* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Multielectron Atoms * The Independent Particle Approximation
Survey
Document related concepts
Electron paramagnetic resonance wikipedia , lookup
Electromotive force wikipedia , lookup
Electromagnetism wikipedia , lookup
Electrical resistivity and conductivity wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Photoelectric effect wikipedia , lookup
Transcript
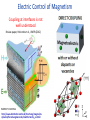
Physics 471: Solid State Physics Professor Micky Holcomb Office: 437 White Hall [email protected] http://community.wvu.edu/~miholcomb/ Today: Introduction to me, you, class structure and topic About Your Professor • • • • • Went to school at Vanderbilt and Berkeley Did graduate work at a national lab Did internship at IBM on quantum computing Toyed with a non-scientific internet startup Decided I liked teaching, so I’m here now My Work: Surface & Interface Physics Magnetic Dead Layers Exchange Bias Delafossites Topological Insulators Definition of a Multiferroic Ferromagnetic Ferroelectric field Before field After field Spontaneous magnetization whose Spontaneous polarization whose direction can be changed with an direction can be changed with an applied electric field (voltage) applied magnetic field Multiferroic Electric Control of Magnetism Coupling at interfaces is not well understood Review paper, Holcomb et al., IJMPB (2012) INDIRECT COUPLING http://www.helmholtz-berlin.de/forschung/magma/mdynamik/forschungsbereiche/multiferroicity_en.html Measurement Techniques WVU: Nonlinear Optics San Francisco Focused on Second Harmonic Generation and MOKE Synchrotron Techniques At National Labs: X-ray Absorption Spectroscopy Provides information on Photoemission Electron symmetry and interfacial time Microscopy (PEEM) dynamics Beyond the standard techniques (XRD, VSM, TEM, etc.), we focus on unique interfacial measurement techniques. As we are about to spend a lot of time together, please introduce yourself. -Name and Year - Taken or in quantum mechanics yet? -Planned career path (or possibilities, if debating) -Why are you taking this course? Important Class Issues • Text: Understanding Solid State Physics by by Holgate (in stock on Amazon, $22 - 85) • Strongly recommended: Intro. to Solid State Physics by Kittel ($1-178) Others • Lecture PPTs will be available online shortly after class. (additional material, not tested) • I expect you to do the Holgate reading (and ideally skim Kittel) before coming to class so that we can focus on details/applications. There is another reason. Why cramming does not work for the long run How Do We Learn? Through Repetition: the more times (and more ways) we repeat something, the more important our brains think it is, and the more likely we are to remember it Another good reason to read multiple books How Do We Learn? Amygdala: Hippocampus: Prefrontal Cortex: Fight or Flight Memory Executive Function My Goal Why Cramming Doesn’t Work Learning Assessment • There are many different physics skills (problem solving, written and oral communication, etc). My assignments reflect this variety. • Homework: Late assignments arriving in my hands will be counted 20% for each day late. • Feel free to work together on HW, but do not copy. Copying of anyone else’s work is an honor code violation. Oral Communication of Science Is one of the more difficult skills to master and grade. Your skill will be evaluated in 2 ways: •In class discussion (with both a partner and to the entire class) on in-class problems Discussion Questions • Some of these questions will be easy to make sure you have important background. If everyone thinks it’s easy, we’ll move through these fast. Otherwise, vital to cover. • Some of the questions will be harder. My goal is to coach you through these problems, not just do them for you. You learn more this way. Oral Communication of Science Is one of the more difficult skills to master and grade. Your skill will be evaluated in 2 ways: •In class discussion (with both a partner and to the entire class) on in-class problems •You will teach through a presentation. Pick a topic and get it approved. Course Grading Course Grade: Homework 25%, Midterm 30%, Final 30%, In-class Discussion 4%, Show & Tell 1%, Presentation 10% Grading scale: A (>85), B (70 - 84), C (60% - 69%), D (50% - 59%), F (<49%) Teaching: A student receiving an A on their presentation will at least 1) make and share organized powerpoint slides, 2) make eye contact with classmates, 3) discuss applications of physics, 4) motivate why topic is interesting, 5) identify key points and 6) stay close to the allotted time. (See syllabus for more) My Teaching Philosophy Regarding Slides •If you become a professor, you will be instructed to (regarding teaching) “beg, borrow and steal.” Preparing a good lecture is hard! So, if someone manages to, then reuse it! • Therefore, many of my slides are adaptations. I pick what I deem to be the most instructive slides. • I find this to work out pretty well, accept that there are many different notations for the same things are used. (e.g. lattice vectors) • In reality, papers use different notation, so it is a useful practice to get used to multiple forms of terminology. Please ask if you are confused about any terminology. Physics 471: Solid State Physics (SSP) Office: 437 White Hall Office hour: Tuesdays 2:30-3:30PM or by appointment [email protected] http://community.wvu.edu/~miholcomb/ Today’s Plan: Take ungraded pre-test. When finished, read over syllabus. If time remains, I will introduce SSP. What is solid state physics? • Solid state physics (SSP) explains the properties of solid materials which follow from Schrödinger’s equation for a collection of atomic nuclei and electrons interacting with electrostatic forces. • SSP, also known as condensed matter physics, is the study of the behavior of atoms when they are placed in close proximity to one another. Many of the concepts relevant to liquids too. What is the point? • Understanding the electrical properties of solids is right at the heart of modern society and technology. • The entire computer and electronics industry relies on tuning of a special class of material, the semiconductor, which lies right at the metalinsulator boundary. Solid state physics provide a background to understand what goes on in semiconductors. schedule Why is Solid State Physics Important? Electronic Properties - Transistors (the heart of electronics) Electron/Photon Interactions - Laser Diodes, Photodiodes, CCDS Electron/Phonon Interactions - Piezoelectric materials Electron Spin/Charge Interactions - Spintronics, Quantum Computing Highly Correlated Electronics Systems - Superconductors, Magnets New technology for the future will involve developing and understanding new classes of materials. By the end of this course we will see why this is a non-trivial task. What do pencil lead and diamond have in common? What property are they complete opposites? Electrical resistivity of three states of solid matter They are all just carbon! How can this be? After all, they each contain a system of atoms and especially electrons of similar density. Graphite is a metal, diamond is an insulator and buckminster-fullerene is a superconductor. With the remaining time today: Let’s remind ourselves of how we deal with individual atoms In the remainder of the semester we’ll focus on the effects of bringing lots of atoms together What is the simplest atom? What can you tell me about it? Potential energy felt by an electron? ke U (r ) r 2 Reminder: k=1/4o Multielectron Atoms (i.e., atoms other than hydrogen) • Because electrostatic forces between electrons are strong, we need to take them into account • We approximate this by treating the force on each electron independently, which includes force from nucleus + force from all other electrons • In this case, inner electrons can shield the nuclear charge, called “screening” Screening electron cloud We write the effective potential energy felt by an electron as +Ze r electron ke 2 U (r ) Z eff (r ) r Zeff is the effective charge that the electron feels and depends on r. Note that Z eff Z when r is inside all other electrons Z eff 1 when r is outside all other electrons Reminder: k=1/4o Energy Levels • As in the hydrogen atom, quantum states of electrons in multielectron atoms are specified by the quantum numbers n, l, m, mS • In hydrogen atom, all states of a given n are degenerate (in zero magnetic field and neglecting the fine structure) • In multielectron atoms the dependence of the potential energy on r due to screening lifts the degeneracy between these states: ke 2 U (r ) Z eff (r ) r Why does the effective radius look like this? In hydrogen, all n orbital (ns,np,nd) states have the same energy I won’t always have time to get to all of the slides. You won’t be tested on them in this case, but it’s not a bad idea to look through them if you have time. Electron distribution • For a multielectron atom, how are the electrons distributed among the different energy levels and orbitals? • Electrons would all crowd the ground state (lowest energy) if it wasn’t for the: Pauli Exclusion Principle: No two electrons (fermions) in a quantum system can occupy the same state (i.e., have the same quantum numbers) Would ground state helium or lithium be easier to ionize (remove an electron)? Helium ground state Helium excited state Lithium ground state How does the number of electrons determine the properties of the elements? The Periodic Table Properties of Helium 2He: Use notation ZE: e.g. 1H, 2He • Ground state: two electrons in 1s state (spin up/down) • Screening of each electron by the other • This results on a relatively large ionization energy (energy to remove an electron from a neutral atom) of 24.6 eV • Also large excitation energy (E2s-E1s) = 19.8 eV vs. 10.2 eV for H • Chemically inactive as a result of the large excitation and ionization energies – will not solidify unless low temperatures (4.2 K) and high pressures are used • Chemically inert gases are called noble or inert Helium ground state Helium excited state In groups, consider (for ~3 minutes): Compare ionization energies and effective radius of the elements Z=3 Lithium & Z=4 Beryllium Draw the ground state and excited states Properties of Lithium 3Li: • Ground state: two electrons in 1s state + one electron in 2s state • Ionization energy significantly smaller than He: expect Zeff ~1 with n=2, resulting in 5.4 eV • Large effective radius due to occupancy of n=2 level • Reactive as a result of the small ionization energy – can form compounds such as LiF Properties of First Ten Elements 4Be: • Ground state: two electrons in 1s state + two electrons in 2s state • Larger Z means larger ionization energy than Li (9.3 eV vs. 5.4 eV) • Excitation energy to 2p state is relatively low (2.7 eV) which makes Be chemically active and allows it to bond to other atoms (forms a solid) • Smaller effective radius due to larger Z than Li Lowest excited state for Be: Properties of First Ten Elements Other elements • Increasing Z causes electrons to be more tightly bound (causing greater ionization energies), however, when the quantum number changes higher energy states are occupied, meaning they are less tightly bound (causing lower ionization energies) • For Z=5 (B) electron goes to 2p state, slightly higher in energy (due to screening) causing binding energy to drop slightly (8.3 eV vs. 9.3 eV for Be). Very pure isolated boron is produced with difficulty, as boron tends to form refractory materials containing small amounts of carbon or other elements. • For Z=6 (Be) to Z=10 (Ne) electrons go into 2p state causing steady increase in ionization energy and decrease in radius due to increase in Z (all electrons go into n=2 level) • In going from Z=10 (Ne) (full n=2 shell) to Z=11 (Na, one electron in 3s) level ionization energy suddenly drops due to large increase in occupation energy of n=3 state (Zeff ~1, similar to Li) In Groups: Rank the Ionization Energies For each of the following sets of atoms, decide which has the highest and lowest ionization energies and why. a. Mg, Si, S b. Mg, Ca, Ba c. F, Cl, Br d. Ba, Cu, Ne e. Si, P, N