Test 1 - UTC.edu
... 14. Which one of the following statements about atoms and subatomic particles is correct? A) The proton and the neutron have identical masses. B) Rutherford discovered the atomic nucleus by bombarding gold foil with electrons C) The neutron's mass is equal to that of a proton plus an electron. D) An ...
... 14. Which one of the following statements about atoms and subatomic particles is correct? A) The proton and the neutron have identical masses. B) Rutherford discovered the atomic nucleus by bombarding gold foil with electrons C) The neutron's mass is equal to that of a proton plus an electron. D) An ...
Practice Unit D Exam - mvhs
... 2. Observations about real gases can be explained at the molecular level according to the kinetic molecular theory of gases and ideas about intermolecular forces. Explain how each of the following observations can be interpreted according to these concepts, including how the observation supports the ...
... 2. Observations about real gases can be explained at the molecular level according to the kinetic molecular theory of gases and ideas about intermolecular forces. Explain how each of the following observations can be interpreted according to these concepts, including how the observation supports the ...
ATOMS, MOLECULES and IONS
... A sample of any given ELEMENT is comprised of many atoms, all of which have the same atomic number; in Carbon, every atom has 6 e - and a nucleus with a charge of + 6 (ie; 6 protons). The atoms of each element have a unique number of protons, neutrons and electrons; it is these differing numbers tha ...
... A sample of any given ELEMENT is comprised of many atoms, all of which have the same atomic number; in Carbon, every atom has 6 e - and a nucleus with a charge of + 6 (ie; 6 protons). The atoms of each element have a unique number of protons, neutrons and electrons; it is these differing numbers tha ...
2009-10 Chemistry 1st Semester Final Exam Topics and Review
... Metric system Significant Figures Dimensional Analysis Density Physical and Chemical properties, Physical and chemical changes Matter- elements and compounds, mixtures and pure substances Elements, atoms, atomic structure- parts, location, charges, and masses. For any atom, ion, or isotope be able t ...
... Metric system Significant Figures Dimensional Analysis Density Physical and Chemical properties, Physical and chemical changes Matter- elements and compounds, mixtures and pure substances Elements, atoms, atomic structure- parts, location, charges, and masses. For any atom, ion, or isotope be able t ...
1305- practise exam 2
... C) Decreases from left to right; increases from bottom to top D) Decreases from left to right; decreases from bottom to top -------10. What is the predicted ionic charge for a S ion? A) 2+. B) 2C) 6+ D)6-------11. The compound H2O is classified as which of the following? A) Binary ionic B) Ternary i ...
... C) Decreases from left to right; increases from bottom to top D) Decreases from left to right; decreases from bottom to top -------10. What is the predicted ionic charge for a S ion? A) 2+. B) 2C) 6+ D)6-------11. The compound H2O is classified as which of the following? A) Binary ionic B) Ternary i ...
Final Exam Chemistry B2A Mr. Kimball`s Class 2003
... 22. Which of the following is FALSE regarding an electron? a) has a relative charge of -1 b) is abbreviated ec) has a mass of approximately 1 amu d) exists outside the nucleus e) exists in energy levels 23. Which of the following is FALSE regarding the nucleus of an atom? a) a small, low density reg ...
... 22. Which of the following is FALSE regarding an electron? a) has a relative charge of -1 b) is abbreviated ec) has a mass of approximately 1 amu d) exists outside the nucleus e) exists in energy levels 23. Which of the following is FALSE regarding the nucleus of an atom? a) a small, low density reg ...
Energy Atoms and Elements Practice Problems
... 18) Which of the following is NOT a statement of Dalton's Atomic Theory. A) All matter is made up of tiny indestructable particles called atoms. B) Atoms are niether created or destroyed during a chemical reaction, just rearranged. C) All atoms of a given element are identical and atoms of different ...
... 18) Which of the following is NOT a statement of Dalton's Atomic Theory. A) All matter is made up of tiny indestructable particles called atoms. B) Atoms are niether created or destroyed during a chemical reaction, just rearranged. C) All atoms of a given element are identical and atoms of different ...
Midterm Review Date
... B) low ionization energy and high electronegativity C) high ionization energy and low electronegativity D) high ionization energy and high electronegativity 22. Which element is an alkali metal? ...
... B) low ionization energy and high electronegativity C) high ionization energy and low electronegativity D) high ionization energy and high electronegativity 22. Which element is an alkali metal? ...
Chemistry Fall-2016 Final
... AA. any metal in Group 2A of the periodic table; generally harder, denser, stronger, and have higher melting points than alkali metals ...
... AA. any metal in Group 2A of the periodic table; generally harder, denser, stronger, and have higher melting points than alkali metals ...
Atoms and Elements
... putting electrons into orbitals that have the same energy as each other. Put one electron into each orbital before pairing them up. Whichever way the first arrow (electron) points, the others must point the same way until they pair up, then they point in opposite directions. ...
... putting electrons into orbitals that have the same energy as each other. Put one electron into each orbital before pairing them up. Whichever way the first arrow (electron) points, the others must point the same way until they pair up, then they point in opposite directions. ...
Valence electrons and Lewis Dot Structures
... The _________ _________ basically says that an atom wants to have 8 electrons in the s and p orbitals. It will gain, lose or share electrons in order to achieve this ...
... The _________ _________ basically says that an atom wants to have 8 electrons in the s and p orbitals. It will gain, lose or share electrons in order to achieve this ...
Name - cloudfront.net
... These are Dalton’s Postulate regarding the nature of the atom: 1. All matter consists of indivisible particles called atoms. 2. Atoms of the same element have the same shape and mass. 3. Atoms cannot be created or destroyed. 4. Atoms of different elements may combine with each other in fixed, simple ...
... These are Dalton’s Postulate regarding the nature of the atom: 1. All matter consists of indivisible particles called atoms. 2. Atoms of the same element have the same shape and mass. 3. Atoms cannot be created or destroyed. 4. Atoms of different elements may combine with each other in fixed, simple ...
1411-practice exam 2(ch4 5) - Chemistry
... Topic: Download(s): 0 Pages: 4 Published: 26 октября 2016 READ FULL DOCUMENT Please sign up to read full document. TEXT PREVIEW ...
... Topic: Download(s): 0 Pages: 4 Published: 26 октября 2016 READ FULL DOCUMENT Please sign up to read full document. TEXT PREVIEW ...
Scanning Electron Microscopy / Electron Probe X
... at sub micron scale. Energy dispersive spectrometers have the advantage that all X-ray energies are recorded simultaneously and the spectrum (see figure 4) is displayed directly by means of a multi-channel analyzer. A disadvantage of EDX is the generally poor detection limit in comparison to WDX. Th ...
... at sub micron scale. Energy dispersive spectrometers have the advantage that all X-ray energies are recorded simultaneously and the spectrum (see figure 4) is displayed directly by means of a multi-channel analyzer. A disadvantage of EDX is the generally poor detection limit in comparison to WDX. Th ...
Chem 430 - Fall 1994
... What is the mole fraction of Argon in air if the atmospheric pressure is 746.2 torr? Assume that air is made up of the following gasses with partial pressures in parenthesis. Nitrogen (595.4 torr); oxygen (141.8 torr) and argon (9.0 torr). ...
... What is the mole fraction of Argon in air if the atmospheric pressure is 746.2 torr? Assume that air is made up of the following gasses with partial pressures in parenthesis. Nitrogen (595.4 torr); oxygen (141.8 torr) and argon (9.0 torr). ...
name - cloudfront.net
... 6. A mass spectrometer works by ionizing atoms or molecules, and then accelerating them through oppositely ...
... 6. A mass spectrometer works by ionizing atoms or molecules, and then accelerating them through oppositely ...
Thursday, March 27, 2008
... What is the total number of hydrogen atoms required to form one molecule of C3H5(OH)3? ...
... What is the total number of hydrogen atoms required to form one molecule of C3H5(OH)3? ...
Atomic number
... • A neutron walks into a bar; he asks the bartender, 'How much for a beer?' The bartender looks at him, and says 'For you, no charge.' Two atoms bump into each other. One says 'I think I lost an electron!' The other asks, 'Are you sure?', to which the first replies, 'I'm positive.' ...
... • A neutron walks into a bar; he asks the bartender, 'How much for a beer?' The bartender looks at him, and says 'For you, no charge.' Two atoms bump into each other. One says 'I think I lost an electron!' The other asks, 'Are you sure?', to which the first replies, 'I'm positive.' ...
1. Which of the following statements best describes the
... Gas particles are packed closely together, but have some ability to move. ...
... Gas particles are packed closely together, but have some ability to move. ...
A Student want to prepare 250mL of .10 M NaCl solution
... correct drawing!! • Some things to look for – Octets on non-central atoms (all atoms if central is 1st row) – Happy elements – Charges • Negative usually carried by single bonded O (one for each charge) • Positive usually carried by a four bond N or P ...
... correct drawing!! • Some things to look for – Octets on non-central atoms (all atoms if central is 1st row) – Happy elements – Charges • Negative usually carried by single bonded O (one for each charge) • Positive usually carried by a four bond N or P ...
CHE 1401 - Fall 2013 - Chapter 7 Homework 7 (Chapter 7: Periodic
... A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more reactive than alkaline earth metals ...
... A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more reactive than alkaline earth metals ...
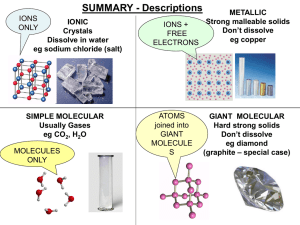
smart_materials_1 - Aldercar High School
... Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions ...
... Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions ...
CHEM1411,chapter 1-2-3 exercises 1. In 1828, the diameter of the
... 19. Calculate the percent composition by mass of carbon in Na2CO3. 20. Commonly used gases in the laboratory are generally obtained from pressurized metal gas cylinders, but for small amounts of occasionally used gases, it is sometimes easier just to prepare them chemically as needed. For example, n ...
... 19. Calculate the percent composition by mass of carbon in Na2CO3. 20. Commonly used gases in the laboratory are generally obtained from pressurized metal gas cylinders, but for small amounts of occasionally used gases, it is sometimes easier just to prepare them chemically as needed. For example, n ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.