any physical system, whether or not it can exchange energy and
... Entropy determines the arrow of time ...
... Entropy determines the arrow of time ...
File
... Laws of thermodynamics The four laws of thermodynamics are: • Zeroth law of thermodynamics: If two systems are in thermal equilibrium separately, with a third system, they must be in thermal equilibrium with each other. This law helps define the notion of temperature. • First law of thermodynamics: ...
... Laws of thermodynamics The four laws of thermodynamics are: • Zeroth law of thermodynamics: If two systems are in thermal equilibrium separately, with a third system, they must be in thermal equilibrium with each other. This law helps define the notion of temperature. • First law of thermodynamics: ...
Entropy change of an ideal gas determination with no reversible
... In simulation (b) the gas reaches the final equilibrium much faster than in simulation (a). This happens because in case (a) the walls have a lower thermal conductivity than in the case (b), which explains why the temperature rises over 400 K, due to the strong gas compression, before it decreases r ...
... In simulation (b) the gas reaches the final equilibrium much faster than in simulation (a). This happens because in case (a) the walls have a lower thermal conductivity than in the case (b), which explains why the temperature rises over 400 K, due to the strong gas compression, before it decreases r ...
thermodynamics - La Salle High School
... Convention: Positive: heat flows into system work done onto system Negative: heat flows out of system work done by system ...
... Convention: Positive: heat flows into system work done onto system Negative: heat flows out of system work done by system ...
Lecture 1
... All heat engines have a maximum efficiency that is much less than 100% because of the second law of thermodynamics. No engine can exceed Carnot efficiency, because heat does not flow spontaneously from cold to hot. “No process is possible whose sole result is the transfer of heat from a body of lowe ...
... All heat engines have a maximum efficiency that is much less than 100% because of the second law of thermodynamics. No engine can exceed Carnot efficiency, because heat does not flow spontaneously from cold to hot. “No process is possible whose sole result is the transfer of heat from a body of lowe ...
1 Lecture: 2 Thermodynamic equilibrium 1
... Expansion against a constant pressure In this case the system does work against a constant force so that we can take it out of the ...
... Expansion against a constant pressure In this case the system does work against a constant force so that we can take it out of the ...
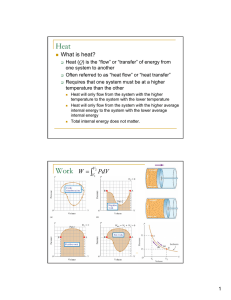
Q - UCSB Physics
... • Q – W has same value for all processes • Q – W depends only on initial, final state • Q – W is path-independent (these are three equivalent statements) ...
... • Q – W has same value for all processes • Q – W depends only on initial, final state • Q – W is path-independent (these are three equivalent statements) ...
Bagian 2 termodinamika
... Three different experiments are run, in which a gas expands from point A to point D along the three paths shown below. Calculate the amount of work done ...
... Three different experiments are run, in which a gas expands from point A to point D along the three paths shown below. Calculate the amount of work done ...