X-ray photoelectron spectroscopy - An introduction
... variations in the band bending and, thus, the work function will vary from point to point. This variation in surface potential produces a broadening of the XPS peaks. -Excitation process such as the shake-up/shake-off processes or vibrational broadening. ...
... variations in the band bending and, thus, the work function will vary from point to point. This variation in surface potential produces a broadening of the XPS peaks. -Excitation process such as the shake-up/shake-off processes or vibrational broadening. ...
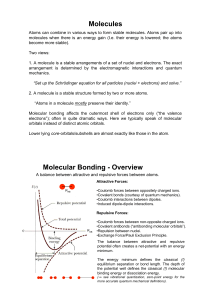
Molecules Molecular Bonding
... Atoms can combine in various ways to form stable molecules. Atoms pair up into molecules when there is an energy gain (I.e. their energy is lowered; the atoms become more stable). Two views: 1. A molecule is a stable arrangements of a set of nuclei and electrons. The exact arrangement is determined ...
... Atoms can combine in various ways to form stable molecules. Atoms pair up into molecules when there is an energy gain (I.e. their energy is lowered; the atoms become more stable). Two views: 1. A molecule is a stable arrangements of a set of nuclei and electrons. The exact arrangement is determined ...
AP Chemistry Study Guide – Chapter 7, Atomic Structure
... 6) Account for each of the following in terms of principles of atomic structure, including the number, properties, and arrangements of subatomic particles. (a) The second ionization energy of sodium is about three times greater than the second ionization energy of magnesium. (b) The difference betwe ...
... 6) Account for each of the following in terms of principles of atomic structure, including the number, properties, and arrangements of subatomic particles. (a) The second ionization energy of sodium is about three times greater than the second ionization energy of magnesium. (b) The difference betwe ...
Easy explanation
... A little more history Rutherford conducts gold foil experiment in 1911 • Directed high-energy, positively-charged alpha particles (helium ions from radon) at gold foil surrounded by cylindrical detector • Thomson’s model = no massive concentration of positive charge = nothing to significantly defle ...
... A little more history Rutherford conducts gold foil experiment in 1911 • Directed high-energy, positively-charged alpha particles (helium ions from radon) at gold foil surrounded by cylindrical detector • Thomson’s model = no massive concentration of positive charge = nothing to significantly defle ...
X-ray photoelectron spectroscopy - An introduction
... variations in the band bending and, thus, the work function will vary from point to point. This variation in surface potential produces a broadening of the XPS peaks. -Excitation process such as the shake-up/shake-off processes or vibrational broadening. ...
... variations in the band bending and, thus, the work function will vary from point to point. This variation in surface potential produces a broadening of the XPS peaks. -Excitation process such as the shake-up/shake-off processes or vibrational broadening. ...
Chem 1st Sem Rev Ch2
... 1. Arrange these elements in order of decreasing atomic size: sulfur, chlorine, aluminum and sodium. Does your arrangement demonstrate a periodic or group trend? 2. Distinguish between the first and second ionization energies of an atom. 3. Indicate which element in each of the following pairs has t ...
... 1. Arrange these elements in order of decreasing atomic size: sulfur, chlorine, aluminum and sodium. Does your arrangement demonstrate a periodic or group trend? 2. Distinguish between the first and second ionization energies of an atom. 3. Indicate which element in each of the following pairs has t ...
The end
... b/ One milliwatt of light of wavelength 4,560A is incident on a caesium surface. Calculate the electron current liberated and the minimum stopping voltage necessary to reduce this current to zero. Work function of caesium is 1.93 volts. Assume a quantum efficiency of 0.5 % (that means only 0,5 % of ...
... b/ One milliwatt of light of wavelength 4,560A is incident on a caesium surface. Calculate the electron current liberated and the minimum stopping voltage necessary to reduce this current to zero. Work function of caesium is 1.93 volts. Assume a quantum efficiency of 0.5 % (that means only 0,5 % of ...
Chapter 21 Powerpoint: Nuclear Chemistry
... 3. Identify the type of particle that has decayed and write it after the arrow. 4. Balance the mass number (top) and the atomic number (bottom). 5. Identify the new element. ...
... 3. Identify the type of particle that has decayed and write it after the arrow. 4. Balance the mass number (top) and the atomic number (bottom). 5. Identify the new element. ...
264-lecture-2015-10
... Quantum Mechanics Mnemonic for p “Now I need a drink, alcoholic of course, after the heavy lectures involving quantum mechanics.” ...
... Quantum Mechanics Mnemonic for p “Now I need a drink, alcoholic of course, after the heavy lectures involving quantum mechanics.” ...
P114 PROBLEM SET 9 Spring 2014
... [2] Your friend, who is exactly the same age as you, travels at a speed of 0.9990c to a star 25 light-years away. She spends 10 years on one of the star’s planets and then returns at 0.999c. (a) How long has she been away as measured by you? (b) How long does she measure for the time she is away? [3 ...
... [2] Your friend, who is exactly the same age as you, travels at a speed of 0.9990c to a star 25 light-years away. She spends 10 years on one of the star’s planets and then returns at 0.999c. (a) How long has she been away as measured by you? (b) How long does she measure for the time she is away? [3 ...
IQSE Banner News Page
... is known. Scattering angles of 120° and 140° have been used with some success in regions where the volume scattering function is similar to the one that is assumed. But this is not always the case— especially in dense plankton blooms. The volume scattering function may even have an azimuthal depende ...
... is known. Scattering angles of 120° and 140° have been used with some success in regions where the volume scattering function is similar to the one that is assumed. But this is not always the case— especially in dense plankton blooms. The volume scattering function may even have an azimuthal depende ...
Modern physics 2330
... 4- ( ) X-rays are produced by bombarding a metal target with energetic electrons having energies of 50 to 100 KeV. 5- (..) A black hole is an object of sufficiently high density can trap light forever. 6- ( ) Millikan’s oil drop experiment is inaccurate because it is difficult to measure the speed o ...
... 4- ( ) X-rays are produced by bombarding a metal target with energetic electrons having energies of 50 to 100 KeV. 5- (..) A black hole is an object of sufficiently high density can trap light forever. 6- ( ) Millikan’s oil drop experiment is inaccurate because it is difficult to measure the speed o ...
lect10
... which the electrons orbit the nucleus in accordance with Newton’s Laws but do not radiate energy an atom emits or absorbs energy only when an electron moves from one stable state to another ...
... which the electrons orbit the nucleus in accordance with Newton’s Laws but do not radiate energy an atom emits or absorbs energy only when an electron moves from one stable state to another ...
Biology Fall Semester Test 1 Study Guide
... Two products of cellular respiration are: In producers, chlorophyll and sunlight are necessary for the process of: The closing of its shell when a clam is removed from its watery environment is an example of how a clam maintains its: In a trophic pyramid, _______% of the energy from a source is pass ...
... Two products of cellular respiration are: In producers, chlorophyll and sunlight are necessary for the process of: The closing of its shell when a clam is removed from its watery environment is an example of how a clam maintains its: In a trophic pyramid, _______% of the energy from a source is pass ...
IB HL Physics More Problems on Quantum and Nuclear Physics_
... In 1924, Davisson and Germer carried out an experiment in which electrons were accelerated through a potential difference of 54 V. The electrons were scattered at the surface of a nickel crystal. (i) ...
... In 1924, Davisson and Germer carried out an experiment in which electrons were accelerated through a potential difference of 54 V. The electrons were scattered at the surface of a nickel crystal. (i) ...
Chemistry Questions
... 4. An atomic mass unit is defined as exactly a. 1/16 the mass of 12C atom b. 1/12 the mass of 12C atom 5. The total number of electrons in the outer shell (energy level) of a sodium ion is 6. As the number of neutrons in the nucleus of a given atom of an element increases, the atomic number of that ...
... 4. An atomic mass unit is defined as exactly a. 1/16 the mass of 12C atom b. 1/12 the mass of 12C atom 5. The total number of electrons in the outer shell (energy level) of a sodium ion is 6. As the number of neutrons in the nucleus of a given atom of an element increases, the atomic number of that ...
Atomic Theory Worksheet
... 1. Describe what was involved in JJ Thomson’s study of the Cathode Ray Tube. ...
... 1. Describe what was involved in JJ Thomson’s study of the Cathode Ray Tube. ...
Intro Biochemistry/Ecology
... We are combining our biochemistry unit and introductory ecology units into one big topic ...
... We are combining our biochemistry unit and introductory ecology units into one big topic ...
Request for Mass Spectrometric Analysis
... Incomplete forms will cause samples to be rejected. Max turnaround time: 2 weeks. Write legibly. If your sample has special requirements (keep frozen until analysis, sensitive to acid, etc) please describe in Extra Notes. Samples can be submitted directly to room 318 or placed on the labeled counter ...
... Incomplete forms will cause samples to be rejected. Max turnaround time: 2 weeks. Write legibly. If your sample has special requirements (keep frozen until analysis, sensitive to acid, etc) please describe in Extra Notes. Samples can be submitted directly to room 318 or placed on the labeled counter ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.