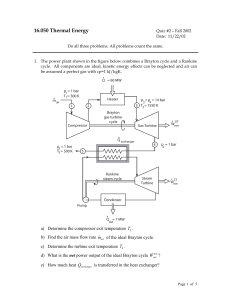
... between pressures p1 = 1.6 bar and p2 = 6 bar. The refrigeration cycle consists of four processes as sketched in the figure below. The refrigerant is compressed ideally from a saturated vapor at station 1 to a superheated gas at station 2, cooled in a heat exchanger from superheated gas to a saturat ...
... between pressures p1 = 1.6 bar and p2 = 6 bar. The refrigeration cycle consists of four processes as sketched in the figure below. The refrigerant is compressed ideally from a saturated vapor at station 1 to a superheated gas at station 2, cooled in a heat exchanger from superheated gas to a saturat ...
Document
... ∆H – T∆S / ∆G ∆G is negative Y Process is spontaneous ∆G is zero Y Process is at equilibrium ∆G is positive Y Process is non-spontaneous ...
... ∆H – T∆S / ∆G ∆G is negative Y Process is spontaneous ∆G is zero Y Process is at equilibrium ∆G is positive Y Process is non-spontaneous ...
Thermodynamics - myersparkphysics
... Heat flows spontaneously from a substance at a higher temperature to a substance at a lower temperature and does not flow spontaneously in the reverse direction. ...
... Heat flows spontaneously from a substance at a higher temperature to a substance at a lower temperature and does not flow spontaneously in the reverse direction. ...
Lecture 1 1 Overview
... another extensive property. Example: V → V /N . 2. Notation. Inconsistent notation has plagued thermodynamics perhaps since the beginning. The key in reading any text is to find out what the notation means before you read. There are basically four types of variables: (a) Extensive: e.g. V , U , N , ...
... another extensive property. Example: V → V /N . 2. Notation. Inconsistent notation has plagued thermodynamics perhaps since the beginning. The key in reading any text is to find out what the notation means before you read. There are basically four types of variables: (a) Extensive: e.g. V , U , N , ...
Equivalence of Kelvin-Planck and Clausius statements
... • A PMM2 according to Kelvin-Planck statement is a device that: Operates in a cycle. Accepts heat from a single reservoir (surroundings). Develops a net work output. ...
... • A PMM2 according to Kelvin-Planck statement is a device that: Operates in a cycle. Accepts heat from a single reservoir (surroundings). Develops a net work output. ...
Lecture_1_ Heat and - Arizona State University
... Sign convention: The work is taken negative if it increases the energy in the system. If the volume of the system is decreased work is done on the system, increasing its energy; hence the positive sign in the equation W pdV . The unusual convention was established to fit the behavior of heat engi ...
... Sign convention: The work is taken negative if it increases the energy in the system. If the volume of the system is decreased work is done on the system, increasing its energy; hence the positive sign in the equation W pdV . The unusual convention was established to fit the behavior of heat engi ...
Introduction in energy systems - Faculty of Mechanical Engineering
... thermodynamics to visualize changes to temperature and specific entropy during a thermodynamic process or cycle. It is a useful and common tool, particularly because it helps to visualize the heat transfer during a process. For reversible (ideal) processes, the area under the T-s curve of a process ...
... thermodynamics to visualize changes to temperature and specific entropy during a thermodynamic process or cycle. It is a useful and common tool, particularly because it helps to visualize the heat transfer during a process. For reversible (ideal) processes, the area under the T-s curve of a process ...