INTRODUCTION TO QUANTUM OPTICS
... Finally, we should not forget how strongly seeing things influences our concepts of and the ways in which we pursue science. We can only speculate whether the current state of science could have been achieved without sight, without our ability to comprehend complicated equations, or to recognize str ...
... Finally, we should not forget how strongly seeing things influences our concepts of and the ways in which we pursue science. We can only speculate whether the current state of science could have been achieved without sight, without our ability to comprehend complicated equations, or to recognize str ...
2.8-2.9 - BYU Physics and Astronomy
... The photon was originally called a “light quantum” (das Lichtquant) by Albert Einstein.[5] The modern name “photon” derives from the Greek word for light, φῶς, (transliterated phôs), and was coined in 1926 by the physical chemist Gilbert N. Lewis, who published a speculative theory[9] in which photo ...
... The photon was originally called a “light quantum” (das Lichtquant) by Albert Einstein.[5] The modern name “photon” derives from the Greek word for light, φῶς, (transliterated phôs), and was coined in 1926 by the physical chemist Gilbert N. Lewis, who published a speculative theory[9] in which photo ...
PHY982 12th week Electromagnetic field and coupling with photons
... Coupling photons and particles Connecting the classical EM description with quantum particle dynamics o free field source flux o particle current to photon production (capture) ...
... Coupling photons and particles Connecting the classical EM description with quantum particle dynamics o free field source flux o particle current to photon production (capture) ...
Lecture 13: Heisenberg and Uncertainty
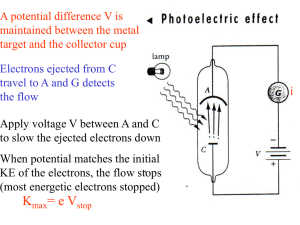
... Light When light strikes E, photoelectrons are emitted Electrons collected at C and passing through the ammeter are a current in the circuit ...
... Light When light strikes E, photoelectrons are emitted Electrons collected at C and passing through the ammeter are a current in the circuit ...
View the Powerpoint Presentation.
... Maximum kinetic energy of photoelectrons based on photon energy and work function Einstein’s equation for conversion of matter to energy ...
... Maximum kinetic energy of photoelectrons based on photon energy and work function Einstein’s equation for conversion of matter to energy ...
What is light?
... electromagnetic disturbance in the form of waves propagated through the electromagnetic field according to electromagnetic laws.” ...
... electromagnetic disturbance in the form of waves propagated through the electromagnetic field according to electromagnetic laws.” ...
example8
... threshold frequency. If the frequency is below this threshold, then no matter how bright the radiation is, the electrons are not given off. Above this frequency the kinetic energy of the electrons increases as the frequency increases. Also, the rate at which the electrons are given off depends on ho ...
... threshold frequency. If the frequency is below this threshold, then no matter how bright the radiation is, the electrons are not given off. Above this frequency the kinetic energy of the electrons increases as the frequency increases. Also, the rate at which the electrons are given off depends on ho ...
Lesson 1 - Tarleton State University
... A similar experiment was performed independently later that year by G.P. Thomson (J.J. Thomson's son). Davidson and Thomson won the Nobel Prize for their scattering experiments. ...
... A similar experiment was performed independently later that year by G.P. Thomson (J.J. Thomson's son). Davidson and Thomson won the Nobel Prize for their scattering experiments. ...
Set 3
... Historical introduction to quantum mechanics - Set no. 3 1) Suppose that a 60 W light bulb radiates primarily at a wavelength 1000 nm. What is the number of photons emitted per second ? 2) What is the number of photons emitted per second by the Sun? Lets assume that the star radiates electromagnetic ...
... Historical introduction to quantum mechanics - Set no. 3 1) Suppose that a 60 W light bulb radiates primarily at a wavelength 1000 nm. What is the number of photons emitted per second ? 2) What is the number of photons emitted per second by the Sun? Lets assume that the star radiates electromagnetic ...
Historic Development by Mihai
... Einstein came up with the hypothesis of the corpuscular nature of light explaining the photoelectric effect. Einstein theory was verified by Millikan experiments (1916) ,he received Nobel price for that. Careful investigations toward the end of the nineteenth century proved that the photoelectric ef ...
... Einstein came up with the hypothesis of the corpuscular nature of light explaining the photoelectric effect. Einstein theory was verified by Millikan experiments (1916) ,he received Nobel price for that. Careful investigations toward the end of the nineteenth century proved that the photoelectric ef ...
V stop f
... work function is 2.2 eV, what is the wavelength of the light? • Ephoton = hf = hc/ • Kmax = Ephoton - = hc/ - = e Vstop • = (hc)/(e Vstop + ) • h = 6.63x10-34 J.s = 6.63x10-34 /1.6x10-19 eV.s = 4.14 x10-15 eV.s • = (4.14 x10-15 eV.s)(3x108m/s)/[5.0 eV+2.2 eV] =170 nm ...
... work function is 2.2 eV, what is the wavelength of the light? • Ephoton = hf = hc/ • Kmax = Ephoton - = hc/ - = e Vstop • = (hc)/(e Vstop + ) • h = 6.63x10-34 J.s = 6.63x10-34 /1.6x10-19 eV.s = 4.14 x10-15 eV.s • = (4.14 x10-15 eV.s)(3x108m/s)/[5.0 eV+2.2 eV] =170 nm ...
Please look over the following review questions
... quite different from the radius predicted by Bohr that agrees with the orbital radius of Bohr ...
... quite different from the radius predicted by Bohr that agrees with the orbital radius of Bohr ...
U2: Day 4 Notes
... o Theory: light is composed of photons that have both particle and wave properties o Light as a WAVE: Has a wavelength, λ Has a frequency, ν Has a speed, c Electromagnetic spectrum is a display of the various types of electromagnetic radiation Visible radiation wavelengths 400 nm(violet)-7 ...
... o Theory: light is composed of photons that have both particle and wave properties o Light as a WAVE: Has a wavelength, λ Has a frequency, ν Has a speed, c Electromagnetic spectrum is a display of the various types of electromagnetic radiation Visible radiation wavelengths 400 nm(violet)-7 ...
Type in the abstract title here
... Entanglement is one of the key features to quantum info-communication technology. Although parametric down-conversion has been used so far to generate highly entangled pairs of photons, entangled photon sources using semiconductors are desired for practical applications [1-3]. We reported the first ...
... Entanglement is one of the key features to quantum info-communication technology. Although parametric down-conversion has been used so far to generate highly entangled pairs of photons, entangled photon sources using semiconductors are desired for practical applications [1-3]. We reported the first ...
Physics116_L31
... J. J. Thomson (1897) identifies electron: very light, negative charge E. Rutherford (1911) bounces “alpha rays” off gold atoms • We now know: α = nucleus of helium: 2 protons + 2 neutrons • “Scattering experiment” = model for modern particle physics – Size of atoms was approximately known from chemi ...
... J. J. Thomson (1897) identifies electron: very light, negative charge E. Rutherford (1911) bounces “alpha rays” off gold atoms • We now know: α = nucleus of helium: 2 protons + 2 neutrons • “Scattering experiment” = model for modern particle physics – Size of atoms was approximately known from chemi ...
The Compton Effect, or Compton scattering – conclusive evidence
... Where did the extra energy go? The energy lost by the x-ray photons, as evidenced by the photons’ increased wavelength, increases the kinetic energy of the scattered electrons. Sound like billiards? It should! The collision is inelastic – kinetic energy is not conserved, although total energy is (se ...
... Where did the extra energy go? The energy lost by the x-ray photons, as evidenced by the photons’ increased wavelength, increases the kinetic energy of the scattered electrons. Sound like billiards? It should! The collision is inelastic – kinetic energy is not conserved, although total energy is (se ...
Chapter 27: Summary
... to be emitted via the photoelectric effect. If we reduce the intensity of the light shining on the metal, without changing its wavelength, what happens? Explain your answer. 4. The work functions of gold, aluminum, and cesium are 5.1 eV, 4.1 eV, and 2.1 eV, respectively. If light of a particular fre ...
... to be emitted via the photoelectric effect. If we reduce the intensity of the light shining on the metal, without changing its wavelength, what happens? Explain your answer. 4. The work functions of gold, aluminum, and cesium are 5.1 eV, 4.1 eV, and 2.1 eV, respectively. If light of a particular fre ...
Chemistry Quiz Ch. 5 Electron Configuration and Quantum Theory
... 17. Circle an example where two elements have vastly different properties, yet are only 1 electron different in their structures a. Sodium and Fluorine b. Fluorine and Chlorine c. Sodium and Neon d. Neon and Argon 18. Quantum physics has established that Light travels as two different properties: a. ...
... 17. Circle an example where two elements have vastly different properties, yet are only 1 electron different in their structures a. Sodium and Fluorine b. Fluorine and Chlorine c. Sodium and Neon d. Neon and Argon 18. Quantum physics has established that Light travels as two different properties: a. ...
Atomic Diffraction Dr. Janine Shertzer College of the Holy Cross
... The wave-particle duality is fundamental to quantum mechanics. Light can behave like a particle (photon); matter can behave like a wave. The wavelength associated with a particle is inversely proportional to its momentum p: λ = h / p, where h is Planck’s constant. For cold atoms, the wavelength is l ...
... The wave-particle duality is fundamental to quantum mechanics. Light can behave like a particle (photon); matter can behave like a wave. The wavelength associated with a particle is inversely proportional to its momentum p: λ = h / p, where h is Planck’s constant. For cold atoms, the wavelength is l ...
THINGSYOUNEEDTOKNOW-modern
... CONTINUOUS SPECTRUM-A smooth spectrum of all colors of the rainbow. Generated by light bulb ABSORPTION SPECTRUM-looks like continuous spectrum with some spectral lines absorbed By a cool gas EMISSION SPECTRUM-a spectrum made up of only a few spectral lines emitted by an excited gas. Energy of photon ...
... CONTINUOUS SPECTRUM-A smooth spectrum of all colors of the rainbow. Generated by light bulb ABSORPTION SPECTRUM-looks like continuous spectrum with some spectral lines absorbed By a cool gas EMISSION SPECTRUM-a spectrum made up of only a few spectral lines emitted by an excited gas. Energy of photon ...
The Photoelectric Effect
... •When the light intensity is very low there should be a time delay before the photoelectron emission •Experimental observation: •Kinetic energy of the photoelectrons is independent of the light intensity but increases with the frequency •No photoelectrons are emitted below some cutoff frequency fc • ...
... •When the light intensity is very low there should be a time delay before the photoelectron emission •Experimental observation: •Kinetic energy of the photoelectrons is independent of the light intensity but increases with the frequency •No photoelectrons are emitted below some cutoff frequency fc • ...
Wang_Project_Summery
... energy of all electron bunches inside the SPEAR ring. The Light Shack takes the synchrotron radiation from the SPEAR ring and filters out the harmful wavelength radiation with a cold finger, which is a piece of metal that blocks out x-ray radiations. As a result from the cold finger, only radiation ...
... energy of all electron bunches inside the SPEAR ring. The Light Shack takes the synchrotron radiation from the SPEAR ring and filters out the harmful wavelength radiation with a cold finger, which is a piece of metal that blocks out x-ray radiations. As a result from the cold finger, only radiation ...
Chapter 3 Electromagnetic Theory, Photons, and Light
... Photons are bosons - many photons can simultaneously be in exactly the same state, i.e. have the same energy When a large number of photons occupy the same state (i.e. have the same energy, polarization and direction), the inherent granularity of the light beam vanishes and the EM field appears as t ...
... Photons are bosons - many photons can simultaneously be in exactly the same state, i.e. have the same energy When a large number of photons occupy the same state (i.e. have the same energy, polarization and direction), the inherent granularity of the light beam vanishes and the EM field appears as t ...
BACH, the Beamline for Advanced diCHroic and scattering
... polarized light comes from the fact that excitations induced in condensed and soft matter by the absorption of a photon are affected by the polarization of the light by selection rules depending on the angular degrees of freedom of the ground and final states. In this case, information concerning th ...
... polarized light comes from the fact that excitations induced in condensed and soft matter by the absorption of a photon are affected by the polarization of the light by selection rules depending on the angular degrees of freedom of the ground and final states. In this case, information concerning th ...
Photon
A photon is an elementary particle, the quantum of light and all other forms of electromagnetic radiation. It is the force carrier for the electromagnetic force, even when static via virtual photons. The effects of this force are easily observable at the microscopic and at the macroscopic level, because the photon has zero rest mass; this allows long distance interactions. Like all elementary particles, photons are currently best explained by quantum mechanics and exhibit wave–particle duality, exhibiting properties of waves and of particles. For example, a single photon may be refracted by a lens or exhibit wave interference with itself, but also act as a particle giving a definite result when its position is measured. Waves and quanta, being two observable aspects of a single phenomenon cannot have their true nature described in terms of any mechanical model. A representation of this dual property of light, which assumes certain points on the wave front to be the seat of the energy is also impossible. Thus, the quanta in a light wave cannot be spatially localized. Some defined physical parameters of a photon are listed. The modern photon concept was developed gradually by Albert Einstein in the first years of the 20th century to explain experimental observations that did not fit the classical wave model of light. In particular, the photon model accounted for the frequency dependence of light's energy, and explained the ability of matter and radiation to be in thermal equilibrium. It also accounted for anomalous observations, including the properties of black-body radiation, that other physicists, most notably Max Planck, had sought to explain using semiclassical models, in which light is still described by Maxwell's equations, but the material objects that emit and absorb light do so in amounts of energy that are quantized (i.e., they change energy only by certain particular discrete amounts and cannot change energy in any arbitrary way). Although these semiclassical models contributed to the development of quantum mechanics, many further experiments starting with Compton scattering of single photons by electrons, first observed in 1923, validated Einstein's hypothesis that light itself is quantized. In 1926 the optical physicist Frithiof Wolfers and the chemist Gilbert N. Lewis coined the name photon for these particles, and after 1927, when Arthur H. Compton won the Nobel Prize for his scattering studies, most scientists accepted the validity that quanta of light have an independent existence, and the term photon for light quanta was accepted.In the Standard Model of particle physics, photons and other elementary particles are described as a necessary consequence of physical laws having a certain symmetry at every point in spacetime. The intrinsic properties of particles, such as charge, mass and spin, are determined by the properties of this gauge symmetry.The photon concept has led to momentous advances in experimental and theoretical physics, such as lasers, Bose–Einstein condensation, quantum field theory, and the probabilistic interpretation of quantum mechanics. It has been applied to photochemistry, high-resolution microscopy, and measurements of molecular distances. Recently, photons have been studied as elements of quantum computers and for applications in optical imaging and optical communication such as quantum cryptography.