Take silver atoms with an electron that has a moment of µz = −g e(e
... one above the other, where z is the up/down direction and the magnets have length l (not to be mixed up with the quantum number). Although in the area between the magnets in the Stern-Gerlach experiment the magnetic ~ is homogeneous, near the end of the passage thru the magnets, it becomes inhomofie ...
... one above the other, where z is the up/down direction and the magnets have length l (not to be mixed up with the quantum number). Although in the area between the magnets in the Stern-Gerlach experiment the magnetic ~ is homogeneous, near the end of the passage thru the magnets, it becomes inhomofie ...
namimg compounds
... As the number of named compounds increased it was obvious that if such common names were used, confusion would result. In 1787, in order to solve the problem, a scientist named Lavoisier established the principles for a systematic naming process. There are, however, some familiar compounds that are ...
... As the number of named compounds increased it was obvious that if such common names were used, confusion would result. In 1787, in order to solve the problem, a scientist named Lavoisier established the principles for a systematic naming process. There are, however, some familiar compounds that are ...
Atomic Structure Study Guide
... (2) Atoms of a given element are ___________ in all ways. (3) Atoms of different elements have different physical and chemical __________. (4) Atoms of different elements combine in simple, whole-number ratios to form _________. (5) In chemical reactions, atoms are rearranged, but are never ________ ...
... (2) Atoms of a given element are ___________ in all ways. (3) Atoms of different elements have different physical and chemical __________. (4) Atoms of different elements combine in simple, whole-number ratios to form _________. (5) In chemical reactions, atoms are rearranged, but are never ________ ...
Worksheet on Ionic and Atomic Size Trends
... 3. The size of atoms decreases from left to right across a period. 4. Na: 1s22s22p63s1 Cl: 1s22s22p63s23p5 Both of these electron configurations contain the same number of energy levels. The size of these atoms decreases because the number of protons increases from left to right, resulting in an inc ...
... 3. The size of atoms decreases from left to right across a period. 4. Na: 1s22s22p63s1 Cl: 1s22s22p63s23p5 Both of these electron configurations contain the same number of energy levels. The size of these atoms decreases because the number of protons increases from left to right, resulting in an inc ...
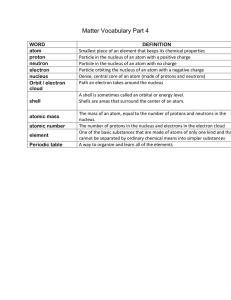
Matter Vocab Part 4
... Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes called an orbital or energy level. Shells are areas that surround the center of an atom. ...
... Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes called an orbital or energy level. Shells are areas that surround the center of an atom. ...
topic 1 sol review homework
... by the nucleus of the gold atoms. 1The nucleus is positively charged and so is the alpha particles so they are not attracted to each other. 2)They were used because the alpha particles would not be attracted to the nucleus. 9. Which atomic model has no subatomic particles? a) Rutherford b) Thomson c ...
... by the nucleus of the gold atoms. 1The nucleus is positively charged and so is the alpha particles so they are not attracted to each other. 2)They were used because the alpha particles would not be attracted to the nucleus. 9. Which atomic model has no subatomic particles? a) Rutherford b) Thomson c ...
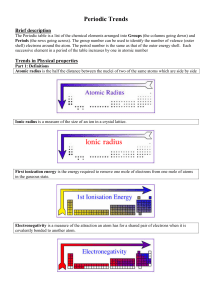
Topic 3 Periodicity notes SL - Chemical Minds
... The ionisation energy and electronegativity decrease because i) there is a decrease in the electrostatic attraction between the positive protons in the nucleus and the negative electrons in the electron shells orbiting the nucleus and ii) the valence electrons are shielded by the inner electron shel ...
... The ionisation energy and electronegativity decrease because i) there is a decrease in the electrostatic attraction between the positive protons in the nucleus and the negative electrons in the electron shells orbiting the nucleus and ii) the valence electrons are shielded by the inner electron shel ...
Modern Physics Lesson 3
... This can obviously only be used for objects with mass (i.e., NOT photons). It is usually used for electrons and protons, since they are the only objects that display noticeable wave-like properties. Normal sized objects (like a book or a basketball) also have a deBroglie wavelength, but it is so sma ...
... This can obviously only be used for objects with mass (i.e., NOT photons). It is usually used for electrons and protons, since they are the only objects that display noticeable wave-like properties. Normal sized objects (like a book or a basketball) also have a deBroglie wavelength, but it is so sma ...
5.2 Quantum Theory and the Atom
... Ground State: When all electrons are in their lowest possible energy level Excited State: When electrons gain energy and move to a higher energy level (when they go back to ground state they release energy in the form of COLOR) ...
... Ground State: When all electrons are in their lowest possible energy level Excited State: When electrons gain energy and move to a higher energy level (when they go back to ground state they release energy in the form of COLOR) ...
Atoms, Molecules, and Ions C Kapler ` , , I 27 O//#W SELF
... a. an Fe atom must gain 3 protons. b. an Fe2+ ion must gain 1 proton. c. an Fe atom must gain 3 electrons. d. an Fe2+ ion must gain 1 electron. e. none of the above will work. 8. Consider the following statements — There are always more neutrons than protons in an atom's nucleus. — The nucleus of an ...
... a. an Fe atom must gain 3 protons. b. an Fe2+ ion must gain 1 proton. c. an Fe atom must gain 3 electrons. d. an Fe2+ ion must gain 1 electron. e. none of the above will work. 8. Consider the following statements — There are always more neutrons than protons in an atom's nucleus. — The nucleus of an ...
1. Modern Physics
... Most positive charge and mass is in a small, centralized region (nucleus). A consequence of this is that there must be positive particles (protons). Electrons are “outside” the nucleus. Suggested that neutral particles consisting of proton plus electron might also be found (in the nucleus). ...
... Most positive charge and mass is in a small, centralized region (nucleus). A consequence of this is that there must be positive particles (protons). Electrons are “outside” the nucleus. Suggested that neutral particles consisting of proton plus electron might also be found (in the nucleus). ...
Regents questions
... Arranging the elements by atomic weight leads to an order slightly different from that in a modern periodic table, where the arrangement is by atomic number. Why does this happen? ...
... Arranging the elements by atomic weight leads to an order slightly different from that in a modern periodic table, where the arrangement is by atomic number. Why does this happen? ...
Chemistry 102B What`s in an atom? Before “Chemistry” Other Early
... given compound always contained the same proportions of certain elements by mass. “Law of Definite Proportions” • John Dalton (early 1800s) – noted that elements that combined to form more than one kind of compound, did so in proportions of integers by mass. “Law of Multiple Proportions” ...
... given compound always contained the same proportions of certain elements by mass. “Law of Definite Proportions” • John Dalton (early 1800s) – noted that elements that combined to form more than one kind of compound, did so in proportions of integers by mass. “Law of Multiple Proportions” ...
Models of the Atom:
... All elements are composed of submicroscopic, indivisible particles called atoms Atoms of the same element are identical. Atoms of different elements are not the same. Atoms of different elements can physically mix together or can chemically combine with one another in simple wholenumber ratios to fo ...
... All elements are composed of submicroscopic, indivisible particles called atoms Atoms of the same element are identical. Atoms of different elements are not the same. Atoms of different elements can physically mix together or can chemically combine with one another in simple wholenumber ratios to fo ...
File
... from the nucleus. • When an atom absorbs energy, the electron jumps to a level further from the nucleus • If it radiated energy, that means that the electron is falling to a level closer to the nucleus. ...
... from the nucleus. • When an atom absorbs energy, the electron jumps to a level further from the nucleus • If it radiated energy, that means that the electron is falling to a level closer to the nucleus. ...
Structure of matter.
... Solution of the Schrödinger Equation The solution of the Schrödinger equation for the electron in the hydrogen atom leads to the values of the energies of the orbital electron. The solution of the Schrödinger equation often leads to numerical coefficients which determine the possible values of ...
... Solution of the Schrödinger Equation The solution of the Schrödinger equation for the electron in the hydrogen atom leads to the values of the energies of the orbital electron. The solution of the Schrödinger equation often leads to numerical coefficients which determine the possible values of ...
Lab Science 9 Pacing Guide
... 6. Explain that the electric force between the nucleus and the electrons hold an atom together. Relate that on a larger scale, electric forces hold solid and liquid materials together (e.g., salt crystals and water). 7. Show how atoms may be bonded together by losing, gaining or sharing electrons an ...
... 6. Explain that the electric force between the nucleus and the electrons hold an atom together. Relate that on a larger scale, electric forces hold solid and liquid materials together (e.g., salt crystals and water). 7. Show how atoms may be bonded together by losing, gaining or sharing electrons an ...
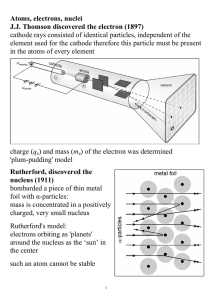
First Semester Honors Chemistry Exam Review (2011
... Dalton's atomic theory agrees with modern atomic theory except for what? The deflection of cathode rays in Thomson's experiments was evidence of the ____ nature of electrons. Whose series of experiments identified the nucleus of the atom? What happened to the alpha particles in Rutherford's experime ...
... Dalton's atomic theory agrees with modern atomic theory except for what? The deflection of cathode rays in Thomson's experiments was evidence of the ____ nature of electrons. Whose series of experiments identified the nucleus of the atom? What happened to the alpha particles in Rutherford's experime ...
HW8 not graded v3 - Department of Physics | Oregon State
... where E0 is an energy constant, the tail of the chlorophyll is 12 bonds long and the C-C bond length is 0.14 nm. ...
... where E0 is an energy constant, the tail of the chlorophyll is 12 bonds long and the C-C bond length is 0.14 nm. ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.