Earth`s Chemistry
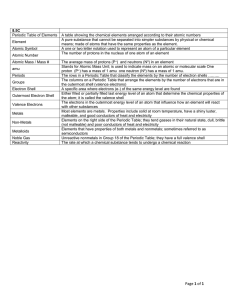
... All matter is made up of elements. Element = any substance that can’t break down any further (Ex. oxygen, iron, nitrogen, etc.). Periodic table --- made up of elements. Universal symbol for elements --- consists of either one letter or two (Note how it’s written) Over 90 elements occur naturally; ot ...
... All matter is made up of elements. Element = any substance that can’t break down any further (Ex. oxygen, iron, nitrogen, etc.). Periodic table --- made up of elements. Universal symbol for elements --- consists of either one letter or two (Note how it’s written) Over 90 elements occur naturally; ot ...
CHEM_1305_Practice_Exam_2
... 2) What is the general term that refers to either visible or invisible radiant energy? A) continuous spectrum ...
... 2) What is the general term that refers to either visible or invisible radiant energy? A) continuous spectrum ...
Quantum Mechanics
... Rutherford’s discovery of nucleus (1911) led to “solar system” model of atom Orbiting electrons contradicted e-m theory Niels Bohr (1913) proposed model of atom with electron orbits based on quantized energy states Difference between energy states always some multiple of Planck’s constant ...
... Rutherford’s discovery of nucleus (1911) led to “solar system” model of atom Orbiting electrons contradicted e-m theory Niels Bohr (1913) proposed model of atom with electron orbits based on quantized energy states Difference between energy states always some multiple of Planck’s constant ...
Document
... from Niels Bohr who explained experimentally observed discrete nature of atomic spectrum of Hydrogen. In spite of its immediate success in providing theoretical account of the spectrum and other nature of Hydrogen atom, a complete understanding of Bohr’s atom came only after de Broglie’s conjecture ...
... from Niels Bohr who explained experimentally observed discrete nature of atomic spectrum of Hydrogen. In spite of its immediate success in providing theoretical account of the spectrum and other nature of Hydrogen atom, a complete understanding of Bohr’s atom came only after de Broglie’s conjecture ...
Budiansky Cover
... "Once at the end of a colloquium I heard Debye saying something like: Schrödinger, you are not working right now on very important problems anyway. Why don't you tell us sometime about that thesis of de Broglie? "So in one of the next colloquia, Schrödinger gave a beautifully clear account of how de ...
... "Once at the end of a colloquium I heard Debye saying something like: Schrödinger, you are not working right now on very important problems anyway. Why don't you tell us sometime about that thesis of de Broglie? "So in one of the next colloquia, Schrödinger gave a beautifully clear account of how de ...
Honors Chemistry Week-At-A-Glance
... Review the basic structure of the atom Review calculation atomic mass Calculate wavelength, frequency, and energy of light emitted from atoms Formative Assessment: History, Discriminate between particles, and Atomic mass Quiz Partner Practice: Wavelength, frequency, energy, and electromagnetic ...
... Review the basic structure of the atom Review calculation atomic mass Calculate wavelength, frequency, and energy of light emitted from atoms Formative Assessment: History, Discriminate between particles, and Atomic mass Quiz Partner Practice: Wavelength, frequency, energy, and electromagnetic ...
Practice Exam 2 - Department of Chemistry and Biochemistry
... Use a softhead pencil, fill in you name, z-number, department name (CHEM), course name (110), and today's date () in the scantron sheet. ...
... Use a softhead pencil, fill in you name, z-number, department name (CHEM), course name (110), and today's date () in the scantron sheet. ...
Review 2nd KEY
... ____ 2. A spherical electron cloud surrounding an atomic nucleus would best represent a. an s orbital. c. a combination of px and py orbitals. b. a px orbital. d. a combination of an s and a px orbital. ____ 3. For an electron in an atom to change from the ground state to an excited state, a. energy ...
... ____ 2. A spherical electron cloud surrounding an atomic nucleus would best represent a. an s orbital. c. a combination of px and py orbitals. b. a px orbital. d. a combination of an s and a px orbital. ____ 3. For an electron in an atom to change from the ground state to an excited state, a. energy ...
S90 Notes U2 Topic 6 Chemical Compounds
... 1. Write the entire name of the first element. 2. Change the ending of the second element to end with –ide 3. Use a prefix to indicate the number of each type of atom in the formula: mono- for one, di- for two, tri- for three, and tetra- for four. The prefix mono- is used only for the second element ...
... 1. Write the entire name of the first element. 2. Change the ending of the second element to end with –ide 3. Use a prefix to indicate the number of each type of atom in the formula: mono- for one, di- for two, tri- for three, and tetra- for four. The prefix mono- is used only for the second element ...
Document
... and have the same de Broglie wavelength. Which of the following are also the same for the two particles: (a) speed, (b) kinetic energy, (c) momentum, (d) frequency? ...
... and have the same de Broglie wavelength. Which of the following are also the same for the two particles: (a) speed, (b) kinetic energy, (c) momentum, (d) frequency? ...
Honors Chemistry
... 9. What are the colors that make up white light? 10. Give the different waves of the magnetic spectrum. 11. Which wave has more energy: red or blue? Short or long? Microwave or x-ray? 12. What does Bohr’s Model say about the hydrogen atom? 13. What does it mean when an electron is excited? What happ ...
... 9. What are the colors that make up white light? 10. Give the different waves of the magnetic spectrum. 11. Which wave has more energy: red or blue? Short or long? Microwave or x-ray? 12. What does Bohr’s Model say about the hydrogen atom? 13. What does it mean when an electron is excited? What happ ...
Honors Chemistry Midterm Review 2008
... b. Volume Space taken up; graduated cylinder (ml); LxWxH (cm3) c. Density Ratio of Mass to volume; Mass/Volume g/ml or g/cm3 d. Time seconds stopwatch e. Temperature Average Kinetic Energy of molecules; thermometer °C or K thermometer. Absolute zero is 0K or -273°C K= °C + 273 f. Heat Form of Energy ...
... b. Volume Space taken up; graduated cylinder (ml); LxWxH (cm3) c. Density Ratio of Mass to volume; Mass/Volume g/ml or g/cm3 d. Time seconds stopwatch e. Temperature Average Kinetic Energy of molecules; thermometer °C or K thermometer. Absolute zero is 0K or -273°C K= °C + 273 f. Heat Form of Energy ...
Total intensity and quasi-elastic light
... The application of laser light-scattering techniques to the study of whole intact micro-organisms provides certain advantages and disadvantages compared with the study of smaller macromolecular assemblies and macromolecules. The advantages include the greater signal to noise ratio (i.e. the ‘dust pr ...
... The application of laser light-scattering techniques to the study of whole intact micro-organisms provides certain advantages and disadvantages compared with the study of smaller macromolecular assemblies and macromolecules. The advantages include the greater signal to noise ratio (i.e. the ‘dust pr ...
Lecture 1.1 Some preliminary chemistry knowledge, ppt file
... atom, the single electron is held in its orbital by its attraction to the proton in the nucleus. ...
... atom, the single electron is held in its orbital by its attraction to the proton in the nucleus. ...
File
... Data can frequently be better presented graphically with the independent variable plotted on the horizontal axis and the dependent variable plotted on the vertical axis. Axes should have regular scales and be clearly labelled with the units shown. The relationship between the variables is shown by a ...
... Data can frequently be better presented graphically with the independent variable plotted on the horizontal axis and the dependent variable plotted on the vertical axis. Axes should have regular scales and be clearly labelled with the units shown. The relationship between the variables is shown by a ...
The Egyptian American International School
... 1. The orbitals are different from the Bohr orbits. 2. Probability maps indicate the likelihood of finding the electron at a given point in space. 3. The size of an atom can be described by a surface that contains 90% of the total electron probability. 11.4 Electron Configurations and Atomic Propert ...
... 1. The orbitals are different from the Bohr orbits. 2. Probability maps indicate the likelihood of finding the electron at a given point in space. 3. The size of an atom can be described by a surface that contains 90% of the total electron probability. 11.4 Electron Configurations and Atomic Propert ...
lecture 7
... • We want to obtain the energy of the hydrogen atom system. We will do this the same way as we got it for the particle-in-a-box: by performing the “energy operation” on the wavefunction which describes the H atom system. ...
... • We want to obtain the energy of the hydrogen atom system. We will do this the same way as we got it for the particle-in-a-box: by performing the “energy operation” on the wavefunction which describes the H atom system. ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.