The Photoelectric Effect in Practice WS Key
... 7. Automatic flushing toilets use the photoelectric effect. When there’s a change from darkness to light, they flush automatically! Most AFTs use reflected light of a 950 nm wavelength to trigger the flush. The site of one manufacturer says they use titanium as the sensing element. Find the work fun ...
... 7. Automatic flushing toilets use the photoelectric effect. When there’s a change from darkness to light, they flush automatically! Most AFTs use reflected light of a 950 nm wavelength to trigger the flush. The site of one manufacturer says they use titanium as the sensing element. Find the work fun ...
Chapter 7 Homework questions
... (a) What colors of light involve less energy than green light? (b) Which color of light has photons of greater energy, yellow or blue? 25. Violet light has a wavelength of about 410 nm. What is its frequency? Calculate the energy of one photon of violet light. What is the energy of 1.0 mol of violet ...
... (a) What colors of light involve less energy than green light? (b) Which color of light has photons of greater energy, yellow or blue? 25. Violet light has a wavelength of about 410 nm. What is its frequency? Calculate the energy of one photon of violet light. What is the energy of 1.0 mol of violet ...
powerpoint Light 00
... Light as a wave! diffraction--will spread out after going through an opening if the opening is small enough ...
... Light as a wave! diffraction--will spread out after going through an opening if the opening is small enough ...
Historical Introduction to the Elementary Particles
... charges. We can now give this model a more sophisticated formulation by attributing the binding force to the exchange of photons between the electrons and the protons in the nucleus. However, for the purposes of atomic physics this is overkill, for in this context quantization of the electromagnetic ...
... charges. We can now give this model a more sophisticated formulation by attributing the binding force to the exchange of photons between the electrons and the protons in the nucleus. However, for the purposes of atomic physics this is overkill, for in this context quantization of the electromagnetic ...
Electronic Structure and the Periodic Table
... Max Planck discovered that the atoms of a solid vibrate with energy of a definite frequency, f, depending on the solid. ...
... Max Planck discovered that the atoms of a solid vibrate with energy of a definite frequency, f, depending on the solid. ...
Fourth lecture, 28.10.03 (dispersion cancellation, time measurement
... Why? No interference between paths leading to different frequencies at the detectors, because in principle one could go back and measure how much energy had been absorbed. Note: it took a long time-integral to enforce this. If the detector had been open only for 1 fs, it would be impossible to tell ...
... Why? No interference between paths leading to different frequencies at the detectors, because in principle one could go back and measure how much energy had been absorbed. Note: it took a long time-integral to enforce this. If the detector had been open only for 1 fs, it would be impossible to tell ...
Biophysics
... •200 MeV: energy released during nuclear decay of 235U atom •13.6 eV: ionizing energy of H atom •2.5 eV: energy of bluish light photon •1/40 eV: kT energy at room temperature ...
... •200 MeV: energy released during nuclear decay of 235U atom •13.6 eV: ionizing energy of H atom •2.5 eV: energy of bluish light photon •1/40 eV: kT energy at room temperature ...
Concepts introduced by the theories of relativity include
... time dilates. • Spacetime: space and time should be considered together and in relation to each other. • The speed of light is nonetheless invariant, the same for all observers. ...
... time dilates. • Spacetime: space and time should be considered together and in relation to each other. • The speed of light is nonetheless invariant, the same for all observers. ...
Electromagnetic Waves In this lecture Waves Speed of
... • X-ray photons are characterised by energy • Planck developed relationship between energy and frequency ...
... • X-ray photons are characterised by energy • Planck developed relationship between energy and frequency ...
Quantum_PPT
... older wave theory: • 1. Wave theory could not explain why a hot body emitted light. • 2. UV-light discharged electricallycharged metal plates (the photoelectric effect). ...
... older wave theory: • 1. Wave theory could not explain why a hot body emitted light. • 2. UV-light discharged electricallycharged metal plates (the photoelectric effect). ...
Chapter 27
... what is the minimum uncertainty in its position? 51. Photons of wavelength 450 nm are incident on a metal. The most energetic electrons ejected from the metal are bent into a circular arc of radius 20.0 cm by a magnetic field with a magnitude of 2.00 × 10–5 T. What is the work function of the meta ...
... what is the minimum uncertainty in its position? 51. Photons of wavelength 450 nm are incident on a metal. The most energetic electrons ejected from the metal are bent into a circular arc of radius 20.0 cm by a magnetic field with a magnitude of 2.00 × 10–5 T. What is the work function of the meta ...
Notes on Quantum Theory
... since Planck's constant is so small that both momentum and location can have very small uncertainties (and thus both be known quite accurately). Imagine trying to simultaneously determine the location and momentum of a very tiny moving particle, however. To detect the particle's position, a photon o ...
... since Planck's constant is so small that both momentum and location can have very small uncertainties (and thus both be known quite accurately). Imagine trying to simultaneously determine the location and momentum of a very tiny moving particle, however. To detect the particle's position, a photon o ...
P316
... function of potassium and compute the value of Planck’s constant. If the angular momentum of the Earth’s orbit around the Sun were quantized what would the Earth’s quantum number be? How much energy would be released in a transition to the next lower quantum number? Would this amount of energy be de ...
... function of potassium and compute the value of Planck’s constant. If the angular momentum of the Earth’s orbit around the Sun were quantized what would the Earth’s quantum number be? How much energy would be released in a transition to the next lower quantum number? Would this amount of energy be de ...
Electrons in Atoms Section 5.1
... • Compare the wave and particle models of light. • Define a quantum of energy and explain how it is related to an energy change of matter. • Contrast continuous electromagnetic spectra and atomic emission spectra. ...
... • Compare the wave and particle models of light. • Define a quantum of energy and explain how it is related to an energy change of matter. • Contrast continuous electromagnetic spectra and atomic emission spectra. ...
Photoelectric Effect When light shines on a metal surface, electrons are emitted
... Does light as photons (“particles” or “packages”)? • An increase in intensity of light means more photons are incident, so more electrons will be ejected; but since the energy of each photon is the same, the max KE of electrons is not changed. • If the frequency is increased, the max KE of the elec ...
... Does light as photons (“particles” or “packages”)? • An increase in intensity of light means more photons are incident, so more electrons will be ejected; but since the energy of each photon is the same, the max KE of electrons is not changed. • If the frequency is increased, the max KE of the elec ...
1 – Foundations of Quantum Theory
... • This formula states that the energy released by an object at a certain frequency is a multiple of the energy of each packet of light (the frequency by Planck’s constant) ...
... • This formula states that the energy released by an object at a certain frequency is a multiple of the energy of each packet of light (the frequency by Planck’s constant) ...
$doc.title
... P. Debye, M. Knudsen, W.L. Bragg, H.A. Kramers, P.A.M. Dirac, A.H. Compton, L. de Broglie, M. Born, N. Bohr; I. Langmuir, M. Planck, M. Skłodowska-Curie, H.A. Lorentz, A. Einstein, P. Langevin, Ch.-E. Guye, C.T.R. Wilson, O.W. Richardson ...
... P. Debye, M. Knudsen, W.L. Bragg, H.A. Kramers, P.A.M. Dirac, A.H. Compton, L. de Broglie, M. Born, N. Bohr; I. Langmuir, M. Planck, M. Skłodowska-Curie, H.A. Lorentz, A. Einstein, P. Langevin, Ch.-E. Guye, C.T.R. Wilson, O.W. Richardson ...
Properties of photons with similarities to waves and or particles
... photon model. The energy of the light was supposed to be carried in the form of a particle or photon, the energy of the photon was defined in terms of the frequency of the radiation, and frequency is an essentially wavelike property! One possible way of dealing with this paradox is to treat light as ...
... photon model. The energy of the light was supposed to be carried in the form of a particle or photon, the energy of the photon was defined in terms of the frequency of the radiation, and frequency is an essentially wavelike property! One possible way of dealing with this paradox is to treat light as ...
Arrangement of Electrons in Atoms
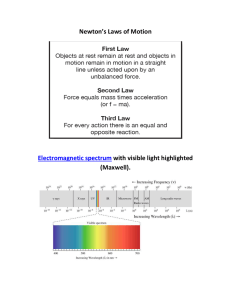
... Called the dual wave-particle nature of light! Electromagnetic Radiation - form of E that exhibits wavelike behavior as it travels thru space Electromagnetic Spectrum - all of the forms of electromagnetic radiation (visible light, x-rays, uv and infrared light, micro and radio waves) ...
... Called the dual wave-particle nature of light! Electromagnetic Radiation - form of E that exhibits wavelike behavior as it travels thru space Electromagnetic Spectrum - all of the forms of electromagnetic radiation (visible light, x-rays, uv and infrared light, micro and radio waves) ...
Problem 1. What is the wavelength of a hydrogen atom... sponding to the mean kinetic energy at thermal equilibrium at...
... size. What is the minimum speed of the virus? [2 points] Problem 4. Consider a monochromatic beam of light of intensity I (units of J · s−1 · m−2 ) striking a perfectly reflecting surface at 90 degrees to it. Using the concept of photons calculate the radiation pressure. [2 points] Problem 5. Photon ...
... size. What is the minimum speed of the virus? [2 points] Problem 4. Consider a monochromatic beam of light of intensity I (units of J · s−1 · m−2 ) striking a perfectly reflecting surface at 90 degrees to it. Using the concept of photons calculate the radiation pressure. [2 points] Problem 5. Photon ...
5.3 section summary
... According to quantum mechanics, the motions of subatomic particles may be described as waves. The wavelength and frequency of light are inversely proportional to each other. When atoms absorb energy, electrons move into higher energy levels. These electrons then lose energy by emitting light when th ...
... According to quantum mechanics, the motions of subatomic particles may be described as waves. The wavelength and frequency of light are inversely proportional to each other. When atoms absorb energy, electrons move into higher energy levels. These electrons then lose energy by emitting light when th ...
Photon
A photon is an elementary particle, the quantum of light and all other forms of electromagnetic radiation. It is the force carrier for the electromagnetic force, even when static via virtual photons. The effects of this force are easily observable at the microscopic and at the macroscopic level, because the photon has zero rest mass; this allows long distance interactions. Like all elementary particles, photons are currently best explained by quantum mechanics and exhibit wave–particle duality, exhibiting properties of waves and of particles. For example, a single photon may be refracted by a lens or exhibit wave interference with itself, but also act as a particle giving a definite result when its position is measured. Waves and quanta, being two observable aspects of a single phenomenon cannot have their true nature described in terms of any mechanical model. A representation of this dual property of light, which assumes certain points on the wave front to be the seat of the energy is also impossible. Thus, the quanta in a light wave cannot be spatially localized. Some defined physical parameters of a photon are listed. The modern photon concept was developed gradually by Albert Einstein in the first years of the 20th century to explain experimental observations that did not fit the classical wave model of light. In particular, the photon model accounted for the frequency dependence of light's energy, and explained the ability of matter and radiation to be in thermal equilibrium. It also accounted for anomalous observations, including the properties of black-body radiation, that other physicists, most notably Max Planck, had sought to explain using semiclassical models, in which light is still described by Maxwell's equations, but the material objects that emit and absorb light do so in amounts of energy that are quantized (i.e., they change energy only by certain particular discrete amounts and cannot change energy in any arbitrary way). Although these semiclassical models contributed to the development of quantum mechanics, many further experiments starting with Compton scattering of single photons by electrons, first observed in 1923, validated Einstein's hypothesis that light itself is quantized. In 1926 the optical physicist Frithiof Wolfers and the chemist Gilbert N. Lewis coined the name photon for these particles, and after 1927, when Arthur H. Compton won the Nobel Prize for his scattering studies, most scientists accepted the validity that quanta of light have an independent existence, and the term photon for light quanta was accepted.In the Standard Model of particle physics, photons and other elementary particles are described as a necessary consequence of physical laws having a certain symmetry at every point in spacetime. The intrinsic properties of particles, such as charge, mass and spin, are determined by the properties of this gauge symmetry.The photon concept has led to momentous advances in experimental and theoretical physics, such as lasers, Bose–Einstein condensation, quantum field theory, and the probabilistic interpretation of quantum mechanics. It has been applied to photochemistry, high-resolution microscopy, and measurements of molecular distances. Recently, photons have been studied as elements of quantum computers and for applications in optical imaging and optical communication such as quantum cryptography.