Study Questions
... b) At what pH is methyl red orange? c) What is the Ka of an indicator that changes color at pH 3.5? d) What is a good indicator to use when titrating sodium nitrite (NaNO2) with HCl? 3. How many ml of 2.00 M HNO3 would be required to neutralize 12.5 ml of 0.0800 M NH3? (optional: What is the pH of t ...
... b) At what pH is methyl red orange? c) What is the Ka of an indicator that changes color at pH 3.5? d) What is a good indicator to use when titrating sodium nitrite (NaNO2) with HCl? 3. How many ml of 2.00 M HNO3 would be required to neutralize 12.5 ml of 0.0800 M NH3? (optional: What is the pH of t ...
+ [O] - MrFisherChemistry
... Provided by mixture of potassium dichromate(VI), K2Cr2O7, and excess dilute sulphuric acid, H2SO4. Oxidant is represented by : [O] ...
... Provided by mixture of potassium dichromate(VI), K2Cr2O7, and excess dilute sulphuric acid, H2SO4. Oxidant is represented by : [O] ...
1 - contentextra
... Worksheet 8.1 Chapter 8: Acids and bases – glossary Acid rain Rain water that has a pH of less than about 5. It is an environmental problem, causing damage to living and non-living structures. It arises from acidic gases being released into the atmosphere from industrial sources. Alkali ...
... Worksheet 8.1 Chapter 8: Acids and bases – glossary Acid rain Rain water that has a pH of less than about 5. It is an environmental problem, causing damage to living and non-living structures. It arises from acidic gases being released into the atmosphere from industrial sources. Alkali ...
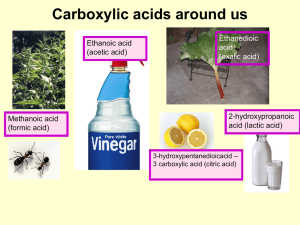
Unit4_Carboxylic Acid ppt
... Acids are named according to standard IUPAC rules But, many are still known under their trivial names, some having been called after characteristic properties or their origin. Formula HCOOH CH3COOH C6H5COOH ...
... Acids are named according to standard IUPAC rules But, many are still known under their trivial names, some having been called after characteristic properties or their origin. Formula HCOOH CH3COOH C6H5COOH ...
acids and bases - Althea`s Academy
... Pharmacology of the nitrate ion: 1. It has no specific action in the body 2. Potassium nitrate is a neutral salt but is the most potent diuretic of all the salts it acts by upsetting th eionic balance of the blood and tissue fluids ...
... Pharmacology of the nitrate ion: 1. It has no specific action in the body 2. Potassium nitrate is a neutral salt but is the most potent diuretic of all the salts it acts by upsetting th eionic balance of the blood and tissue fluids ...
Exp`t 88 - Chemistry Courses
... Ester syntheses are one of the more enjoyable experiments in organic chemistry, as one can't help but notice the special odors associated with esters - some fruity, some more like nail polish. In contrast, the acids which are used in their syntheses usually have a rotten odor. The reverse reaction, ...
... Ester syntheses are one of the more enjoyable experiments in organic chemistry, as one can't help but notice the special odors associated with esters - some fruity, some more like nail polish. In contrast, the acids which are used in their syntheses usually have a rotten odor. The reverse reaction, ...
Calculating a Ka Value from a Known pH - Chemwiki
... Ka, the acid ionization constant, is the equilibrium constant for chemical reactions involving weak acids in aqueous solution. The numerical value of Ka is used to predict the extent of acid dissociation. A large Ka value indicates a stronger acid (more of the acid dissociates) and small Ka val ...
... Ka, the acid ionization constant, is the equilibrium constant for chemical reactions involving weak acids in aqueous solution. The numerical value of Ka is used to predict the extent of acid dissociation. A large Ka value indicates a stronger acid (more of the acid dissociates) and small Ka val ...
Section II - School District 27J
... What is the molecular formula for this acid? (EC ~ NAME IT!) ...
... What is the molecular formula for this acid? (EC ~ NAME IT!) ...
Oxoacids of Phosphorus
... the phosphorous center being split by a single hydride (δ = 4 ppm, J P-H = 700 Hz). The 1 H NMR spectrum shows a doublet for the hydride and a single resonance of twice the intensity for the hydroxide. As expected, in water phosphorous acid is dibasic, (3). The acid (and the anions) are strong reduc ...
... the phosphorous center being split by a single hydride (δ = 4 ppm, J P-H = 700 Hz). The 1 H NMR spectrum shows a doublet for the hydride and a single resonance of twice the intensity for the hydroxide. As expected, in water phosphorous acid is dibasic, (3). The acid (and the anions) are strong reduc ...
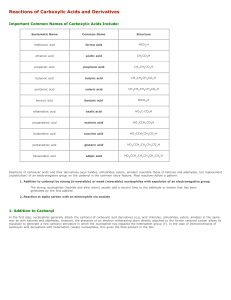
Reactions of Carboxylic Acids and Derivatives
... a reaction with a heteroatom nucleophile will work, the scheme shown below is useful. ...
... a reaction with a heteroatom nucleophile will work, the scheme shown below is useful. ...
Chem 30CL - Lecture 1d - UCLA Chemistry and Biochemistry
... convenient time for you to wake up again!) • In the lab, a racemic mixture of trans-1,2-diaminocyclohexane is provided • In order to synthesize the chiral ligand and the chiral catalyst in high enantiomeric purity, one enantiomer of the diamine is isolated that serves as a chiral backbone • (L)-(+)- ...
... convenient time for you to wake up again!) • In the lab, a racemic mixture of trans-1,2-diaminocyclohexane is provided • In order to synthesize the chiral ligand and the chiral catalyst in high enantiomeric purity, one enantiomer of the diamine is isolated that serves as a chiral backbone • (L)-(+)- ...
Acid Rain - Controlled Assessment
... During the last century the rain water in some parts of the world has become far more acidic. This acid rain has been caused by the emission of pollutant gases such as carbon dioxide, sulphur dioxide and nitrogen oxides. When coal is burned in electricity power stations, gases are released into the ...
... During the last century the rain water in some parts of the world has become far more acidic. This acid rain has been caused by the emission of pollutant gases such as carbon dioxide, sulphur dioxide and nitrogen oxides. When coal is burned in electricity power stations, gases are released into the ...
chemical equilibrium in a liquid phase
... water in the reaction mixtures. Methyl alcohol gives the highest value of K while ethyl alcohol gives the lowest. Branching of the chain as in i-butyl alcohol causes an appreciable rise in K over %-butyl alcohol, while the substitution of chlorine in ethyl alcohol has little effect. It is evident fr ...
... water in the reaction mixtures. Methyl alcohol gives the highest value of K while ethyl alcohol gives the lowest. Branching of the chain as in i-butyl alcohol causes an appreciable rise in K over %-butyl alcohol, while the substitution of chlorine in ethyl alcohol has little effect. It is evident fr ...
Module 02.indd
... prevent loss of any volatile production reactant. The reaction mixture needs to be heated to speed up the reaction and refluxing is used to prevent loss of alcohol reactant. ...
... prevent loss of any volatile production reactant. The reaction mixture needs to be heated to speed up the reaction and refluxing is used to prevent loss of alcohol reactant. ...
Biochemistry 311 Problem Set: pH and Buffer 1. Calculate the [H+] of
... 3. What is the [OH-] of 0.1 M HCl solution? 4. The K b for ammonia (NH3) is 1.8 x 10 -5 mole liter-l. What is its Ka value? 5. Calculate the pH of a 0.001 M solution of dichloroacetic acid, Ka 5.0 x 10 -2. 6. The pH of 0.1 M HCl is 1.0, of 0.1 M acetic acid is 2.8. What volume of 0.1 N NaOH would be ...
... 3. What is the [OH-] of 0.1 M HCl solution? 4. The K b for ammonia (NH3) is 1.8 x 10 -5 mole liter-l. What is its Ka value? 5. Calculate the pH of a 0.001 M solution of dichloroacetic acid, Ka 5.0 x 10 -2. 6. The pH of 0.1 M HCl is 1.0, of 0.1 M acetic acid is 2.8. What volume of 0.1 N NaOH would be ...
Carboxylic Acid Questions 1.-Arrange the following in order of (i
... Resonance stabilization in carboxylates makes carboxylic acids more acidic than alcohols or thiols. Electron withdrawing groups, here -Cl, increase the acidity due to further stabilization of the carboxylate. Thiols are more acidic than alcohols due to weaker X-H bond and the ability of S to accommo ...
... Resonance stabilization in carboxylates makes carboxylic acids more acidic than alcohols or thiols. Electron withdrawing groups, here -Cl, increase the acidity due to further stabilization of the carboxylate. Thiols are more acidic than alcohols due to weaker X-H bond and the ability of S to accommo ...
Session #31: homework Solution
... The structure of cysteine at pH = 7 shows that the side group is protonated. So we must conclude that even though the pKa is 8.33, the sulfhydryl (−SH) is acting as an acid. The isoelectric point, pI, is the pH at which the zwitterion is the dominant species. Let’s start with extreme acid conditions ...
... The structure of cysteine at pH = 7 shows that the side group is protonated. So we must conclude that even though the pKa is 8.33, the sulfhydryl (−SH) is acting as an acid. The isoelectric point, pI, is the pH at which the zwitterion is the dominant species. Let’s start with extreme acid conditions ...
Section 1 Sulfuric Acid, 50% v/v (1:1) Product
... Suitable Extinguishing Media If in laboratory setting, follow laboratory fire suppression procedures. Use appropriate suppression agents for adjacent combustibe materials or sources ignition. Use dry chemical, foam, or carbon dioxide to extinguish fire. Unsuitable Extinguishing Media Do not use wate ...
... Suitable Extinguishing Media If in laboratory setting, follow laboratory fire suppression procedures. Use appropriate suppression agents for adjacent combustibe materials or sources ignition. Use dry chemical, foam, or carbon dioxide to extinguish fire. Unsuitable Extinguishing Media Do not use wate ...
Preparation of Esters
... Esters are a group of organic compounds best known for their interesting odors and flavors. Many natural odors and flavors were discovered to be esters and therefore, many synthesized esters are used in perfumes and foods. ...
... Esters are a group of organic compounds best known for their interesting odors and flavors. Many natural odors and flavors were discovered to be esters and therefore, many synthesized esters are used in perfumes and foods. ...
Review Sheet Exam 2 3.4-4.7
... 15. Which one of the following is NOT a strong electrolyte? a. NaOH b. HBr c. KF d. HF 16. Which of the following is a weak electrolyte, circle all that apply. a. C2H6 b. CH3OH c. NH3 d. HNO3 e. CH3CO2H 17. Label the following with Strong, Weak, or Non- electrolyte. a. KOH b. H2SO4 c. C6H12O6(glucos ...
... 15. Which one of the following is NOT a strong electrolyte? a. NaOH b. HBr c. KF d. HF 16. Which of the following is a weak electrolyte, circle all that apply. a. C2H6 b. CH3OH c. NH3 d. HNO3 e. CH3CO2H 17. Label the following with Strong, Weak, or Non- electrolyte. a. KOH b. H2SO4 c. C6H12O6(glucos ...
Document
... reaction mixture at its boiling temperature to form products, without losing any of the compounds in the reaction flask. In practice, a condenser is set vertically into the top of the reaction flask. Any compound that vaporizes will condense when it enters the cool environment of the reflux condense ...
... reaction mixture at its boiling temperature to form products, without losing any of the compounds in the reaction flask. In practice, a condenser is set vertically into the top of the reaction flask. Any compound that vaporizes will condense when it enters the cool environment of the reflux condense ...
Problem: How would you prepare the following esters? a) Butyl
... Problem: Which method would you use to prepare cyclohexyl benzoate, Fischer esterfication or a reaction of an acid chloride with an alcohol? ...
... Problem: Which method would you use to prepare cyclohexyl benzoate, Fischer esterfication or a reaction of an acid chloride with an alcohol? ...
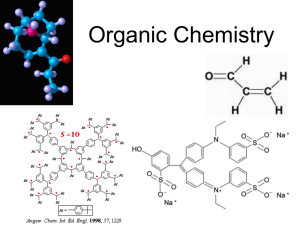
Organic Chemistry
... • How many isomers are there for pentane? • How many are there for hexane? • Draw and name as many octane isomers as you can (as a group) before the end of class. (1 pt. each) • Do the isomers page for homework. ...
... • How many isomers are there for pentane? • How many are there for hexane? • Draw and name as many octane isomers as you can (as a group) before the end of class. (1 pt. each) • Do the isomers page for homework. ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.
![+ [O] - MrFisherChemistry](http://s1.studyres.com/store/data/008194573_1-9c1e57b3af8f6a74ecb3216d2ce704f3-300x300.png)










![Biochemistry 311 Problem Set: pH and Buffer 1. Calculate the [H+] of](http://s1.studyres.com/store/data/016276514_1-cc9bfff072c2adb68721959b3f97d8e4-300x300.png)