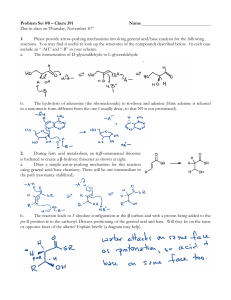
2. In aqueous solution, sodium hydroxide is a strong base and
... Define the terms strong acid and weak acid. Make sure you know properties of strong and weak acids in terms of dissociation, conductivity and reactivity. Using hydrochloric and ethanoic acid as examples, write equations to show the dissociation of each i n aqueous solution. *Be sure you are aware of ...
... Define the terms strong acid and weak acid. Make sure you know properties of strong and weak acids in terms of dissociation, conductivity and reactivity. Using hydrochloric and ethanoic acid as examples, write equations to show the dissociation of each i n aqueous solution. *Be sure you are aware of ...
8.1 Classifying inorganic compounds
... Acidity is a measure of the relative amounts of H+ and OH- ions in solution (Table 4 p.205) – the higher the number of H+ ions in solution the more acidic it is (opposite is true for basic ...
... Acidity is a measure of the relative amounts of H+ and OH- ions in solution (Table 4 p.205) – the higher the number of H+ ions in solution the more acidic it is (opposite is true for basic ...
IE EA
... bond dissociation energies of the N-H and P-H bonds and the electron affinities of NH2 and PH2 are needed. However, if one assumes that the differences in these quantities are about the same as the differences in the average bond dissociation energies (386 kJ mol-1 for NH3 and 322 kJ mol-1 for PH3) ...
... bond dissociation energies of the N-H and P-H bonds and the electron affinities of NH2 and PH2 are needed. However, if one assumes that the differences in these quantities are about the same as the differences in the average bond dissociation energies (386 kJ mol-1 for NH3 and 322 kJ mol-1 for PH3) ...
Iridoids and Valerian are not mentioned in the archive so please
... which is a plant in Europe but it's cultured in many countries, mainly in Africa but Kenya also produces different insecticide substance from this plant. The constituent of pyrethroid are monoterpenoid substances different in their structure of our common nucleus of GPP, by this condensation as IPP ...
... which is a plant in Europe but it's cultured in many countries, mainly in Africa but Kenya also produces different insecticide substance from this plant. The constituent of pyrethroid are monoterpenoid substances different in their structure of our common nucleus of GPP, by this condensation as IPP ...
Ester - SCH4U-SRB
... Esters are responsible for the “fruity” odours and flavours of many naturally occurring products. Chemists can reproduce these odours or flavours in the lab by mixing the right alcohol and carboxylic acid together; thus, producing “artificial” or “synthetic” versions of these naturally occurring com ...
... Esters are responsible for the “fruity” odours and flavours of many naturally occurring products. Chemists can reproduce these odours or flavours in the lab by mixing the right alcohol and carboxylic acid together; thus, producing “artificial” or “synthetic” versions of these naturally occurring com ...
(Acid Base 1).
... 2. The acid in question, if we have two equally concentrated solutions of acids, the solution of a strong acid will have a lower pH than that of a weak acid, because it is more fully dissociated and therefore produces more H3O+ ions. HCl, for example, is completely dissociated. ...
... 2. The acid in question, if we have two equally concentrated solutions of acids, the solution of a strong acid will have a lower pH than that of a weak acid, because it is more fully dissociated and therefore produces more H3O+ ions. HCl, for example, is completely dissociated. ...
Renal Physiology 9 (Acid Base 1)
... BASES (a.k.a. alkalis) – ions or molecules that can ACCEPT H+ (e.g., HCO3- + H+ H2CO3). • STRONG bases – dissociate easily in H2O and quickly bind H+. • WEAK bases – accept H+ more slowly (e.g., HCO3- and NH3) Proteins in body function as weak bases as some constituent AMINO ACIDS have net negat ...
... BASES (a.k.a. alkalis) – ions or molecules that can ACCEPT H+ (e.g., HCO3- + H+ H2CO3). • STRONG bases – dissociate easily in H2O and quickly bind H+. • WEAK bases – accept H+ more slowly (e.g., HCO3- and NH3) Proteins in body function as weak bases as some constituent AMINO ACIDS have net negat ...
A Floral Fragrance, Methyl Benzoate
... concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8°C higher than the boiling points o ...
... concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8°C higher than the boiling points o ...
DETERMINING THE CONCENTRATION OF A SOLUTION:
... compound that is produced when a carboxylic acid reacts with an alcohol. They are probably the most pleasant and delicious organic compounds one can study. A functional group is a specific arrangement of atoms in an organic compound that is capable of characteristic chemical reactions. The chemistry ...
... compound that is produced when a carboxylic acid reacts with an alcohol. They are probably the most pleasant and delicious organic compounds one can study. A functional group is a specific arrangement of atoms in an organic compound that is capable of characteristic chemical reactions. The chemistry ...
Solutions
... in a tautomeric form different from the one I usually draw, so that N9 is not protonated). ...
... in a tautomeric form different from the one I usually draw, so that N9 is not protonated). ...
Carboxylic Acids
... the name of the parent chain and is replaced -anoic acid. Since a carboxylic acid group must always lie at the end of a carbon chain, it is always is given the #1 location position in numbering and it is not necessary to include it in the name. Many carboxylic acids are called by the common names. T ...
... the name of the parent chain and is replaced -anoic acid. Since a carboxylic acid group must always lie at the end of a carbon chain, it is always is given the #1 location position in numbering and it is not necessary to include it in the name. Many carboxylic acids are called by the common names. T ...
lab – preparation of esters name
... Fill a 400-mL beaker about half full with water. Heat the water to boiling, and then turn off the flame or the hot plate. 2. Prepare the ester. Place 10 drops of one of the organic acids in a dry test tube. If using a solid acid, use a small spatula full, about 0.08 g. Add 10 drops of one of the alc ...
... Fill a 400-mL beaker about half full with water. Heat the water to boiling, and then turn off the flame or the hot plate. 2. Prepare the ester. Place 10 drops of one of the organic acids in a dry test tube. If using a solid acid, use a small spatula full, about 0.08 g. Add 10 drops of one of the alc ...
An ester is a chemical compound that is formed when an organic
... Fill a 400-mL beaker about half full with water. Heat the water to boiling, and then turn off the flame or the hot plate. 2. Prepare the ester. Place 10 drops of one of the organic acids in a dry test tube. If using a solid acid, use a small spatula full, about 0.08 g. Add 10 drops of one of the alc ...
... Fill a 400-mL beaker about half full with water. Heat the water to boiling, and then turn off the flame or the hot plate. 2. Prepare the ester. Place 10 drops of one of the organic acids in a dry test tube. If using a solid acid, use a small spatula full, about 0.08 g. Add 10 drops of one of the alc ...
Esterification
... Pre- Lab Discussion: Esters are responsible for the smell of many fruits and perfumes. In this experiment, a microscale technique is used to prepare four different esters. The esters are identified by their odors. Objectives: Students will use qualitative skills to determine the esters formed in thi ...
... Pre- Lab Discussion: Esters are responsible for the smell of many fruits and perfumes. In this experiment, a microscale technique is used to prepare four different esters. The esters are identified by their odors. Objectives: Students will use qualitative skills to determine the esters formed in thi ...
Nitric Acid Fact Sheet
... 1. Nitric acid is not just another acid, it is a very powerful oxidising agent and can form explosive substances. 2. All uses of nitric acid even for simple glassware washing must be fully risk assessed and approved by your supervisor. Both the incidents above occurred during cleaning operations. 3. ...
... 1. Nitric acid is not just another acid, it is a very powerful oxidising agent and can form explosive substances. 2. All uses of nitric acid even for simple glassware washing must be fully risk assessed and approved by your supervisor. Both the incidents above occurred during cleaning operations. 3. ...
Acids, Bases, and Salts Section 1 Acids and Bases
... pH is determined by a universal indicator paper or pH meter Buffers – solutions containing ions that react with additional acids or bases to minimize their effects on pH. ...
... pH is determined by a universal indicator paper or pH meter Buffers – solutions containing ions that react with additional acids or bases to minimize their effects on pH. ...
Acids, Bases and Salts - White Plains Public Schools
... ending in –ic. (HCl is hydrochloric acid) ...
... ending in –ic. (HCl is hydrochloric acid) ...
Acids and Bases Intr.. - hrsbstaff.ednet.ns.ca
... Substances that can act like an acid in one reaction, and like a base in another type of reaction. • Example: baking soda’s anion HCO31. HCO3- + OH- < -- > CO3-2 + H2O (donates a H+, so acts like an acid) 2. HCO3- + H3O+ < -- > H2CO3 + H2O (accepts a H+, so acts like a base) ...
... Substances that can act like an acid in one reaction, and like a base in another type of reaction. • Example: baking soda’s anion HCO31. HCO3- + OH- < -- > CO3-2 + H2O (donates a H+, so acts like an acid) 2. HCO3- + H3O+ < -- > H2CO3 + H2O (accepts a H+, so acts like a base) ...
K b
... For any respectable weak acid, [H+] from HA will be much greater than [H+] from H2O If dissociation of HA is much greater than H2O dissociation, [A-] >>[OH-] ...
... For any respectable weak acid, [H+] from HA will be much greater than [H+] from H2O If dissociation of HA is much greater than H2O dissociation, [A-] >>[OH-] ...
Lecture 7a
... Food vs. Fuel debate (i.e., 80 gal/acre for soy and sunflower) 42 billion gallons of diesel in 2008 in the US: 525,000,000 acre (21 % of the US) If the gasoline is also included (135 billion gallons) about 85 % of the area is needed! ...
... Food vs. Fuel debate (i.e., 80 gal/acre for soy and sunflower) 42 billion gallons of diesel in 2008 in the US: 525,000,000 acre (21 % of the US) If the gasoline is also included (135 billion gallons) about 85 % of the area is needed! ...
CHEM121 Exam 4 ObjectivesW16
... Solvent, solute, electrolyte, nonelectrolyte, saturated, dilute, concentrated , solubility rules -calculate Molarity -use Molarity to find # of moles or volume Concentration-dependent Properties: -vapor pressure lowering -boiling point elevation -freezing/melting point depression -osmotic pressure T ...
... Solvent, solute, electrolyte, nonelectrolyte, saturated, dilute, concentrated , solubility rules -calculate Molarity -use Molarity to find # of moles or volume Concentration-dependent Properties: -vapor pressure lowering -boiling point elevation -freezing/melting point depression -osmotic pressure T ...
Weak Acids and Bases Practice -- Chemistry 121A
... Weak Acids and Bases Practice -- Chemistry 121A Hanson OK, what’s new here really is just percent ionization. The way to think of this is that it is just a form of percent yield. It runs from 0% (all reactants) to 100% (all products). This is simply more convenient than Q, which runs from 0 to infin ...
... Weak Acids and Bases Practice -- Chemistry 121A Hanson OK, what’s new here really is just percent ionization. The way to think of this is that it is just a form of percent yield. It runs from 0% (all reactants) to 100% (all products). This is simply more convenient than Q, which runs from 0 to infin ...
Ch2hon ppt part 3
... (a)Because there are four chlorine atoms present, the compound is silicon tetrachloride. (b)There are four phosphorus atoms and ten oxygen atoms present, so the compound is tetraphosphorus decoxide. Note that the “a” is omitted in “deca.” ...
... (a)Because there are four chlorine atoms present, the compound is silicon tetrachloride. (b)There are four phosphorus atoms and ten oxygen atoms present, so the compound is tetraphosphorus decoxide. Note that the “a” is omitted in “deca.” ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.