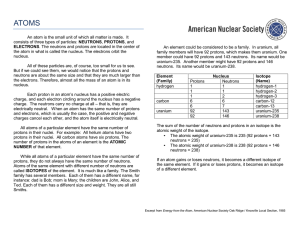
ATOMS AND ELEMENTS Evolution of Atomic Theory
... The ancient Greek scientist Democritus is often credited with developing the idea of the atom ¨ Democritus proposed that matter was, on the smallest scale, composed of particles described as atomos, meaning “indivisible” ¨ While atoms can in fact be broken down into smaller particles, the atoms ...
... The ancient Greek scientist Democritus is often credited with developing the idea of the atom ¨ Democritus proposed that matter was, on the smallest scale, composed of particles described as atomos, meaning “indivisible” ¨ While atoms can in fact be broken down into smaller particles, the atoms ...
Chemistry Atoms Learning Objectives Atoms Essential knowledge
... The atomic number of an element is the same as the number of protons. In a neutral atom, the number of electrons is the same as the number of protons. All atoms of an element have the same number of protons. The average atomic mass for each element is the weighted average of that element’s naturally ...
... The atomic number of an element is the same as the number of protons. In a neutral atom, the number of electrons is the same as the number of protons. All atoms of an element have the same number of protons. The average atomic mass for each element is the weighted average of that element’s naturally ...
Review Questions
... electricity, high density, high melting temperature. Nonmetals tend to be gases or brittle solids at room temperature, poor conductors of heat and electricity (insulators), low density, low melting temperature Metalloids (Semi-metals) dull, brittle, semi-conductors (used in computer chips), properti ...
... electricity, high density, high melting temperature. Nonmetals tend to be gases or brittle solids at room temperature, poor conductors of heat and electricity (insulators), low density, low melting temperature Metalloids (Semi-metals) dull, brittle, semi-conductors (used in computer chips), properti ...
File
... Isotopes are atoms of the same element that have different masses. Why are these masses different?? Since number of protons doesn’t change, the change in mass must be due to different number of neutrons. ...
... Isotopes are atoms of the same element that have different masses. Why are these masses different?? Since number of protons doesn’t change, the change in mass must be due to different number of neutrons. ...
atom
... Isotopes are identified by their mass numbers. For example, carbon has three isotopes—carbon-12, carbon-13, and carbon-14. Each isotope has a different number of neutrons. ...
... Isotopes are identified by their mass numbers. For example, carbon has three isotopes—carbon-12, carbon-13, and carbon-14. Each isotope has a different number of neutrons. ...
Periodic Table for class
... In other words, electron shells have a limited capacity for electrons. As you might expect, the farther an electron shell is from the nucleus, the larger it is. You can calculate the total capacity of an electron shell using the formula 2n2, where n equals the number of the electron shell. For examp ...
... In other words, electron shells have a limited capacity for electrons. As you might expect, the farther an electron shell is from the nucleus, the larger it is. You can calculate the total capacity of an electron shell using the formula 2n2, where n equals the number of the electron shell. For examp ...
Atomic Structure Notes Blank
... c. Very dense (Extremely small % of total volume of atom, BUT 99.97% of its _________) 2. Outside nucleus a. 99.9% of atom is this empty space through which the _____________ travel. b. Overall __________________ charge C. How they fit together 1. Electrons are held within the atom due to their attr ...
... c. Very dense (Extremely small % of total volume of atom, BUT 99.97% of its _________) 2. Outside nucleus a. 99.9% of atom is this empty space through which the _____________ travel. b. Overall __________________ charge C. How they fit together 1. Electrons are held within the atom due to their attr ...
Chapter 4 Review ans.. - hrsbstaff.ednet.ns.ca
... density, high melting temperature. Nonmetals tend to be gases or brittle solids at room temperature, poor conductors of heat and electricity (insulators), low density, low melting temperature Metalloids (Semi-metals) dull, brittle, semi-conductors (used in computer chips), properties of both metals ...
... density, high melting temperature. Nonmetals tend to be gases or brittle solids at room temperature, poor conductors of heat and electricity (insulators), low density, low melting temperature Metalloids (Semi-metals) dull, brittle, semi-conductors (used in computer chips), properties of both metals ...
Periodic Table for class
... In other words, electron shells have a limited capacity for electrons. As you might expect, the farther an electron shell is from the nucleus, the larger it is. You can calculate the total capacity of an electron shell using the formula 2n2, where n equals the number of the electron shell. For examp ...
... In other words, electron shells have a limited capacity for electrons. As you might expect, the farther an electron shell is from the nucleus, the larger it is. You can calculate the total capacity of an electron shell using the formula 2n2, where n equals the number of the electron shell. For examp ...
The Atom
... A. To find the number of neutrons an atom has 1. First you have to know the number of protons – that’s the atomic number 2. Then you have to know the atomic mass number 3. When you subtract the atomic number from the mass number rounded to nearest whole number 4. You get the number of neutrons Mass ...
... A. To find the number of neutrons an atom has 1. First you have to know the number of protons – that’s the atomic number 2. Then you have to know the atomic mass number 3. When you subtract the atomic number from the mass number rounded to nearest whole number 4. You get the number of neutrons Mass ...
A Few Laws • Conservation of Matter-For any
... • In chemistry, we deal with matter on the large scale • Dalton’s Law tells us we need to measure elements in some manner that can be looked at as an “atomic measure” • If all apples weighed 23.0ounces and all oranges weighed 13.2ounces, I can readily scale these up to any other weight and be assure ...
... • In chemistry, we deal with matter on the large scale • Dalton’s Law tells us we need to measure elements in some manner that can be looked at as an “atomic measure” • If all apples weighed 23.0ounces and all oranges weighed 13.2ounces, I can readily scale these up to any other weight and be assure ...
Why are atoms of lead different to those of gold and why can we not
... turn lead into gold, but why can we not? Why are atoms of lead different to those of gold and why can we not just simply change them? ...
... turn lead into gold, but why can we not? Why are atoms of lead different to those of gold and why can we not just simply change them? ...
Chapter 4 Chemical Foundations: Elements, Atoms, and Ions
... 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same relative numbers and types of atoms. 5. Atoms are indivisible in chemical processes. That is, atoms are not created or destroyed in chemical reactions. A chemical reaction simply ...
... 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same relative numbers and types of atoms. 5. Atoms are indivisible in chemical processes. That is, atoms are not created or destroyed in chemical reactions. A chemical reaction simply ...
Chapter 4
... • he determined that the ray was made of tiny negatively charged particles we call electrons • his measurements led him to conclude that these electrons were smaller than a hydrogen atom • if electrons are smaller than atoms, they must be pieces of atoms • if atoms have pieces, they must be breakabl ...
... • he determined that the ray was made of tiny negatively charged particles we call electrons • his measurements led him to conclude that these electrons were smaller than a hydrogen atom • if electrons are smaller than atoms, they must be pieces of atoms • if atoms have pieces, they must be breakabl ...
The Modern Atomic Model
... • Every atom of an element will always have the same number of protons • Carbon will always have 6 protons, oxygen will always have 8 protons, and iron will always have 26 protons. ...
... • Every atom of an element will always have the same number of protons • Carbon will always have 6 protons, oxygen will always have 8 protons, and iron will always have 26 protons. ...
Atomic Structure
... • Atoms acquire this charge by gaining or losing electrons from their outermost level = valence electrons. ...
... • Atoms acquire this charge by gaining or losing electrons from their outermost level = valence electrons. ...
atom - BobcatChemistry
... The Chapter Resources Menu will allow you to access chapter specific resources from the Chapter Menu or any Chapter Outline slide. From within any feature, click the Resources tab to return to this slide. The “Return” button will allow you to return to the slide that you were viewing when you clicke ...
... The Chapter Resources Menu will allow you to access chapter specific resources from the Chapter Menu or any Chapter Outline slide. From within any feature, click the Resources tab to return to this slide. The “Return” button will allow you to return to the slide that you were viewing when you clicke ...
An atom is the small unit of which all matter is made. It consists of
... neutrons are about the same size and that they are much larger than the electrons. Therefore, almost all the mass of an atom is in its nucleus. Each proton in an atom’s nucleus has a positive electric charge, and each electron circling around the nucleus has a negative charge. The neutrons carry no ...
... neutrons are about the same size and that they are much larger than the electrons. Therefore, almost all the mass of an atom is in its nucleus. Each proton in an atom’s nucleus has a positive electric charge, and each electron circling around the nucleus has a negative charge. The neutrons carry no ...
MODERN PHYSICS MASS DEFECT (∆m)
... In the radioactive series whenever the mass number changes it changes by _____ The time required for half of the atoms of a radioactive substance to disintegrate is known as _____ The decay constant λ = _____ Atoms of different elements having same mass number but different atomic number are called ...
... In the radioactive series whenever the mass number changes it changes by _____ The time required for half of the atoms of a radioactive substance to disintegrate is known as _____ The decay constant λ = _____ Atoms of different elements having same mass number but different atomic number are called ...
CP NT Ch. 4 and 25 v2
... C. He proved that nuclear reactions can be produced artificially. D. Induced transmutation can occur by bombarding an atom with _______ particles, protons or neutrons. III. Transuranium Elements A. Elements with atomic number above _______. B. All transuranium elements undergo transmutation C. None ...
... C. He proved that nuclear reactions can be produced artificially. D. Induced transmutation can occur by bombarding an atom with _______ particles, protons or neutrons. III. Transuranium Elements A. Elements with atomic number above _______. B. All transuranium elements undergo transmutation C. None ...
Atomic number - KCPE-KCSE
... When an unstable nucleus emits an alpha particle its atomic number falls by _______ and its mass number by ______. Beta particles are emitted by nuclei with too many ________. In this case the atomic number increases by ______ while the ...
... When an unstable nucleus emits an alpha particle its atomic number falls by _______ and its mass number by ______. Beta particles are emitted by nuclei with too many ________. In this case the atomic number increases by ______ while the ...
atomic number - Teacher Pages
... • Most of the elements are metals, which are found to the left of the zig-zag line in the periodic table. • Chemists classify an element as a metal based on physical properties such as: hardness, shininess, malleability, and ductility. – Malleable means to pound into shape. – Ductile means that the ...
... • Most of the elements are metals, which are found to the left of the zig-zag line in the periodic table. • Chemists classify an element as a metal based on physical properties such as: hardness, shininess, malleability, and ductility. – Malleable means to pound into shape. – Ductile means that the ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.