File - Mrs. Dawson`s Classroom
... MASS NUMBER The mass number is the total number of protons and neutrons that make up the nucleus of an isotope. Examples: ...
... MASS NUMBER The mass number is the total number of protons and neutrons that make up the nucleus of an isotope. Examples: ...
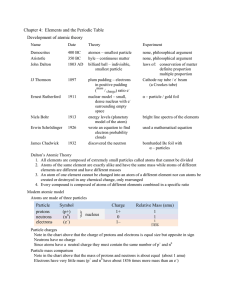
Chapter 4: Elements and the Periodic Table Development of atomic
... Actinides – the bottom row of which only Ac, Th, Pa, and U occur naturally on earth Most of the actinides are synthetic elements formed in particle accelerators Mixed group metals – metals found in the bottom left corner of the p block The most familiar of these metals are Al, Sn, and Pb Pb was used ...
... Actinides – the bottom row of which only Ac, Th, Pa, and U occur naturally on earth Most of the actinides are synthetic elements formed in particle accelerators Mixed group metals – metals found in the bottom left corner of the p block The most familiar of these metals are Al, Sn, and Pb Pb was used ...
1020 Chapter 4 Lecture Notes
... 2) Atomic Mass Deficit (more subtle and harder to understand) Protons and neutrons are held together to form nuclei by the strong nuclear force. Energy must be expended to separate a nucleus into individual nucleons. At the subatomic level, energy and mass are equivalent. When a system gains energy, ...
... 2) Atomic Mass Deficit (more subtle and harder to understand) Protons and neutrons are held together to form nuclei by the strong nuclear force. Energy must be expended to separate a nucleus into individual nucleons. At the subatomic level, energy and mass are equivalent. When a system gains energy, ...
Ch 17 Properties of Atoms - Effingham County Schools
... The organization of elements by their properties is the periodic table. Each small square on the periodic table shows the name of one element and the letter symbol for that element. The elements are arranged based on the number of protons an atom of that element has in its nucleus. Periodic la ...
... The organization of elements by their properties is the periodic table. Each small square on the periodic table shows the name of one element and the letter symbol for that element. The elements are arranged based on the number of protons an atom of that element has in its nucleus. Periodic la ...
atom
... J. J. Thomson had discovered the electron, a negatively charged, low mass particle present within all atoms. Properties of Electrical Charge: Electrical charge is a fundamental property of some of the particles that compose atoms and results in attractive and repulsive forces—called electrostatic fo ...
... J. J. Thomson had discovered the electron, a negatively charged, low mass particle present within all atoms. Properties of Electrical Charge: Electrical charge is a fundamental property of some of the particles that compose atoms and results in attractive and repulsive forces—called electrostatic fo ...
LAB ACTIVITY CALCULATING ATOMIC MASS
... Press Release from the Virginia Beach News: Virginia Beach, VA- Chemists, working on a project for the city of Virginia Beach, have recently discovered what is believed to be element 114. The researchers have proposed that the element be named vabeachium (va-beach-e-m), after the city in which it wa ...
... Press Release from the Virginia Beach News: Virginia Beach, VA- Chemists, working on a project for the city of Virginia Beach, have recently discovered what is believed to be element 114. The researchers have proposed that the element be named vabeachium (va-beach-e-m), after the city in which it wa ...
Periodic Table Trends
... • You only need to know the group number to know the valence electrons • But be careful of He in group VIII • The pattern does not quite hold for the transition elements ...
... • You only need to know the group number to know the valence electrons • But be careful of He in group VIII • The pattern does not quite hold for the transition elements ...
Element - Faculty
... Chemical formulas can be determined by measuring the mass of each element present in a sample of the compound. The mass of each element (grams) is converted to number of moles, or molecules of each element presenting the compound. You will need to do such calculations in order to determine the amoun ...
... Chemical formulas can be determined by measuring the mass of each element present in a sample of the compound. The mass of each element (grams) is converted to number of moles, or molecules of each element presenting the compound. You will need to do such calculations in order to determine the amoun ...
Module 4 Trivia Review
... Semi means half or partial. So semiconductors (metalloids) have electrical conductivity half way between those of a conductor and an insulator (non-metal). Since they are solid and ductile, these metalloids have been found to be indispensable to the technology industry. Metals would conduct too much ...
... Semi means half or partial. So semiconductors (metalloids) have electrical conductivity half way between those of a conductor and an insulator (non-metal). Since they are solid and ductile, these metalloids have been found to be indispensable to the technology industry. Metals would conduct too much ...
Study Guide Answer Key
... 2. Consider an element Z that has two naturally occurring isotopes with the following percent abundances: the isotope with a mass number of 19.0 is 55.0% abundant; the isotope with a mass number of 21.0 is 45.0% abundant. What is the average atomic mass for element Z? [(mass A) (%A)] + [(mass B) (%B ...
... 2. Consider an element Z that has two naturally occurring isotopes with the following percent abundances: the isotope with a mass number of 19.0 is 55.0% abundant; the isotope with a mass number of 21.0 is 45.0% abundant. What is the average atomic mass for element Z? [(mass A) (%A)] + [(mass B) (%B ...
Summer - Honors Chemistry
... are made from only one kind of atom (which all share the same atomic number and elemental symbol), and all atoms of a given element have identical chemical properties, but not the same mass (isotopes). Elements can come in atomic form (e.g. Fe) or in molecular form (O2 or C60). About 80% of the elem ...
... are made from only one kind of atom (which all share the same atomic number and elemental symbol), and all atoms of a given element have identical chemical properties, but not the same mass (isotopes). Elements can come in atomic form (e.g. Fe) or in molecular form (O2 or C60). About 80% of the elem ...
DEFINING THE ATOM - Southgate Schools
... Part B True-False Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. ________ 11. The atomic number of an element is the sum of the protons and electrons in an atom of that element. ________ 12. The atomic number of an atom is the total number of protons in ...
... Part B True-False Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. ________ 11. The atomic number of an element is the sum of the protons and electrons in an atom of that element. ________ 12. The atomic number of an atom is the total number of protons in ...
Atoms, Elements, Compounds, and Mixtures
... be divided and still maintain its years, scientists have characteristics. designed many different models for this structure. Atoms are the building blocks of the universe. There are 92 Each one was the best different kinds of atoms that model at the time, but as occur naturally, although more ne ...
... be divided and still maintain its years, scientists have characteristics. designed many different models for this structure. Atoms are the building blocks of the universe. There are 92 Each one was the best different kinds of atoms that model at the time, but as occur naturally, although more ne ...
periodic table - Mesa Community College
... involving hydrogen are usually named as if hydrogen were a metal, and the compound a binary ionic one. This does not, however, make hydrogen a metal. 2. Metal has more than one possible oxidation number. These compounds are named by giving the name of the metal followed by its oxidation number, expr ...
... involving hydrogen are usually named as if hydrogen were a metal, and the compound a binary ionic one. This does not, however, make hydrogen a metal. 2. Metal has more than one possible oxidation number. These compounds are named by giving the name of the metal followed by its oxidation number, expr ...
Unit C3, C3.1
... However, other chemists did not accept Newlands’ ideas. It was not until much later that his contribution to the development of the modern periodic table was recognised. Reproduced courtesy of the library and information centre Royal Society of Chemistry ...
... However, other chemists did not accept Newlands’ ideas. It was not until much later that his contribution to the development of the modern periodic table was recognised. Reproduced courtesy of the library and information centre Royal Society of Chemistry ...
Elements, Compounds, and Mixtures
... You will be shown a series of photos. Tell if each photo represents an item composed of an element, compound, or mixture. Review: An element contains just one type of atom. A compound contains two or more different atoms joined together. A mixture contains two or more different substances that ...
... You will be shown a series of photos. Tell if each photo represents an item composed of an element, compound, or mixture. Review: An element contains just one type of atom. A compound contains two or more different atoms joined together. A mixture contains two or more different substances that ...
b. Elements as Mixtures - Isotopes
... Elements are made from atoms having the same atomic number, protons Are all atoms of one particular atom the same or are they mixtures? 1) All atom nuclei for an element have the same number of protons. 2) Every atom in an element has the same number of protons & electrons 3) However, elements are ...
... Elements are made from atoms having the same atomic number, protons Are all atoms of one particular atom the same or are they mixtures? 1) All atom nuclei for an element have the same number of protons. 2) Every atom in an element has the same number of protons & electrons 3) However, elements are ...
isotopes and atomic mass
... 1. Which of the data in the table must be measured and which must be calculated? 2. In all except step 11, the “Total” is calculated by adding the numbers across each row. Step 11 is an exception because it does not take into account the fact that there are different numbers of each isotope. Rather ...
... 1. Which of the data in the table must be measured and which must be calculated? 2. In all except step 11, the “Total” is calculated by adding the numbers across each row. Step 11 is an exception because it does not take into account the fact that there are different numbers of each isotope. Rather ...
Atomic Structure
... How would the results of Rutherford's experiments have been different if atoms had the positive charges on the outside and negative charges in the middle? ...
... How would the results of Rutherford's experiments have been different if atoms had the positive charges on the outside and negative charges in the middle? ...
Yr11 Chemistry Title Page:TourismContents
... Atoms of different elements can combine with one another in simple (whole number) ratios to form compounds. ...
... Atoms of different elements can combine with one another in simple (whole number) ratios to form compounds. ...
Chapter 2: Atoms Molecules and Ions
... reactant molecules into a mass ratio for a chemical reaction to be useful. 2) Mass ratios are determined by using atomic masses for the elements. i) Atomic masses (atomic weights) are found in periodic table beneath the chemical symbol, and represent the average of all the naturally occurring isotop ...
... reactant molecules into a mass ratio for a chemical reaction to be useful. 2) Mass ratios are determined by using atomic masses for the elements. i) Atomic masses (atomic weights) are found in periodic table beneath the chemical symbol, and represent the average of all the naturally occurring isotop ...
Sub Unit Plan 1 Chem Periodic Table
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...