Plasmonics: Fundamentals and Applications
... The remaining e( Za –Z) are the tightly bound electrons (known as core electrons). When these isolated atoms condense to form a metal, the core electrons remain bounded to the nucleus to form the metallic ions, but the valence electrons are free to move around and away from their parent atom. They ...
... The remaining e( Za –Z) are the tightly bound electrons (known as core electrons). When these isolated atoms condense to form a metal, the core electrons remain bounded to the nucleus to form the metallic ions, but the valence electrons are free to move around and away from their parent atom. They ...
12.50, £6.25. GA Jones. Pp. 175. London" Edward
... in the analysis of electron and X-ray diffraction patterns obtained in the study of materials. This volume contains 223 such diagrams, each of which displays some 300 plane normals. There are 23 projections of the cubic system, 28 of the hexagonal, and so on. To provide a distribution of axial ratio ...
... in the analysis of electron and X-ray diffraction patterns obtained in the study of materials. This volume contains 223 such diagrams, each of which displays some 300 plane normals. There are 23 projections of the cubic system, 28 of the hexagonal, and so on. To provide a distribution of axial ratio ...
Giant spin Seebeck effect in a non-magneticmaterial
... moments. This past year, the OSU team discovered that the effect is 1000 times larger in a material that contains no magnetism. This strange result is explained since the electrical charges in the material can also have magnetism due to a property called spin. The effect is strong enough that the OS ...
... moments. This past year, the OSU team discovered that the effect is 1000 times larger in a material that contains no magnetism. This strange result is explained since the electrical charges in the material can also have magnetism due to a property called spin. The effect is strong enough that the OS ...
SAMPLE QUESTIONS_and_ANSWERS
... dimensional photonic crystals. Can you think of analogous structures formed by atomic scale materials ? ...
... dimensional photonic crystals. Can you think of analogous structures formed by atomic scale materials ? ...
Chapter 2 - Cengage Learning
... • Oldest method of moving electrons – Permanently displaces an electron from an atom – Prolonged or steady flow of current is not possible – When charges are equalized, flow stops – Usually caused by friction ...
... • Oldest method of moving electrons – Permanently displaces an electron from an atom – Prolonged or steady flow of current is not possible – When charges are equalized, flow stops – Usually caused by friction ...
$doc.title
... Colloidal quantum dots (QD), also known as semiconductor nanocrystals, are a promising material platform for solution-‐processable optoelectronic devices, such as solar cells, light-‐ emitting diodes, thermoelectric ...
... Colloidal quantum dots (QD), also known as semiconductor nanocrystals, are a promising material platform for solution-‐processable optoelectronic devices, such as solar cells, light-‐ emitting diodes, thermoelectric ...
Periodic Table Test – Study Guide What state of matter are almost all
... What state of matter are almost all metals in at room temperature? Solid, liquid, or gas? What state of matter are nonmetals in at room temperature? Solid, liquid, or gas? What is similar for elements in a group/family on the periodic table? (2 things) number of valence e-, properties/reactivity Wha ...
... What state of matter are almost all metals in at room temperature? Solid, liquid, or gas? What state of matter are nonmetals in at room temperature? Solid, liquid, or gas? What is similar for elements in a group/family on the periodic table? (2 things) number of valence e-, properties/reactivity Wha ...
unit 4b hw packet File
... Pauli exclusion principle – maximum of 2 electrons in one orbital. Two electrons in one orbital will have opposite electron spin. ...
... Pauli exclusion principle – maximum of 2 electrons in one orbital. Two electrons in one orbital will have opposite electron spin. ...
1. The central core of the atom, containing protons and usually
... A representation of the number of valence electrons in an atom, using dots placed around the symbol of an element ...
... A representation of the number of valence electrons in an atom, using dots placed around the symbol of an element ...
Int. to Basic Electronics - Kashif Bashir
... •Such electrons that can move freely from one atom to atom to the next are often called free electrons. The movement of free electrons that provides electric current in a metal conductor. •When electrons can move easily from atom to atom in a material, it is a conductor. • In general all the metals ...
... •Such electrons that can move freely from one atom to atom to the next are often called free electrons. The movement of free electrons that provides electric current in a metal conductor. •When electrons can move easily from atom to atom in a material, it is a conductor. • In general all the metals ...
Ceramics Ceramics are inorganic and nonmetallic materials
... Ceramics are inorganic and nonmetallic materials. Ceramic Structures Because ceramics are composed of at least two elements, and often more, their crystal structures are generally more complex than those for metals Crystal Structures For those ceramic materials for which the atomic bonding is predom ...
... Ceramics are inorganic and nonmetallic materials. Ceramic Structures Because ceramics are composed of at least two elements, and often more, their crystal structures are generally more complex than those for metals Crystal Structures For those ceramic materials for which the atomic bonding is predom ...
Engineering Electromagnetic
... A conductor is something which allows electricity to flow through. Exam.: Scissors, Paper clip, Aluminum foil,Iron,Steel,Copper. In Conductors Valence electronics are present. When an electric field is applied ,then these electrons start moving which constitute an electric current. Conductors ...
... A conductor is something which allows electricity to flow through. Exam.: Scissors, Paper clip, Aluminum foil,Iron,Steel,Copper. In Conductors Valence electronics are present. When an electric field is applied ,then these electrons start moving which constitute an electric current. Conductors ...
on Fast Moving Electrons
... THE MINIMUM ENERGY REQUIRED TO EMIT AN ELECTRON FROM THE METAL SURFACE IS CALLED THE WORK FUNCTION. LOWER THE WORK FUNCTION, BETTER THE METAL IS AS A THERMION EMITTER. ...
... THE MINIMUM ENERGY REQUIRED TO EMIT AN ELECTRON FROM THE METAL SURFACE IS CALLED THE WORK FUNCTION. LOWER THE WORK FUNCTION, BETTER THE METAL IS AS A THERMION EMITTER. ...
Multielectron Atoms * The Independent Particle Approximation
... • Ground state: two electrons in 1s state (spin up/down) • Screening of each electron by the other • This results on a relatively large ionization energy (energy to remove an electron from a neutral atom) of 24.6 eV • Also large excitation energy (E2s-E1s) = 19.8 eV vs. 10.2 eV for H • Chemically in ...
... • Ground state: two electrons in 1s state (spin up/down) • Screening of each electron by the other • This results on a relatively large ionization energy (energy to remove an electron from a neutral atom) of 24.6 eV • Also large excitation energy (E2s-E1s) = 19.8 eV vs. 10.2 eV for H • Chemically in ...
The Drude Model and DC Conductivity
... In the absence of an electric field, the conduction electrons move in random directions through the conductor with average speeds v ~ 106 m/s. The drift velocity of the free electrons is zero. There is no current in the conductor since there is no net flow of ...
... In the absence of an electric field, the conduction electrons move in random directions through the conductor with average speeds v ~ 106 m/s. The drift velocity of the free electrons is zero. There is no current in the conductor since there is no net flow of ...
AO Electricity
... electrically neutral. Negative charges, being associated with electrons, are far more mobile in materials than positive charges are. A very small excess or deficit of negative charges in a material produces noticeable electric forces. Different kinds of materials respond differently to electric fo ...
... electrically neutral. Negative charges, being associated with electrons, are far more mobile in materials than positive charges are. A very small excess or deficit of negative charges in a material produces noticeable electric forces. Different kinds of materials respond differently to electric fo ...
Helpful PPT on Atoms and Bonding
... • If atom gains an electron it has an negative charge called an Anion ...
... • If atom gains an electron it has an negative charge called an Anion ...
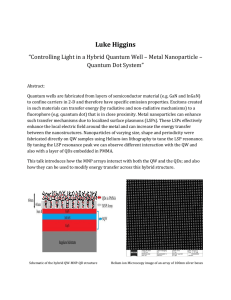
Luke Higgins Quantum Dot System”
... Quantum wells are fabricated from layers of semiconductor material (e.g. GaN and InGaN) to confine carriers in 2-D and therefore have specific emission properties. Excitons created in such materials can transfer energy (by radiative and non-radiative mechanisms) to a fluorophore (e.g. quantum dot) t ...
... Quantum wells are fabricated from layers of semiconductor material (e.g. GaN and InGaN) to confine carriers in 2-D and therefore have specific emission properties. Excitons created in such materials can transfer energy (by radiative and non-radiative mechanisms) to a fluorophore (e.g. quantum dot) t ...
III-Nitride Heterojunction Field Effect Transistors
... The physical basis, technology and performance of AlGaN/GaN FET’s with their undoped polarizationinduced, two-dimensional-electron-gases (2DEG’s) will be presented. The unique properties of the Wurtzite heterojunction crystals involved include 1x1013/cm2 electron sheet density, 3 megavolt/cm electri ...
... The physical basis, technology and performance of AlGaN/GaN FET’s with their undoped polarizationinduced, two-dimensional-electron-gases (2DEG’s) will be presented. The unique properties of the Wurtzite heterojunction crystals involved include 1x1013/cm2 electron sheet density, 3 megavolt/cm electri ...
Electricity (1)
... atoms…they can be moved. A concentration of electrons in an atom creates a net negative charge. If electrons are stripped away, the atom becomes positively charged. ...
... atoms…they can be moved. A concentration of electrons in an atom creates a net negative charge. If electrons are stripped away, the atom becomes positively charged. ...
Electricity and Magnetism Review Name: Directions: Answer the
... 1. Current electricity is the continuous flow of electrons through a conductor. 2. Materials that are a cross between conductors and insulators such as diodes and transistors are called semiconductors. 3. _____static electricity____ is the buildup of electrons on an object. 4. The interactions/prope ...
... 1. Current electricity is the continuous flow of electrons through a conductor. 2. Materials that are a cross between conductors and insulators such as diodes and transistors are called semiconductors. 3. _____static electricity____ is the buildup of electrons on an object. 4. The interactions/prope ...
semiconductor
... A semiconductor is a material which has electrical conductivity between that of a conductor such as copper and an insulator such as glass. The conductivity of a semiconductor increases with increasing temperature, behavior opposite to that of a metal. Semiconductors can display a range of useful pro ...
... A semiconductor is a material which has electrical conductivity between that of a conductor such as copper and an insulator such as glass. The conductivity of a semiconductor increases with increasing temperature, behavior opposite to that of a metal. Semiconductors can display a range of useful pro ...
Semiconductor
A semiconductor material has an electrical conductivity value falling between that of a conductor, such as copper, and an insulator, such as glass. Semiconductors are the foundation of modern electronics. Semiconducting materials exist in two types - elemental materials and compound materials. The modern understanding of the properties of a semiconductor relies on quantum physics to explain the movement of electrons and holes in a crystal lattice. The unique arrangement of the crystal lattice makes silicon and germanium the most commonly used elements in the preparation of semiconducting materials. An increased knowledge of semiconductor materials and fabrication processes has made possible continuing increases in the complexity and speed of microprocessors and memory devices. Some of the information on this page may be outdated within a year because new discoveries are made in the field frequently.The electrical conductivity of a semiconductor material increases with increasing temperature, which is behaviour opposite to that of a metal. Semiconductor devices can display a range of useful properties such as passing current more easily in one direction than the other, showing variable resistance, and sensitivity to light or heat. Because the electrical properties of a semiconductor material can be modified by controlled addition of impurities, or by the application of electrical fields or light, devices made from semiconductors can be used for amplification, switching, and energy conversion.Current conduction in a semiconductor occurs through the movement of free electrons and ""holes"", collectively known as charge carriers. Adding impurity atoms to a semiconducting material, known as ""doping"", greatly increases the number of charge carriers within it. When a doped semiconductor contains mostly free holes it is called ""p-type"", and when it contains mostly free electrons it is known as ""n-type"". The semiconductor materials used in electronic devices are doped under precise conditions to control the concentration and regions of p- and n-type dopants. A single semiconductor crystal can have many p- and n-type regions; the p–n junctions between these regions are responsible for the useful electronic behaviour.Some of the properties of semiconductor materials were observed throughout the mid 19th and first decades of the 20th century. Development of quantum physics in turn allowed the development of the transistor in 1947. Although some pure elements and many compounds display semiconductor properties, silicon, germanium, and compounds of gallium are the most widely used in electronic devices. Elements near the so-called ""metalloid staircase"", where the metalloids are located on the periodic table, are usually used as semiconductors.The nickname of the southern area of Northern California is Silicon Valley because of all the influential tech companies that have their headquarters there. An integral part of today’s technology is built upon semiconductors, which are made primarily of silicon. Some major companies include Marvell Technology Group, National Semiconductor (now part of Texas Instruments), and Advanced Micro Devices.