Question Bank (Class XI - Chemistry)
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
Heterogeneous Catalysis and Solid Catalysts
... Catalysis is a phenomenon by which chemical reactions are accelerated by small quantities of foreign substances, called catalysts. A suitable catalyst can enhance the rate of a thermodynamically feasible reaction but cannot change the position of the thermodynamic equilibrium. Most catalysts are sol ...
... Catalysis is a phenomenon by which chemical reactions are accelerated by small quantities of foreign substances, called catalysts. A suitable catalyst can enhance the rate of a thermodynamically feasible reaction but cannot change the position of the thermodynamic equilibrium. Most catalysts are sol ...
2nd Semester Practice Chemistry Final 2009
... 1. What happens to the volume of a gas during compression? a. The volume increases. b. The volume decreases. c. The volume remains constant. d. It is impossible to tell because all gases are different. 2. Name the compound Fe(NO3)2. a. iron(II) nitrate c. iron(I) nitrite b. iron(II) nitrite d. iron ...
... 1. What happens to the volume of a gas during compression? a. The volume increases. b. The volume decreases. c. The volume remains constant. d. It is impossible to tell because all gases are different. 2. Name the compound Fe(NO3)2. a. iron(II) nitrate c. iron(I) nitrite b. iron(II) nitrite d. iron ...
TR-00-13 - Svensk Kärnbränslehantering AB
... species in the system: Cu - H2O - H+ - H2 - F– - Cl– - S2– - SO42– - NO3– NO2– - NH4+ - PO43– - CO32–. For some reactions and compounds, for which no experimental information on temperature effects was available, entropy and heat capacity values have been estimated. The compiled data were used to ca ...
... species in the system: Cu - H2O - H+ - H2 - F– - Cl– - S2– - SO42– - NO3– NO2– - NH4+ - PO43– - CO32–. For some reactions and compounds, for which no experimental information on temperature effects was available, entropy and heat capacity values have been estimated. The compiled data were used to ca ...
Teacher Edition Calculations
... terms of mole ratios Compare mass changes in samples of metals when they combine with oxygen Perform a first-hand investigation to meas ure and identify the mass ratios of metal to non metal(s) in a common compound and calculate its empirical formula Describe the contribution of Gay-Lussac to the un ...
... terms of mole ratios Compare mass changes in samples of metals when they combine with oxygen Perform a first-hand investigation to meas ure and identify the mass ratios of metal to non metal(s) in a common compound and calculate its empirical formula Describe the contribution of Gay-Lussac to the un ...
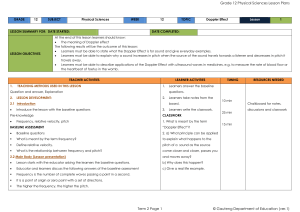
Physical Sciences Grade 12 Term 2
... The very high frequency that ultrasound has a small wavelength. Therefore it can be ...
... The very high frequency that ultrasound has a small wavelength. Therefore it can be ...
Part 1-ICHO-21-25
... This publication contains the competition problems (Volume 2) from the 21st – 40th International Chemistry Olympiads (ICHO) organized in the years 1989 – 2008 and is a continuation of the publication that appeared last year as Volume 1 and contained competition problems from the first twenty ICHOs. ...
... This publication contains the competition problems (Volume 2) from the 21st – 40th International Chemistry Olympiads (ICHO) organized in the years 1989 – 2008 and is a continuation of the publication that appeared last year as Volume 1 and contained competition problems from the first twenty ICHOs. ...
ANNEX (Manuscrits posteriors a la Comissió de Doctorat de Juliol del...
... or amines,7 phenolate, dialkyl or diarylphosphite8 and Nalkylcarbamoyldiphenylphosphine oxides9 resulting in one anionic species as a consequence of the opening of the dioxane ring. But mono or di B(8)-R (R= alkyl) substituted derivatives of [1]- have not yet been reported. In this work we report on ...
... or amines,7 phenolate, dialkyl or diarylphosphite8 and Nalkylcarbamoyldiphenylphosphine oxides9 resulting in one anionic species as a consequence of the opening of the dioxane ring. But mono or di B(8)-R (R= alkyl) substituted derivatives of [1]- have not yet been reported. In this work we report on ...
By Martin S. Silberberg
... Fahrenheit temperature scales Figure 1.12 8 Precision and accuracy in a laboratory calibration Figure 1.16 9 End of chapter problems 1.1, 1.74, 1.76 10 Elements, compounds, and mixtures on the atomic scale Figure 2.1 11 Experiments to determine the properties of cathode rays Figure 2.5 12 Millikan’s ...
... Fahrenheit temperature scales Figure 1.12 8 Precision and accuracy in a laboratory calibration Figure 1.16 9 End of chapter problems 1.1, 1.74, 1.76 10 Elements, compounds, and mixtures on the atomic scale Figure 2.1 11 Experiments to determine the properties of cathode rays Figure 2.5 12 Millikan’s ...
ch15
... Lead(II) chromate has been used as a bright yellow pigment in some paints. It can be produced by the reaction of potassium chromate with lead nitrate. a Write a full equation for this reaction. b What mass of potassium chromate is required to produce 6.0 g of lead chromate? c Suggest a reason why le ...
... Lead(II) chromate has been used as a bright yellow pigment in some paints. It can be produced by the reaction of potassium chromate with lead nitrate. a Write a full equation for this reaction. b What mass of potassium chromate is required to produce 6.0 g of lead chromate? c Suggest a reason why le ...
Chapter 15 Calculations in chemistry: stoichiometry
... Lead(II) chromate has been used as a bright yellow pigment in some paints. It can be produced by the reaction of potassium chromate with lead nitrate. a Write a full equation for this reaction. b What mass of potassium chromate is required to produce 6.0 g of lead chromate? c Suggest a reason why le ...
... Lead(II) chromate has been used as a bright yellow pigment in some paints. It can be produced by the reaction of potassium chromate with lead nitrate. a Write a full equation for this reaction. b What mass of potassium chromate is required to produce 6.0 g of lead chromate? c Suggest a reason why le ...
Chemistry Midterm Exam Review
... ____ 46. A change in the force of gravity on an object will affect its a. mass. c. weight. b. density. d. kinetic energy. ____ 47. Which of these is a measure of the amount of material? a. density c. volume b. weight d. mass ____ 48. Which of these statements about mass is true? a. Mass is expressed ...
... ____ 46. A change in the force of gravity on an object will affect its a. mass. c. weight. b. density. d. kinetic energy. ____ 47. Which of these is a measure of the amount of material? a. density c. volume b. weight d. mass ____ 48. Which of these statements about mass is true? a. Mass is expressed ...
Chapter 4 - Chemistry
... Strategy: In order to break a redox reaction down into an oxidation half-reaction and a reduction halfreaction, you should first assign oxidation numbers to all the atoms in the reaction. In this way, you can determine which element is oxidized (loses electrons) and which element is reduced (gains e ...
... Strategy: In order to break a redox reaction down into an oxidation half-reaction and a reduction halfreaction, you should first assign oxidation numbers to all the atoms in the reaction. In this way, you can determine which element is oxidized (loses electrons) and which element is reduced (gains e ...
PART 6-ICHO-26-30
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
ch final rvw
... a. Substances are compounds, and mixtures are not. b. Mixtures are groupings of elements, and compounds are not. c. Samples of the same substance can have different intensive properties. d. Mixtures can be separated physically, while compounds cannot. The first figure in a properly written chemical ...
... a. Substances are compounds, and mixtures are not. b. Mixtures are groupings of elements, and compounds are not. c. Samples of the same substance can have different intensive properties. d. Mixtures can be separated physically, while compounds cannot. The first figure in a properly written chemical ...
Stoichiometry - Milton
... and light. To account for this emission, scientists proposed a theory that combustion depended on the emission of a substance called phlogiston, which appeared as a combination of energy as heat and light while the material was burning but which could not be detected beforehand. The phlogiston theor ...
... and light. To account for this emission, scientists proposed a theory that combustion depended on the emission of a substance called phlogiston, which appeared as a combination of energy as heat and light while the material was burning but which could not be detected beforehand. The phlogiston theor ...
Answers Chapters 1-3 bookwork - Dunmore High School
... Chemical property. Oxygen gas is consumed in a combustion reaction; its composition and identity are changed. ...
... Chemical property. Oxygen gas is consumed in a combustion reaction; its composition and identity are changed. ...
9 SHS CH 9 LECTURE shs_ch_9_lecture
... How many moles of NaOH will react with 3.21 moles of sulfuric acid? Step 1. Balanced the chemical equation. Step 2. Write down the given numeric info. If it is not in mole units convert it to moles using the molecular mass as a conversion factor. Step 3. Now convert from the moles of starting substa ...
... How many moles of NaOH will react with 3.21 moles of sulfuric acid? Step 1. Balanced the chemical equation. Step 2. Write down the given numeric info. If it is not in mole units convert it to moles using the molecular mass as a conversion factor. Step 3. Now convert from the moles of starting substa ...
The role of aqueous-phase oxidation in the A
... partitioning of reactants, intermediates, and products. This allows for the first direct comparison of aqueous oxidation under drastically different partitioning conditions. These experiments are presented in Chapter 3. In order to address the final research question (how do aqueous-phase oxidants a ...
... partitioning of reactants, intermediates, and products. This allows for the first direct comparison of aqueous oxidation under drastically different partitioning conditions. These experiments are presented in Chapter 3. In order to address the final research question (how do aqueous-phase oxidants a ...
Part 2-ICHO-26-30
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
Moles 1 - pedagogics.ca
... and stoichiometry is the study of the ratios in which chemical substances combine. In order to know the exact quantity of each substance that is required to react we need to know the number of atoms, molecules or ions present in a specific amount of that substance. However, the mass of an individual ...
... and stoichiometry is the study of the ratios in which chemical substances combine. In order to know the exact quantity of each substance that is required to react we need to know the number of atoms, molecules or ions present in a specific amount of that substance. However, the mass of an individual ...
Oxidative Alihatic Carbon-Carbon Bond Cleavage Reactions
... and in developing new reactions for organic synthesis of fine chemicals including pharmaceuticals. Ideally these reactions would be carried out with high atom economy at low temperatures and pressures, and using earth-abundant elements as reagents and catalysts. With these points in mind, nature pro ...
... and in developing new reactions for organic synthesis of fine chemicals including pharmaceuticals. Ideally these reactions would be carried out with high atom economy at low temperatures and pressures, and using earth-abundant elements as reagents and catalysts. With these points in mind, nature pro ...
CS SuppT7(E).indd
... 2CO2(g) ΔH = –788 kJ Which of the following statements is correct? A 286 kJ of heat are released when 1 mole of steam is formed from its elements. B The enthalpy change of combustion of hydrogen is more exothermic than that of ...
... 2CO2(g) ΔH = –788 kJ Which of the following statements is correct? A 286 kJ of heat are released when 1 mole of steam is formed from its elements. B The enthalpy change of combustion of hydrogen is more exothermic than that of ...
Chapter 15 Calculations in chemistry: stoichiometry
... potassium hydroxide. The amount of each is found using n = cV, where c is the concentration in mol L–1, and V is the volume in litres. n(KOH) = 1.00 × 0.0100 = 0.0100 mol n(H3PO4) = 2.0 × 0.0325 = 0.0650 mol Use n(KOH) provided to find n(H3PO4) required to react completely with the KOH. From the equ ...
... potassium hydroxide. The amount of each is found using n = cV, where c is the concentration in mol L–1, and V is the volume in litres. n(KOH) = 1.00 × 0.0100 = 0.0100 mol n(H3PO4) = 2.0 × 0.0325 = 0.0650 mol Use n(KOH) provided to find n(H3PO4) required to react completely with the KOH. From the equ ...