lecture2
... Crystalline and noncrystalline Materials: There are seven kinds of crystal systems shown on page ____. Next, step back from the single crystal and look at groupings of crystals. A material may have different levels of crystalline. a. Single crystal: entire substance formed from one large crystal ...
... Crystalline and noncrystalline Materials: There are seven kinds of crystal systems shown on page ____. Next, step back from the single crystal and look at groupings of crystals. A material may have different levels of crystalline. a. Single crystal: entire substance formed from one large crystal ...
Exploring the Science of Electricity
... Materials that are neither good conductors nor good insulators. Semiconductors are manufactured from elements having atoms with four electrons in their valence rings. Silicon and germanium are widely used in making semi-conducting materials. ...
... Materials that are neither good conductors nor good insulators. Semiconductors are manufactured from elements having atoms with four electrons in their valence rings. Silicon and germanium are widely used in making semi-conducting materials. ...
electron theory of metals
... It is a phenomenon related to the thermal conductivity of electrons in that it too is a consequence of the drift of electrons under a thermal gradient. These are two aspects to thermoelectric phenomena. The first is the Seeback effect in which a temperature between the two junctions of two dissimila ...
... It is a phenomenon related to the thermal conductivity of electrons in that it too is a consequence of the drift of electrons under a thermal gradient. These are two aspects to thermoelectric phenomena. The first is the Seeback effect in which a temperature between the two junctions of two dissimila ...
Chemistry and Material Science 1. Physical Properties of Materials
... as a solute dissolving in the copper solvent. This particular configuration is referred to as substitutional solid solution because the nickel atoms are substituting for Cu atoms on the fcc atom sites. This configuration will tend to occur when the atoms do not differ greatly in size. Solid Soluti ...
... as a solute dissolving in the copper solvent. This particular configuration is referred to as substitutional solid solution because the nickel atoms are substituting for Cu atoms on the fcc atom sites. This configuration will tend to occur when the atoms do not differ greatly in size. Solid Soluti ...
Plasmonics
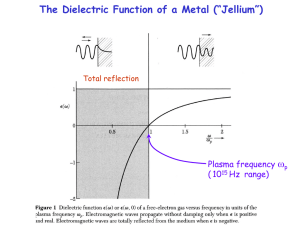
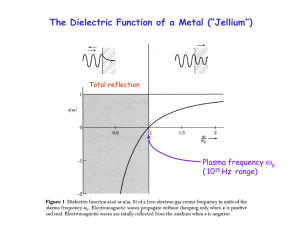
... fast for the electrons to follow. A metal loses its reflectivity. The corresponding photon energy is the plasmon energy Ep = ħp , typically 10-30 eV (deep into the ultraviolet). The reflectivity of aluminum cuts off at its plasmon energy Data (dashed) are compared to the jellium model (full). The d ...
... fast for the electrons to follow. A metal loses its reflectivity. The corresponding photon energy is the plasmon energy Ep = ħp , typically 10-30 eV (deep into the ultraviolet). The reflectivity of aluminum cuts off at its plasmon energy Data (dashed) are compared to the jellium model (full). The d ...
Lecture 30/31
... fast for the electrons to follow. A metal loses its reflectivity. The corresponding photon energy is the plasmon energy Ep = ħp , typically 10-30 eV (deep into the ultraviolet). The reflectivity of aluminum cuts off at its plasmon energy Data (dashed) are compared to the jellium model (full). The d ...
... fast for the electrons to follow. A metal loses its reflectivity. The corresponding photon energy is the plasmon energy Ep = ħp , typically 10-30 eV (deep into the ultraviolet). The reflectivity of aluminum cuts off at its plasmon energy Data (dashed) are compared to the jellium model (full). The d ...
electric field
... and a negative charge has to do with how they behave and the forces they exert. A FORCE is a push or a pull on an object. • When charged particles come near each other two different types of forces can occur… ...
... and a negative charge has to do with how they behave and the forces they exert. A FORCE is a push or a pull on an object. • When charged particles come near each other two different types of forces can occur… ...
Traditional versus PIN Diode Geiger Counter
... circumference of the wire the electrical field strength becomes very high and accelerates the charged carriers massively as they approach the wire such that they are able to cause a further ionization when they collide with further atoms in the vicinity of the wire. This causes an avalanche of charg ...
... circumference of the wire the electrical field strength becomes very high and accelerates the charged carriers massively as they approach the wire such that they are able to cause a further ionization when they collide with further atoms in the vicinity of the wire. This causes an avalanche of charg ...
Homework 1 - Devin Gatherwright IET 307 Portfolio
... bonds, like ionic bonds, are good insulators of heat and electricity. 4. What is Van der Waals bonding? How is it formed? What are its characteristics? Answer: According to the Materials Science textbook, Van der Waals or Secondary bonding is defined as a “secondary interatomic bond between neighbor ...
... bonds, like ionic bonds, are good insulators of heat and electricity. 4. What is Van der Waals bonding? How is it formed? What are its characteristics? Answer: According to the Materials Science textbook, Van der Waals or Secondary bonding is defined as a “secondary interatomic bond between neighbor ...
Quantum Confinement
... artificial periodicity will “fold” the Brillouin Zones into smaller BZ’s “mini-zones”. ...
... artificial periodicity will “fold” the Brillouin Zones into smaller BZ’s “mini-zones”. ...
electrictiy note packet Unit 6
... ~1850 James Joule - studied how energy can be transferred between different systems...... ........heat, mechanical, and electrical systems joules (J) - unit of energy II. Electrostatics A. Definition - study of electric charges, forces, and fields static electricity-electricity at rest, 'non-moving' ...
... ~1850 James Joule - studied how energy can be transferred between different systems...... ........heat, mechanical, and electrical systems joules (J) - unit of energy II. Electrostatics A. Definition - study of electric charges, forces, and fields static electricity-electricity at rest, 'non-moving' ...
Electric Charge
... Note that this behavior is different than gravitation in which the force is always attractive ...
... Note that this behavior is different than gravitation in which the force is always attractive ...
Covalent Bonds
... • Both atoms have very high ionization energies so neither atom will be able to take valence electrons completely off the other…they will share them • Because no electrons are transferred, there are no ...
... • Both atoms have very high ionization energies so neither atom will be able to take valence electrons completely off the other…they will share them • Because no electrons are transferred, there are no ...
Ceramic Crystal Structures
... structure similar to that of diamond. Alumina has a close packed hexagonal structure, with a mixture of covalent and ionic bonds, with one-third of the potential aluminum sites vacant in order to satisfy the valency requirements of the two elements. 2- Complex silicate structures: The majority of ce ...
... structure similar to that of diamond. Alumina has a close packed hexagonal structure, with a mixture of covalent and ionic bonds, with one-third of the potential aluminum sites vacant in order to satisfy the valency requirements of the two elements. 2- Complex silicate structures: The majority of ce ...
What`s This Thing Called “Current”?
... The usual definition of current is "the flow of electrons." This definition has been around for over a hundred years. If you do a search on Google for "flow of electrons" you will get over two million hits, most of them somehow related to this definition. The formal definition of one amp of current ...
... The usual definition of current is "the flow of electrons." This definition has been around for over a hundred years. If you do a search on Google for "flow of electrons" you will get over two million hits, most of them somehow related to this definition. The formal definition of one amp of current ...
Applied Materials Science
... An ability to conduct, analyze, and interpret experiments, as evidenced by the data and data analyses associated with numerous laboratory reports. ...
... An ability to conduct, analyze, and interpret experiments, as evidenced by the data and data analyses associated with numerous laboratory reports. ...
STRUCTURE OF AN ATOM
... gases are poor conductors of electricity at normal pressures but Discharge tube experiment-Production of by decreasing pressure they can behave as electrolytes ...
... gases are poor conductors of electricity at normal pressures but Discharge tube experiment-Production of by decreasing pressure they can behave as electrolytes ...
Polymers composed of a large number of repeating units. Isomers
... molecules with the same molecular formula but different chemical structures. ...
... molecules with the same molecular formula but different chemical structures. ...
AP TOPIC 3: Electronic Configuration
... Wave component of electrons All the work by Bohr suggested that the electron was a discrete particle. However this did not explain several of its properties. Work by De Broglie suggested that the electron also had some wave like characteristics. The description of the electron in this way is extreme ...
... Wave component of electrons All the work by Bohr suggested that the electron was a discrete particle. However this did not explain several of its properties. Work by De Broglie suggested that the electron also had some wave like characteristics. The description of the electron in this way is extreme ...
Part One
... In the pure form it is an electrical insulator When doped to be a semi-conductor it exhibits positive temperature of co-efficient of resistivity (PTCR) properties in the polycrystalline form Has high Tc and Pr over singe crystals Also BaTiO3 exhibits ferroelectric properties and is an excellen ...
... In the pure form it is an electrical insulator When doped to be a semi-conductor it exhibits positive temperature of co-efficient of resistivity (PTCR) properties in the polycrystalline form Has high Tc and Pr over singe crystals Also BaTiO3 exhibits ferroelectric properties and is an excellen ...
final1-executive-summary-harwin
... performance figures of U-value (0.4 W/m²/K) and VLT (75%) comparable or even superior to existing triple glazing can be achieved at almost 50% lower weight. The new extremely stiff and thermally insulating frame, based on polymer foam coreglass fiber reinforced polymer skin materials offers addition ...
... performance figures of U-value (0.4 W/m²/K) and VLT (75%) comparable or even superior to existing triple glazing can be achieved at almost 50% lower weight. The new extremely stiff and thermally insulating frame, based on polymer foam coreglass fiber reinforced polymer skin materials offers addition ...
MATERIALS
... B. Held together by chemical, physical bonds 1. Bonds holding atoms together a) Covalent bonding--two atoms share electrons. Very strong and rigid. Found in organic molecules and sometimes ceramics. Strongly directional. ...
... B. Held together by chemical, physical bonds 1. Bonds holding atoms together a) Covalent bonding--two atoms share electrons. Very strong and rigid. Found in organic molecules and sometimes ceramics. Strongly directional. ...
Semiconductor
A semiconductor material has an electrical conductivity value falling between that of a conductor, such as copper, and an insulator, such as glass. Semiconductors are the foundation of modern electronics. Semiconducting materials exist in two types - elemental materials and compound materials. The modern understanding of the properties of a semiconductor relies on quantum physics to explain the movement of electrons and holes in a crystal lattice. The unique arrangement of the crystal lattice makes silicon and germanium the most commonly used elements in the preparation of semiconducting materials. An increased knowledge of semiconductor materials and fabrication processes has made possible continuing increases in the complexity and speed of microprocessors and memory devices. Some of the information on this page may be outdated within a year because new discoveries are made in the field frequently.The electrical conductivity of a semiconductor material increases with increasing temperature, which is behaviour opposite to that of a metal. Semiconductor devices can display a range of useful properties such as passing current more easily in one direction than the other, showing variable resistance, and sensitivity to light or heat. Because the electrical properties of a semiconductor material can be modified by controlled addition of impurities, or by the application of electrical fields or light, devices made from semiconductors can be used for amplification, switching, and energy conversion.Current conduction in a semiconductor occurs through the movement of free electrons and ""holes"", collectively known as charge carriers. Adding impurity atoms to a semiconducting material, known as ""doping"", greatly increases the number of charge carriers within it. When a doped semiconductor contains mostly free holes it is called ""p-type"", and when it contains mostly free electrons it is known as ""n-type"". The semiconductor materials used in electronic devices are doped under precise conditions to control the concentration and regions of p- and n-type dopants. A single semiconductor crystal can have many p- and n-type regions; the p–n junctions between these regions are responsible for the useful electronic behaviour.Some of the properties of semiconductor materials were observed throughout the mid 19th and first decades of the 20th century. Development of quantum physics in turn allowed the development of the transistor in 1947. Although some pure elements and many compounds display semiconductor properties, silicon, germanium, and compounds of gallium are the most widely used in electronic devices. Elements near the so-called ""metalloid staircase"", where the metalloids are located on the periodic table, are usually used as semiconductors.The nickname of the southern area of Northern California is Silicon Valley because of all the influential tech companies that have their headquarters there. An integral part of today’s technology is built upon semiconductors, which are made primarily of silicon. Some major companies include Marvell Technology Group, National Semiconductor (now part of Texas Instruments), and Advanced Micro Devices.