Overall Score: _____ / 22 (each question is worth
... Metals are more dense than nonmetals. The particles in a metal are more tightly packed together than nonmetals (remember that metals are solid at room temperature, while nonmetals are gas at room temperature). Therefore, the ratio of mass to volume (density) is greater in metals than in nonmetals. M ...
... Metals are more dense than nonmetals. The particles in a metal are more tightly packed together than nonmetals (remember that metals are solid at room temperature, while nonmetals are gas at room temperature). Therefore, the ratio of mass to volume (density) is greater in metals than in nonmetals. M ...
atoms - Fort Bend ISD
... Sizing up the Atom Elements are able to be subdivided into smaller and smaller particles – these are the atoms, and they still have properties of that element If you could line up 100,000,000 copper atoms in a single file, they would be approximately 1 cm long Despite their small size, individu ...
... Sizing up the Atom Elements are able to be subdivided into smaller and smaller particles – these are the atoms, and they still have properties of that element If you could line up 100,000,000 copper atoms in a single file, they would be approximately 1 cm long Despite their small size, individu ...
Fall 2015 Review-2
... ____ 37. The metals in Groups 1A, 2A, and 3A ____. a. gain electrons when they form ions c. all have ions with a 1 charge b. all form ions with a negative charge d. lose electrons when they form ions ____ 38. Which of the following statements correctly compares the relative size of an ion to its neu ...
... ____ 37. The metals in Groups 1A, 2A, and 3A ____. a. gain electrons when they form ions c. all have ions with a 1 charge b. all form ions with a negative charge d. lose electrons when they form ions ____ 38. Which of the following statements correctly compares the relative size of an ion to its neu ...
Physical Science – Chapter 4 Study Guide
... Know and understand John Dalton’s atomic theory. Know and understand J.J. Thomson’s experiments with the atom. Who provided evidence for the existence of a nucleus in an atom? Know and understand the atomic model and the nucleus. Know and understand neutrons, electrons, and protons of the elements. ...
... Know and understand John Dalton’s atomic theory. Know and understand J.J. Thomson’s experiments with the atom. Who provided evidence for the existence of a nucleus in an atom? Know and understand the atomic model and the nucleus. Know and understand neutrons, electrons, and protons of the elements. ...
I. Atoms are the smallest forms
... – Atoms changing identity • Chemical reactions do not effect the nucleus of an atom • Certain conditions can change the number of protons • Each atom has isotopes with different numbers of neutrons • Stability of nucleus depends on the right number of protons and neutrons • Too few or too many neutr ...
... – Atoms changing identity • Chemical reactions do not effect the nucleus of an atom • Certain conditions can change the number of protons • Each atom has isotopes with different numbers of neutrons • Stability of nucleus depends on the right number of protons and neutrons • Too few or too many neutr ...
Chapter 2 – Atoms and Elements
... receptor molecules in our taste buds. Due to their structure, the ethanol molecules travel easily through the human body (they are soluble in both water and fat) until they reach their target, the brain. Chemistry is often termed “the central science”. It lies between biology and physics, and chemic ...
... receptor molecules in our taste buds. Due to their structure, the ethanol molecules travel easily through the human body (they are soluble in both water and fat) until they reach their target, the brain. Chemistry is often termed “the central science”. It lies between biology and physics, and chemic ...
Chapter 2 – Atoms and Elements
... receptor molecules in our taste buds. Due to their structure, the ethanol molecules travel easily through the human body (they are soluble in both water and fat) until they reach their target, the brain. Chemistry is often termed “the central science”. It lies between biology and physics, and chemic ...
... receptor molecules in our taste buds. Due to their structure, the ethanol molecules travel easily through the human body (they are soluble in both water and fat) until they reach their target, the brain. Chemistry is often termed “the central science”. It lies between biology and physics, and chemic ...
Document
... Analyze: We’re told that a rock sample has a certain amount of lead-206 for every unit weight of uranium238 and asked to estimate the age of the rock. Plan: Presumably the lead-206 is due entirely to radioactive decay of uranium-228 to form lead-206, with a known half-life. To apply first-order kine ...
... Analyze: We’re told that a rock sample has a certain amount of lead-206 for every unit weight of uranium238 and asked to estimate the age of the rock. Plan: Presumably the lead-206 is due entirely to radioactive decay of uranium-228 to form lead-206, with a known half-life. To apply first-order kine ...
Big Science from the Small World of Atom
... periodic table. On clicking a particular element in the alive periodic table, the atomic structure of the element, including the number of protons, neutrons and electrons, atomic mass, electron occupation in the different shells (energy levels) so that the number of core electrons and the valence el ...
... periodic table. On clicking a particular element in the alive periodic table, the atomic structure of the element, including the number of protons, neutrons and electrons, atomic mass, electron occupation in the different shells (energy levels) so that the number of core electrons and the valence el ...
Name: Date: ______ Period: _____ Chemistry 1st semester final
... 40. What type of elements is found in the lower left hand part of the periodic table?metals 41. Where are metalloids found on the periodic table?Along the dark (or red) stair case line 42. From which orbital in a lithium atom is an electron transferred to form Li ion? Looses electrons from 2s 43. Wh ...
... 40. What type of elements is found in the lower left hand part of the periodic table?metals 41. Where are metalloids found on the periodic table?Along the dark (or red) stair case line 42. From which orbital in a lithium atom is an electron transferred to form Li ion? Looses electrons from 2s 43. Wh ...
Chapter 4 power point notes
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
ch 4 ppt - Madison County Schools
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
Chapter 4 PPT
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
Radioactive Decay Mechanisms
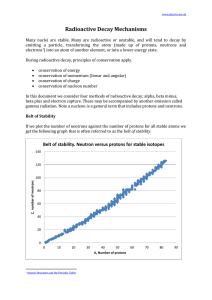
... Note that 1. there are no stable nuclei with an atomic number of 84 or greater. 2. for smaller atoms the ratio neutrons:protons is 1:1 3. for larger atoms the ratio of neutrons:protons approaches about 1.5:1 Since the strong nuclear force weakens significantly with distance, as the atom gets bigger. ...
... Note that 1. there are no stable nuclei with an atomic number of 84 or greater. 2. for smaller atoms the ratio neutrons:protons is 1:1 3. for larger atoms the ratio of neutrons:protons approaches about 1.5:1 Since the strong nuclear force weakens significantly with distance, as the atom gets bigger. ...
CMC Chapter 04
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
... surrounding an atom. Nuclear reactions involve changes in the nucleus of an atom. ...
Chapter 18 Comparing Atoms Lab
... many marbles, and of as many colors as they need but must take at least as many total marbles as they put in. For example, a player can trade 2 yellows for 1 yellow, 1 blue, and 1 red. 11. Which particles are found in an atom’s nucleus? Which particles are found outside the nucleus? 12. What four el ...
... many marbles, and of as many colors as they need but must take at least as many total marbles as they put in. For example, a player can trade 2 yellows for 1 yellow, 1 blue, and 1 red. 11. Which particles are found in an atom’s nucleus? Which particles are found outside the nucleus? 12. What four el ...
2.1 The Nature of Matter Notes
... positively charged nucleus but remain outside the nucleus because of the energy of their motion. Energy level - e- have more energy the farther they are from the nucleus ...
... positively charged nucleus but remain outside the nucleus because of the energy of their motion. Energy level - e- have more energy the farther they are from the nucleus ...
atoms - schultz915
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
The Periodic Table
... You will learn how to…….. Compute the atomic mass and mass number of an atom. Identify isotopes of common elements Interpret the average atomic mass of an element This is important because most elements exist in more than one form. Some are radioactive, and others are not. ...
... You will learn how to…….. Compute the atomic mass and mass number of an atom. Identify isotopes of common elements Interpret the average atomic mass of an element This is important because most elements exist in more than one form. Some are radioactive, and others are not. ...
Atomic Structure 3: ISOTOPES
... element have the same number of protons, as given by the atomic number. You also know that the number of electrons in an uncharged atom equals the number of protons. The number of neutrons in the nucleus of an atom is not so predictable. The periodic table helps us out, though, by giving the atomic ...
... element have the same number of protons, as given by the atomic number. You also know that the number of electrons in an uncharged atom equals the number of protons. The number of neutrons in the nucleus of an atom is not so predictable. The periodic table helps us out, though, by giving the atomic ...
AP CHEMISTRY SUMMER ASSIGNMENT AP Chemistry is a
... conductors, shiny, malleable and ductile. They are solids except for liquid mercury. Nonmetals – elements located to the right of the staircase, plus Hydrogen; they share or gain electrons. Nonmetals can be solids, liquid (Br2) or gases. They are brittle and are poor conductors of electricity. Metal ...
... conductors, shiny, malleable and ductile. They are solids except for liquid mercury. Nonmetals – elements located to the right of the staircase, plus Hydrogen; they share or gain electrons. Nonmetals can be solids, liquid (Br2) or gases. They are brittle and are poor conductors of electricity. Metal ...
Chapter 3—Time and Geology
... atom (34): The smallest particle of matter that can exist as a chemical element. atomic mass (34): A quantity essentially equivalent to the number of neutrons plus the number of protons in an atomic nucleus. atomic number (34): The number of protons in the nuclei of atoms of a particular element. An ...
... atom (34): The smallest particle of matter that can exist as a chemical element. atomic mass (34): A quantity essentially equivalent to the number of neutrons plus the number of protons in an atomic nucleus. atomic number (34): The number of protons in the nuclei of atoms of a particular element. An ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.