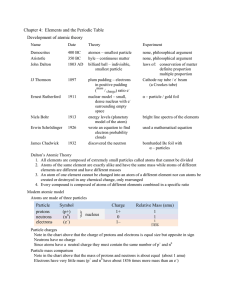
Chapter 4, Lesson 2: The Periodic Table
... or neutron). The majority of the atomic mass is contributed by the protons and neutrons. For any element in the periodic table, the number of electrons in an atom of that element always equals the number of protons in the nucleus. But this is not true for neutrons. Atoms of the same element can have ...
... or neutron). The majority of the atomic mass is contributed by the protons and neutrons. For any element in the periodic table, the number of electrons in an atom of that element always equals the number of protons in the nucleus. But this is not true for neutrons. Atoms of the same element can have ...
isotopes
... Ions An atom that carries an electrical charge is called an ion If the atom loses electrons, the atom becomes positively charged (because the number of positively charged protons will be more the number of electrons) If the atom gains electrons, the atom becomes negatively charged (because there are ...
... Ions An atom that carries an electrical charge is called an ion If the atom loses electrons, the atom becomes positively charged (because the number of positively charged protons will be more the number of electrons) If the atom gains electrons, the atom becomes negatively charged (because there are ...
Chapter 4: Elements and the Periodic Table Development of atomic
... patterns repeated (Li, Na, and K for example) so he started a new column to match properties He noticed two important things: A few element properties did not match exactly in order of atomic mass – he moved these He noticed three missing elements and predicted their properties All three of these el ...
... patterns repeated (Li, Na, and K for example) so he started a new column to match properties He noticed two important things: A few element properties did not match exactly in order of atomic mass – he moved these He noticed three missing elements and predicted their properties All three of these el ...
only that they did. democritus, an early greek philosopher, even had
... Being asked what animal you'd like to be is a trick question; you're already an animal. Doug Coupland ...
... Being asked what animal you'd like to be is a trick question; you're already an animal. Doug Coupland ...
Chemistry Basics Review
... of an element as well as its location on the Periodic Table. No two different elements will have the __________ atomic number. 3.The ______________________of an element is the average mass of an element ’s naturally occurring atom, or isotopes, taking into account the ______________________of each i ...
... of an element as well as its location on the Periodic Table. No two different elements will have the __________ atomic number. 3.The ______________________of an element is the average mass of an element ’s naturally occurring atom, or isotopes, taking into account the ______________________of each i ...
Electron Proton Neutron
... The mass number of an element is the sum of the number of protons and neutrons present in the atom of that element. For example, the atom of boron has 5 protons and 6 neutrons. So, the mass number of boron is 5 + 6 = 11. ...
... The mass number of an element is the sum of the number of protons and neutrons present in the atom of that element. For example, the atom of boron has 5 protons and 6 neutrons. So, the mass number of boron is 5 + 6 = 11. ...
2.1 Elements
... •An element is a pure substance that cannot be broken down into simpler substances by a chemical reaction. •Each element is identified by a one- or two-letter symbol. •Elements are arranged in the periodic table. •The position of an element in the periodic table tells us much about its chemical prop ...
... •An element is a pure substance that cannot be broken down into simpler substances by a chemical reaction. •Each element is identified by a one- or two-letter symbol. •Elements are arranged in the periodic table. •The position of an element in the periodic table tells us much about its chemical prop ...
8b Isotopes and Ions2
... What happens if the number of neutrons change??? If an atom gains neutrons… 1. The mass increases by 1 for every neutron added. 2. The number of protons and electrons stay the same. 3. The charge remains neutral. (Neutrons don’t have a charge.) 4. The identity of the atom does not change ...
... What happens if the number of neutrons change??? If an atom gains neutrons… 1. The mass increases by 1 for every neutron added. 2. The number of protons and electrons stay the same. 3. The charge remains neutral. (Neutrons don’t have a charge.) 4. The identity of the atom does not change ...
Periodic Table of Elements
... Write this Families of Nonmetals • The Halogen family is group 17. Each element has 7 valence electrons, therefore it commonly takes one electron away from group one metals to make compounds. ...
... Write this Families of Nonmetals • The Halogen family is group 17. Each element has 7 valence electrons, therefore it commonly takes one electron away from group one metals to make compounds. ...
Catalyst
... The mass number or atomic mass: This number tells the mass of one atom, which is approximately the sum of protons and neutrons in the nucleus, since each proton and each neutron has a mass equal to one mass unit, and the electrons ...
... The mass number or atomic mass: This number tells the mass of one atom, which is approximately the sum of protons and neutrons in the nucleus, since each proton and each neutron has a mass equal to one mass unit, and the electrons ...
Chapter 4:ааAtomic Structure Section 4.1анаDefining the Atom
... Dalton’s Atomic Theory ● John Dalton: English chemist and school teacher (17661844) ● by using experimental methods, Dalton transformed Democritus’s ideas on atoms into a scientific theory ○ studied the ratios in which elements combine in chemical reactions ● Dalton’s Atomic Theory ○ all ele ...
... Dalton’s Atomic Theory ● John Dalton: English chemist and school teacher (17661844) ● by using experimental methods, Dalton transformed Democritus’s ideas on atoms into a scientific theory ○ studied the ratios in which elements combine in chemical reactions ● Dalton’s Atomic Theory ○ all ele ...
PERIODIC PROPERTY: SIZE OF THE ATOM/ ATOMIC RADIUS
... Periodic Table is the address book of elements. Elements are arranged here in the increasing order of their atomic number. It keeps all the information about elements like size, nature, behaviour towards other element, strength and weakness. However these informations are hidden inside the periodic ...
... Periodic Table is the address book of elements. Elements are arranged here in the increasing order of their atomic number. It keeps all the information about elements like size, nature, behaviour towards other element, strength and weakness. However these informations are hidden inside the periodic ...
atomic mass
... 1. Explain how the periodic table of elements is arranged. 2. Elements at the left of the periodic table are known as ______. 3. Elements at the right of the periodic table are known as _______. 4. Explain some of the properties of metals. 5. Explain some of the properties of ...
... 1. Explain how the periodic table of elements is arranged. 2. Elements at the left of the periodic table are known as ______. 3. Elements at the right of the periodic table are known as _______. 4. Explain some of the properties of metals. 5. Explain some of the properties of ...
Atoms and their structure
... 2. 1st part FALSE b/c of discovery of ions and isotopes for atoms of the same element (Ex: Carbon12 vs. Carbon-14 and H+ vs. H); 2nd part TRUE 3. TRUE - A compound has to have the same ratio of elements to be considered that particular compound – Ex H2O or H2O2 two different ratios mean, two differe ...
... 2. 1st part FALSE b/c of discovery of ions and isotopes for atoms of the same element (Ex: Carbon12 vs. Carbon-14 and H+ vs. H); 2nd part TRUE 3. TRUE - A compound has to have the same ratio of elements to be considered that particular compound – Ex H2O or H2O2 two different ratios mean, two differe ...
Atoms
... •Isotopes are atoms of the same element that differ in the number of neutrons. Isotopes of the same element have the same chemical properties, because they have the same number of protons and electrons. Isotopes are identified by mass number. Neutrons affect mass, so, isotopes with more neutrons ar ...
... •Isotopes are atoms of the same element that differ in the number of neutrons. Isotopes of the same element have the same chemical properties, because they have the same number of protons and electrons. Isotopes are identified by mass number. Neutrons affect mass, so, isotopes with more neutrons ar ...
Chapter 4 Atomic Structure
... a) Cathode rays have identical properties regardless of the element used to produce them. All elements must contain identically charged electrons. b) Atoms are neutral, so there must be positive particles in the atom to balance the negative charge of the electrons c) Electrons have so little mass th ...
... a) Cathode rays have identical properties regardless of the element used to produce them. All elements must contain identically charged electrons. b) Atoms are neutral, so there must be positive particles in the atom to balance the negative charge of the electrons c) Electrons have so little mass th ...
DO NOW - PBworks
... Reminder: Enter the classroom, begin the Do Now immediately, silently and independently. ...
... Reminder: Enter the classroom, begin the Do Now immediately, silently and independently. ...
Week 9 CCA Test Review
... Why do elements with similar valence level of electrons have similar chemical properties? They will react the same way, because they ...
... Why do elements with similar valence level of electrons have similar chemical properties? They will react the same way, because they ...
Unit Nuclear Chemistry
... › Emission of subatomic particles or high-energy electromagnetic radiation by nuclei › Such atoms/isotopes said to be radioactive › All nuclides of elements beyond Bismuth (#83) in the periodic table are radioactive with only Polonium (84), Radon (86), Actinium ((89), Thorium (90), Uranium (92) and ...
... › Emission of subatomic particles or high-energy electromagnetic radiation by nuclei › Such atoms/isotopes said to be radioactive › All nuclides of elements beyond Bismuth (#83) in the periodic table are radioactive with only Polonium (84), Radon (86), Actinium ((89), Thorium (90), Uranium (92) and ...
atomic number
... b) Atoms are neutral, so there must be a positive substance in the atom to balance the negative charge of the electrons c) Electrons have so little mass that atoms must contain other particles that account for most of their mass ...
... b) Atoms are neutral, so there must be a positive substance in the atom to balance the negative charge of the electrons c) Electrons have so little mass that atoms must contain other particles that account for most of their mass ...
Ch. 3 - My CCSD
... b) Atoms are neutral, so there must be a positive substance in the atom to balance the negative charge of the electrons c) Electrons have so little mass that atoms must contain other particles that account for most of their mass ...
... b) Atoms are neutral, so there must be a positive substance in the atom to balance the negative charge of the electrons c) Electrons have so little mass that atoms must contain other particles that account for most of their mass ...
Atomic Structure
... • The mass of an electron is so small that we often approximate it to zero. • The size and the sign are both important for the relative charges. • The absolute values for mass and charge are extremely small. We use the relative values because they are more convenient. ...
... • The mass of an electron is so small that we often approximate it to zero. • The size and the sign are both important for the relative charges. • The absolute values for mass and charge are extremely small. We use the relative values because they are more convenient. ...
Notes 4.3 filled in
... C. Mass number (MN) of an atom ( in amu’s) MN is not indicated on the PT. To calculate the MN, simply add up #p+ and # no each having the mass of 1 amu. Q: an atom has 5 protons and 7 neutrons, calculate the MN. A: 5 amu + 7 amu = 12 amu Q: Which element is that? (Look in the PT) A: 5 protons, ...
... C. Mass number (MN) of an atom ( in amu’s) MN is not indicated on the PT. To calculate the MN, simply add up #p+ and # no each having the mass of 1 amu. Q: an atom has 5 protons and 7 neutrons, calculate the MN. A: 5 amu + 7 amu = 12 amu Q: Which element is that? (Look in the PT) A: 5 protons, ...
1 TEST DATE:
... fewer____________________________ than will other atoms of the same element. Atoms of the same element with different numbers of neutrons are called ___________________________ . Hydrogen has three isotopes. A hydrogen atom may contain zero, one, or two___________________________ . Every atom of car ...
... fewer____________________________ than will other atoms of the same element. Atoms of the same element with different numbers of neutrons are called ___________________________ . Hydrogen has three isotopes. A hydrogen atom may contain zero, one, or two___________________________ . Every atom of car ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.