Geophysical Journal International
... the rock’s frame shear modulus (μsat ) should remain constant regardless of the saturant (Berryman & Milton 1991). However, there are a number of experimental observation in which both sandstones (Wyllie et al. 1958; Khazanehdari & Sothcott 2003) and carbonates (Peselnik 1962; Assefa et al. 2003; Rø ...
... the rock’s frame shear modulus (μsat ) should remain constant regardless of the saturant (Berryman & Milton 1991). However, there are a number of experimental observation in which both sandstones (Wyllie et al. 1958; Khazanehdari & Sothcott 2003) and carbonates (Peselnik 1962; Assefa et al. 2003; Rø ...
Auto-ignition Characteristics of Selected Ionic Liquids
... nonflammability. Thus, ionic liquids are deemed to be green solvent replacements for the common used volatile organic solvents in part because of aforementioned characteristics [1, 2]. However, recent researches have indicated that ionic liquids are flammable. Fox et al. have pointed significant dec ...
... nonflammability. Thus, ionic liquids are deemed to be green solvent replacements for the common used volatile organic solvents in part because of aforementioned characteristics [1, 2]. However, recent researches have indicated that ionic liquids are flammable. Fox et al. have pointed significant dec ...
Thermochemistry
... When the units for the expressions for work and energy are collected together, in both cases, the resultant unit is kg m2 s -2. This corresponds to the SI unit of energy called the joule (J). That is, 1 joule (J) = 1 kg m2 s -2. The bouncing ball in Figure 7-2 suggests something about the nature of ...
... When the units for the expressions for work and energy are collected together, in both cases, the resultant unit is kg m2 s -2. This corresponds to the SI unit of energy called the joule (J). That is, 1 joule (J) = 1 kg m2 s -2. The bouncing ball in Figure 7-2 suggests something about the nature of ...
Symmetric and asymmetric modes of the thermohaline circulation
... of the overturning and the equator-to-pole density difference in the dominant hemisphere essentially follows the classical one-hemisphere scaling. In this case, the shape of the surface density field is kept invariant. However, they pointed out that the classical scaling is inadequate for describing ...
... of the overturning and the equator-to-pole density difference in the dominant hemisphere essentially follows the classical one-hemisphere scaling. In this case, the shape of the surface density field is kept invariant. However, they pointed out that the classical scaling is inadequate for describing ...
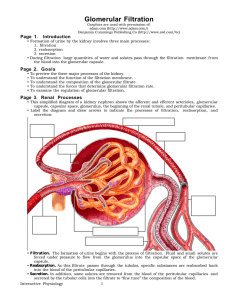
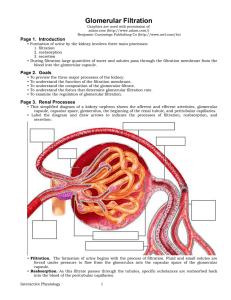
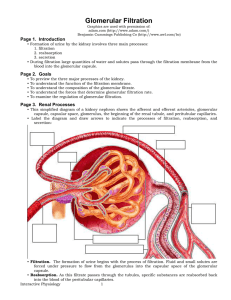
Glomerular Filtration - Interactive Physiology
... • The blood pressure in the glomerulus averages 60 millimeters of mercury. This unusually high capillary pressure is the result of the short, large diameter afferent arterioles conveying blood at high arterial pressure directly to the glomerular capillaries. • The smaller diameter of the efferent ar ...
... • The blood pressure in the glomerulus averages 60 millimeters of mercury. This unusually high capillary pressure is the result of the short, large diameter afferent arterioles conveying blood at high arterial pressure directly to the glomerular capillaries. • The smaller diameter of the efferent ar ...
Heat and Thermodynamics
... In a thermodynamic system with fixed volume and pressure, enthalpy has the same meaning as internal energy and it is also measured in kilojoules (kJ). The reference condition regarding enthalpy differs from one substance to another. For water (H2O), the reference condition is defined at a temperatur ...
... In a thermodynamic system with fixed volume and pressure, enthalpy has the same meaning as internal energy and it is also measured in kilojoules (kJ). The reference condition regarding enthalpy differs from one substance to another. For water (H2O), the reference condition is defined at a temperatur ...
A Pool Boiling Map: Water on a Horizontal Surface at
... being slightly greater than the vapor temperature predicted by equation (!II-5.) The Situation Addressed For the situation where the nucleation site is filled with a gas, an expression for the superheat needed to form a vapor bubble was developed by Bergles (1981) ...
... being slightly greater than the vapor temperature predicted by equation (!II-5.) The Situation Addressed For the situation where the nucleation site is filled with a gas, an expression for the superheat needed to form a vapor bubble was developed by Bergles (1981) ...
book - University of Guelph Physics
... and theoretically, because their physical properties do not change with time. The framework of thermodynamics applies equally well to all such macroscopic systems; it is a powerful and very general framework. An example of a thermodynamic system is a fluid (a gas or a liquid) confined to a beaker of ...
... and theoretically, because their physical properties do not change with time. The framework of thermodynamics applies equally well to all such macroscopic systems; it is a powerful and very general framework. An example of a thermodynamic system is a fluid (a gas or a liquid) confined to a beaker of ...
Knowledge Check (Answer Key)
... generator – which is “almost” 100% saturated. The addition of heat to a saturated vapor results in an increase in temperature. A vapor that exists at a temperature that is greater than its saturation temperature is a superheated vapor. In a superheated vapor (like a subcooled liquid), the pressure a ...
... generator – which is “almost” 100% saturated. The addition of heat to a saturated vapor results in an increase in temperature. A vapor that exists at a temperature that is greater than its saturation temperature is a superheated vapor. In a superheated vapor (like a subcooled liquid), the pressure a ...
Countercurrent exchange

Countercurrent exchange is a mechanism occurring in nature and mimicked in industry and engineering, in which there is a crossover of some property, usually heat or some component, between two flowing bodies flowing in opposite directions to each other. The flowing bodies can be liquids, gases, or even solid powders, or any combination of those. For example, in a distillation column, the vapors bubble up through the downward flowing liquid while exchanging both heat and mass.The maximum amount of heat or mass transfer that can be obtained is higher with countercurrent than co-current (parallel) exchange because countercurrent maintains a slowly declining difference or gradient (usually temperature or concentration difference). In cocurrent exchange the initial gradient is higher but falls off quickly, leading to wasted potential. For example, in the diagram at the right, the fluid being heated (exiting top) has a higher exiting temperature than the cooled fluid (exiting bottom) that was used for heating. With cocurrent or parallel exchange the heated and cooled fluids can only approach one another. The result is that countercurrent exchange can achieve a greater amount of heat or mass transfer than parallel under otherwise similar conditions. See: flow arrangement.Countercurrent exchange when set up in a circuit or loop can be used for building up concentrations, heat, or other properties of flowing liquids. Specifically when set up in a loop with a buffering liquid between the incoming and outgoing fluid running in a circuit, and with active transport pumps on the outgoing fluid's tubes, the system is called a Countercurrent multiplier, enabling a multiplied effect of many small pumps to gradually build up a large concentration in the buffer liquid.Other countercurrent exchange circuits where the incoming and outgoing fluids touch each other are used for retaining a high concentration of a dissolved substance or for retaining heat, or for allowing the external buildup of the heat or concentration at one point in the system.Countercurrent exchange circuits or loops are found extensively in nature, specifically in biologic systems. In vertebrates, they are called a Rete mirabile, originally the name of an organ in fish gills for absorbing oxygen from the water. It is mimicked in industrial systems. Countercurrent exchange is a key concept in chemical engineering thermodynamics and manufacturing processes, for example in extracting sucrose from sugar beet roots.Countercurrent multiplication is a similar but different concept where liquid moves in a loop followed by a long length of movement in opposite directions with an intermediate zone. The tube leading to the loop passively building up a gradient of heat (or cooling) or solvent concentration while the returning tube has a constant small pumping action all along it, so that a gradual intensification of the heat or concentration is created towards the loop. Countercurrent multiplication has been found in the kidneys as well as in many other biological organs.