Condensed Phase Ethanol Conversion to Higher Alcohols Tyler L
... The Peng-Robinson-Wong-Sandler (PRWS), predictive Soave-Redlich-Kwong (PSRK), and Schwartzentruber--Renon (SR-Polar) equations of state were chosen for initial model screening. These equations of state are known for accurate prediction of vapor pressures because they incorporate the acentric (ω) fac ...
... The Peng-Robinson-Wong-Sandler (PRWS), predictive Soave-Redlich-Kwong (PSRK), and Schwartzentruber--Renon (SR-Polar) equations of state were chosen for initial model screening. These equations of state are known for accurate prediction of vapor pressures because they incorporate the acentric (ω) fac ...
Test3_sp2012with answers
... _B__11. If H2O(s) → H2O(l) is endothermic and H2O(l) → H2O(g) is endothermic then H2O(g) → H2O(s) A) is endothermic B) is exothermic C) could be either exo or endothermic D) is equal to the sum of the first two reactions. _B__12. The temperature of a liquid is decreased. What happens to the vapor p ...
... _B__11. If H2O(s) → H2O(l) is endothermic and H2O(l) → H2O(g) is endothermic then H2O(g) → H2O(s) A) is endothermic B) is exothermic C) could be either exo or endothermic D) is equal to the sum of the first two reactions. _B__12. The temperature of a liquid is decreased. What happens to the vapor p ...
Solutions Foldable
... have defined freezing and boiling points. A solutions boiling and freezing point is determined by the concentration of the solution (remember a solution is NOT a pure substance)! Pure water freezes at 0°C but salt water will freeze at a lower temperature (the concentration of the salt water determin ...
... have defined freezing and boiling points. A solutions boiling and freezing point is determined by the concentration of the solution (remember a solution is NOT a pure substance)! Pure water freezes at 0°C but salt water will freeze at a lower temperature (the concentration of the salt water determin ...
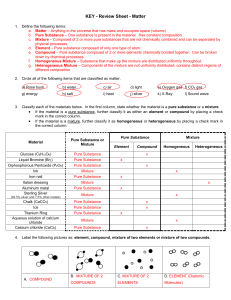
Matter Test Review Sheet
... a. Matter – Anything in the universe that has mass and occupies space (volume) b. Pure Substance – One substance is present in the material. Has constant composition. c. Mixture – Composed of 2 or more pure substances that are not chemically combined and can be separated by physical processes. d. El ...
... a. Matter – Anything in the universe that has mass and occupies space (volume) b. Pure Substance – One substance is present in the material. Has constant composition. c. Mixture – Composed of 2 or more pure substances that are not chemically combined and can be separated by physical processes. d. El ...
1) A clear glass bottle contains white sand, some nails, salt water
... (q) Green dye that separates into blue and yellow dye. homogeneous mixture (r) A green liquid that will separate into two distinct layers if left unattended for a period of time. homogeneous mixture (s) A liquid that boils away completely at 136°C. When the liquid is strongly heated in a closed con ...
... (q) Green dye that separates into blue and yellow dye. homogeneous mixture (r) A green liquid that will separate into two distinct layers if left unattended for a period of time. homogeneous mixture (s) A liquid that boils away completely at 136°C. When the liquid is strongly heated in a closed con ...
The Development of a Carbon Dioxide Fluoromethane Azeotropic
... The R-717/R-170 mixture forms positive azeotropes up to the critical region. With an ideal binary mixture, the bubble and dew-point lines are separate for the entire range of compositions, and only converge when the composition reaches 100% or 0% of the components. However, for the R717/R-170 blend ...
... The R-717/R-170 mixture forms positive azeotropes up to the critical region. With an ideal binary mixture, the bubble and dew-point lines are separate for the entire range of compositions, and only converge when the composition reaches 100% or 0% of the components. However, for the R717/R-170 blend ...
gas–liquid equilibrium prediction of system co2
... literature. The carbon dioxide and water binary system was also studied by Alain et al. [5], who reported new empirical observations for VLE data at a wide range of temperature (278.2–318.2 K) and pressure around 80 bar. These observations were consistent with the ones already present in the literat ...
... literature. The carbon dioxide and water binary system was also studied by Alain et al. [5], who reported new empirical observations for VLE data at a wide range of temperature (278.2–318.2 K) and pressure around 80 bar. These observations were consistent with the ones already present in the literat ...
Physical properties
... • Distillation is used to purify a compound by separating it from a non-volatile or less-volatile material. When different compounds in a mixture have different boiling points, they separate into individual components when the mixture is carefully distilled. • Distillation is the process of heating ...
... • Distillation is used to purify a compound by separating it from a non-volatile or less-volatile material. When different compounds in a mixture have different boiling points, they separate into individual components when the mixture is carefully distilled. • Distillation is the process of heating ...
Classification of
... e) ______heterogeneous_________________ - mixture with individual parts visible f) _______states of matter_______________________ - solid, liquid, gas g) ___________solid___________________ - definite volume and shape h) ____________liquid__________________ - definite volume, changeable shape i) ___ ...
... e) ______heterogeneous_________________ - mixture with individual parts visible f) _______states of matter_______________________ - solid, liquid, gas g) ___________solid___________________ - definite volume and shape h) ____________liquid__________________ - definite volume, changeable shape i) ___ ...
Properties and Changes of Matter
... all parts of the mixture have the same concentration of matter. Different parts of the mixture can not be identified (transparent appearance for solutions) Alloys: solution of metal also called a solution ...
... all parts of the mixture have the same concentration of matter. Different parts of the mixture can not be identified (transparent appearance for solutions) Alloys: solution of metal also called a solution ...
CHAPTER 1 CHEMICAL FOUNDATIONS 1 CHAPTER ONE
... a. Distillation separates components of a mixture, so the orange liquid is a mixture (has an average color of the yellow liquid and the red solid). Distillation utilizes boiling point differences to separate out the components of a mixture. Distillation is a physical change because the components of ...
... a. Distillation separates components of a mixture, so the orange liquid is a mixture (has an average color of the yellow liquid and the red solid). Distillation utilizes boiling point differences to separate out the components of a mixture. Distillation is a physical change because the components of ...
Mixtures
... solute is dissolved in water. Several factors affect how fast the solid will dissolve. You can see why you will enjoy a glass of lemonade sooner if you stir granulated sugar into the lemonade before adding ice! ...
... solute is dissolved in water. Several factors affect how fast the solid will dissolve. You can see why you will enjoy a glass of lemonade sooner if you stir granulated sugar into the lemonade before adding ice! ...
Separation of a Mixture
... Divide class into 4 groups. Set-up 4 stations around the classroom/laboratory with all the aforementioned materials. Ask students to brainstorm in their Journals: o What are some of the physical properties of the components of the mixture? o How can the differences in physical properties be us ...
... Divide class into 4 groups. Set-up 4 stations around the classroom/laboratory with all the aforementioned materials. Ask students to brainstorm in their Journals: o What are some of the physical properties of the components of the mixture? o How can the differences in physical properties be us ...
chem – mixtures elements compounds for ib 1 10-10
... I can write and recognize a chemical symbol for an element using the periodic table. I can give examples of chemical compounds. I can explain that elements are the basic building blocks of matter and discuss how they can ...
... I can write and recognize a chemical symbol for an element using the periodic table. I can give examples of chemical compounds. I can explain that elements are the basic building blocks of matter and discuss how they can ...
Metathesis Problems (and Some Solutions) Identified Through
... where S is solubility (mass of solute per unit volume of solvent), kH is Henry’s law constant (for gas and liquid at a given temperature), P is partial pressure of the gas ...
... where S is solubility (mass of solute per unit volume of solvent), kH is Henry’s law constant (for gas and liquid at a given temperature), P is partial pressure of the gas ...
SampleTest3withAnswers
... D) more than one response is correct _D___11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation _C___12. The vapor pressure of a liquid A) decreases with increasing temperature B) is independent of temperature C) is equal to one at ...
... D) more than one response is correct _D___11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation _C___12. The vapor pressure of a liquid A) decreases with increasing temperature B) is independent of temperature C) is equal to one at ...
File
... solvent because the solution has a lower vapor pressure than the water (Raoult’s Law) . the temperature of the solution has be higher to produce enough vapor pressure to equal the atmospheric pressure (i.e., boiling) (ii) the amount of boiling point elevation depends on the number of non-volatile pa ...
... solvent because the solution has a lower vapor pressure than the water (Raoult’s Law) . the temperature of the solution has be higher to produce enough vapor pressure to equal the atmospheric pressure (i.e., boiling) (ii) the amount of boiling point elevation depends on the number of non-volatile pa ...
Separation and Purification Methods
... The effect of temperature on the volatility of compounds is well-known, but the impact of reduced pressure is much less appreciated. However, distillation at reduced pressure, or vacuum distillation, brings many advantages. For example, consider a liquid that has a boiling point of 180◦ C at atmosph ...
... The effect of temperature on the volatility of compounds is well-known, but the impact of reduced pressure is much less appreciated. However, distillation at reduced pressure, or vacuum distillation, brings many advantages. For example, consider a liquid that has a boiling point of 180◦ C at atmosph ...
Intro to Chem
... ◦ Physical methods that are used to separate mixtures cannot be used to break a compound into simpler substances. ◦ Chemical change is a change that produces matter with a different composition than the orginal matter. Sugar broken down into C and H2O(g) when heated. Broken down into H2 and O2 b ...
... ◦ Physical methods that are used to separate mixtures cannot be used to break a compound into simpler substances. ◦ Chemical change is a change that produces matter with a different composition than the orginal matter. Sugar broken down into C and H2O(g) when heated. Broken down into H2 and O2 b ...
Are You suprised ?
... A system at equilibrium is described by the equation: Heat + SO2Cl2(l ) ⇌ SO2(g) + Cl2(g) One of the following sentences is correct. A) Adding Cl2 will increase heat. B) The equilibrium will move to the left when we remove Cl2. C) Increasing the pressure has no effect on this system. D) Adding catal ...
... A system at equilibrium is described by the equation: Heat + SO2Cl2(l ) ⇌ SO2(g) + Cl2(g) One of the following sentences is correct. A) Adding Cl2 will increase heat. B) The equilibrium will move to the left when we remove Cl2. C) Increasing the pressure has no effect on this system. D) Adding catal ...
Chem A Week 2 Matter Notes
... chromatography. It is the physical separation of a mixture into its individual components. It involves using a solvent to pass through the mixture. What solvent should I use? The solvent used depends upon the solubility of the mixture you are trying to separate. Examples are water, isopropyl alcohol ...
... chromatography. It is the physical separation of a mixture into its individual components. It involves using a solvent to pass through the mixture. What solvent should I use? The solvent used depends upon the solubility of the mixture you are trying to separate. Examples are water, isopropyl alcohol ...
SampleTest3
... D) more than one response is correct ____11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation ____12. The vapor pressure of a liquid A) decreases with increasing temperature B) is independent of temperature C) is equal to one atmo ...
... D) more than one response is correct ____11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation ____12. The vapor pressure of a liquid A) decreases with increasing temperature B) is independent of temperature C) is equal to one atmo ...
CHEMISTRY 313 PHYSICAL CHEMISTRY I Additional Problems for
... with its vapor in a closed vessel. (a) How many phases and components are present. (b) What is the variance of the system? Identify the independent variables. III.10. Suppose that the solution referred to in Problem III.9 is not saturated. (a) How many phases and components are present. (b) What is ...
... with its vapor in a closed vessel. (a) How many phases and components are present. (b) What is the variance of the system? Identify the independent variables. III.10. Suppose that the solution referred to in Problem III.9 is not saturated. (a) How many phases and components are present. (b) What is ...
Mixture Solution Notes
... 1. What does “dissolve” mean? 2. What kinds of things dissolve? 3. What do things dissolve in? ...
... 1. What does “dissolve” mean? 2. What kinds of things dissolve? 3. What do things dissolve in? ...
Azeotrope

An azeotrope or a constant boiling mixture is a mixture of two or more liquids whose proportions cannot be altered by simple distillation. This happens because, when an azeotrope is boiled, the vapour has the same proportions of constituents as the unboiled mixture.Because their composition is unchanged by distillation, azeotropes are also called (especially in older texts) constant boiling mixtures. The word azeotrope is derived from the Greek words ζέειν (boil) and τρόπος (turning) combined with the prefix α- (no) to give the overall meaning, ""no change on boiling"". The term ""azeotrope"" was coined in 1911 by John Wade and Richard William Merriman.Many azeotropic mixtures of pairs of compounds are known, and many azeotropes of three or more compounds are also known. In such a case it is not possible to separate the components by fractional distillation. There are two types of azeotropes: minimum boiling azeotrope and maximum boiling azeotrope. A solution that shows greater positive deviation from Raoult's law forms a minimum boiling azeotrope at a specific composition. For example, an ethanol-water mixture (obtained by fermentation of sugars) on fractional distillation yields a solution containing approximately 95% by volume of ethanol. Once this composition has been achieved, the liquid and vapour have the same composition, and no further separation occurs. A solution that shows large negative deviation from Raoult's law forms a maximum boiling azeotrope at a specific composition. Nitric acid and water is an example of this class of azeotrope. This azeotrope has an approximate composition of 68% nitric acid and 32% water by mass, with a boiling point of 393.5 K.