The atom - KCPE-KCSE
... More about atomic number Each element has a definite and fixed number of protons. If the number of protons changes, then the atom becomes a different element. Changes in the number of particles in the nucleus (protons or neutrons) is very rare. It only takes place in nuclear processes such as: ...
... More about atomic number Each element has a definite and fixed number of protons. If the number of protons changes, then the atom becomes a different element. Changes in the number of particles in the nucleus (protons or neutrons) is very rare. It only takes place in nuclear processes such as: ...
Chapter 4
... a. Atoms are tiny indivisible particles. b. Atoms of the same element are identical. c. Compounds are made by combining atoms. d. Atoms of different elements can combine with one another in simple whole number ratios. The comparison of the number of atoms in a copper coin the size of a penny with th ...
... a. Atoms are tiny indivisible particles. b. Atoms of the same element are identical. c. Compounds are made by combining atoms. d. Atoms of different elements can combine with one another in simple whole number ratios. The comparison of the number of atoms in a copper coin the size of a penny with th ...
“atom”?
... at a piece of Dssue paper and it came back and hit you. On consideraDon, I realized that this scaGering backward must be the result of a single collision, and when I made calculaDons I saw th ...
... at a piece of Dssue paper and it came back and hit you. On consideraDon, I realized that this scaGering backward must be the result of a single collision, and when I made calculaDons I saw th ...
Mass Number, A
... • Protons: Atomic Number (from periodic table) • Neutrons: Mass Number minus the number of protons (mass number is protons and neutrons because the mass of electrons is negligible) ...
... • Protons: Atomic Number (from periodic table) • Neutrons: Mass Number minus the number of protons (mass number is protons and neutrons because the mass of electrons is negligible) ...
Chapter #4 Section Assessment #1 - 33
... nonmetals, and elements touching the staircase mostly metalloids. ...
... nonmetals, and elements touching the staircase mostly metalloids. ...
Excerpt - Assets - Cambridge
... shown in the Appendix and explained, much more fully, in Chapter 7.) All matter is composed of extremely small particles, called atoms. About 100 years ago, the accepted model for atoms included the assumptions that (i) atoms were tiny particles, which could not be divided further or destroyed, and ...
... shown in the Appendix and explained, much more fully, in Chapter 7.) All matter is composed of extremely small particles, called atoms. About 100 years ago, the accepted model for atoms included the assumptions that (i) atoms were tiny particles, which could not be divided further or destroyed, and ...
Slide 1
... number of protons in each atom of a particular element. ☺The atomic number _________ the element! ...
... number of protons in each atom of a particular element. ☺The atomic number _________ the element! ...
Atoms and the Periodic Table
... 3. Using the simulation, talk with your group as you play with the parts of atoms to find: A. What parts go in the center of the atom? _________________________________________ B. You can call the center of the atom, the nucleus. Most atoms in our environment have a stable nucleus. Explore the simul ...
... 3. Using the simulation, talk with your group as you play with the parts of atoms to find: A. What parts go in the center of the atom? _________________________________________ B. You can call the center of the atom, the nucleus. Most atoms in our environment have a stable nucleus. Explore the simul ...
1 - PTO
... atom’s!nucleus!and!places!those!with!similar! chemical!properties!in!columns.!The!repeating! patterns!of!this!table!reflect!patterns!of!outer! electron!states.!(HSEPS1E1),(HSEPS1E2)! PS1.A:!Structure!and!Properties!of!Matter!! ...
... atom’s!nucleus!and!places!those!with!similar! chemical!properties!in!columns.!The!repeating! patterns!of!this!table!reflect!patterns!of!outer! electron!states.!(HSEPS1E1),(HSEPS1E2)! PS1.A:!Structure!and!Properties!of!Matter!! ...
weighted average atomic mass
... • Find out the names that 110, 111, 112, 114, and 116 have now been given. What is the latest news about element 118? • Who/what makes the decisions about element names? • How long does it take for a name to be decided upon? • Record your source(s) using MLA formatting. Read your notes about Chromat ...
... • Find out the names that 110, 111, 112, 114, and 116 have now been given. What is the latest news about element 118? • Who/what makes the decisions about element names? • How long does it take for a name to be decided upon? • Record your source(s) using MLA formatting. Read your notes about Chromat ...
atom - geraldinescience
... • Because gamma rays are massless, the emission of gamma rays by themselves cannot result in the formation of a new atom. ...
... • Because gamma rays are massless, the emission of gamma rays by themselves cannot result in the formation of a new atom. ...
Atomic History and Structure:
... 1. matter is composed of indivisible particles Atoms Can Be Divided, but only in a nuclear reaction 2. all atoms of a particular element are identical Does Not Account for Isotopes (atoms of the same element but a different mass due to a different number of neutrons)! 3. different elements have diff ...
... 1. matter is composed of indivisible particles Atoms Can Be Divided, but only in a nuclear reaction 2. all atoms of a particular element are identical Does Not Account for Isotopes (atoms of the same element but a different mass due to a different number of neutrons)! 3. different elements have diff ...
Periodicity - Teach-n-Learn-Chem
... The Modern periodic table is based on the periodic law. Periodic law - properties of elements tend to change with increasing atomic number in a periodic way ...
... The Modern periodic table is based on the periodic law. Periodic law - properties of elements tend to change with increasing atomic number in a periodic way ...
Biochemistry-Macromolecules
... Very small is size, but has a very large mass A: What is a nucleus? S2C06 Jeopardy Review ...
... Very small is size, but has a very large mass A: What is a nucleus? S2C06 Jeopardy Review ...
ppt notes
... different than A and BA element Atoms of element A and B can be can be physically chemically combined mixed together as a compound ...
... different than A and BA element Atoms of element A and B can be can be physically chemically combined mixed together as a compound ...
atomic - Humble ISD
... The Atom The smallest particle of an element that still retains the properties of the element. Atoms are much too small to see ...
... The Atom The smallest particle of an element that still retains the properties of the element. Atoms are much too small to see ...
Mass/Mole Conversions
... Isotopes • Isotopes: atoms of the same element that have different masses due to different numbers of neutrons. • Mass Number: the total number of protons and neutrons that make up the nucleus of an isotope ~ Isotopes are written with the mass number written after the element name or symbol with a ...
... Isotopes • Isotopes: atoms of the same element that have different masses due to different numbers of neutrons. • Mass Number: the total number of protons and neutrons that make up the nucleus of an isotope ~ Isotopes are written with the mass number written after the element name or symbol with a ...
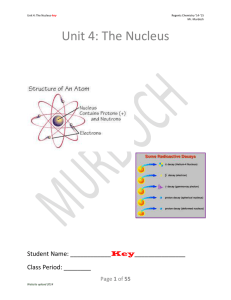
Unit 4: The Nucleus
... 2. Atomic Mass Unit (amu): 1/12 the mass of a C-12 atom; the approximate mass of either a proton or neutron. 3. Atomic Number: The number that identifies an element, equal to an atom’s number of protons. 4. Deflect: Change in direction due to an outside force. 5. Emit: To give off something. 6. Half ...
... 2. Atomic Mass Unit (amu): 1/12 the mass of a C-12 atom; the approximate mass of either a proton or neutron. 3. Atomic Number: The number that identifies an element, equal to an atom’s number of protons. 4. Deflect: Change in direction due to an outside force. 5. Emit: To give off something. 6. Half ...
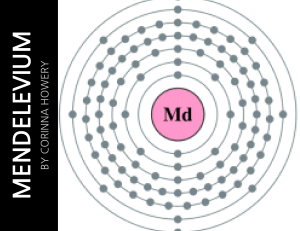
Mendelevium
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
Chapter 4 Atoms and Elements
... SKILLBUILDER 4.4 Determining Ion Charge from Numbers of Protons and Electrons Determine the charge of each ion. (a) a nickel ion with 26 electrons (b) a bromine ion with 36 electrons (c) a phosphorus ion with 18 electrons ...
... SKILLBUILDER 4.4 Determining Ion Charge from Numbers of Protons and Electrons Determine the charge of each ion. (a) a nickel ion with 26 electrons (b) a bromine ion with 36 electrons (c) a phosphorus ion with 18 electrons ...
Name
... element and the relative abundance of its isotopes. a. In nature, most elements occur as a mixture of two or more isotopes. b. Isotopes of an element do not have a specific natural percent abundance. c. The average atomic mass of an element is usually closest to that of the isotope with the highest ...
... element and the relative abundance of its isotopes. a. In nature, most elements occur as a mixture of two or more isotopes. b. Isotopes of an element do not have a specific natural percent abundance. c. The average atomic mass of an element is usually closest to that of the isotope with the highest ...
electrons and the structure of atoms
... element and the relative abundance of its isotopes. a. In nature, most elements occur as a mixture of two or more isotopes. b. Isotopes of an element do not have a specific natural percent abundance. c. The average atomic mass of an element is usually closest to that of the isotope with the highest ...
... element and the relative abundance of its isotopes. a. In nature, most elements occur as a mixture of two or more isotopes. b. Isotopes of an element do not have a specific natural percent abundance. c. The average atomic mass of an element is usually closest to that of the isotope with the highest ...
Sub Unit Plan 1 Chem Periodic Table
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
Chapter 11 section 2 questions - the atom
... Electrons are the negatively charged particles found in the energy levels that surround the nucleus - this like the rings on a target! Electrons have a VERY small mass - almost inconsequential to the atomic mass. Electrons carry a negative charge and are held in place by the positively charged proto ...
... Electrons are the negatively charged particles found in the energy levels that surround the nucleus - this like the rings on a target! Electrons have a VERY small mass - almost inconsequential to the atomic mass. Electrons carry a negative charge and are held in place by the positively charged proto ...
Promethium

Promethium, originally prometheum, is a chemical element with symbol Pm and atomic number 61. All of its isotopes are radioactive; it is one of only two such elements that are followed in the periodic table by elements with stable forms, a distinction shared with technetium. Chemically, promethium is a lanthanide, which forms salts when combined with other elements. Promethium shows only one stable oxidation state of +3; however, a few +2 compounds may exist.In 1902, Bohuslav Brauner suggested there was an element with properties intermediate between those of the known elements neodymium (60) and samarium (62); this was confirmed in 1914 by Henry Moseley who, having measured the atomic numbers of all the elements then known, found there was an element with atomic number 61. In 1926, an Italian and an American group claimed to have isolated a sample of element 61; both ""discoveries"" were soon proven to be false. In 1938, during a nuclear experiment conducted at Ohio State University, a few radioactive nuclides were produced that certainly were not radioisotopes of neodymium or samarium, but there was a lack of chemical proof that element 61 was produced, and the discovery was not generally recognized. Promethium was first produced and characterized at Oak Ridge National Laboratory in 1945 by the separation and analysis of the fission products of uranium fuel irradiated in a graphite reactor. The discoverers proposed the name ""prometheum"" (the spelling was subsequently changed), derived from Prometheus, the Titan in Greek mythology who stole fire from Mount Olympus and brought it down to humans, to symbolize ""both the daring and the possible misuse of mankind's intellect"". However, a sample of the metal was made only in 1963.There are two possible sources for natural promethium: rare decays of natural europium-151 (producing promethium-147), and uranium (various isotopes). Practical applications exist only for chemical compounds of promethium-147, which are used in luminous paint, atomic batteries, and thickness measurement devices, even though promethium-145 is the most stable promethium isotope. Because natural promethium is exceedingly scarce, it is typically synthesized by bombarding uranium-235 (enriched uranium) with thermal neutrons to produce promethium-147.