ppt-nuclear - SandersScienceStuff
... Fission • Fission means to break apart. Nuclear fission occurs when a nucleus splits apart into different fragments. • This generally occurs with atoms that have a mass number heavier than 60. • The nuclei do not always split the same way. Scientists have found 200 different products from the fissi ...
... Fission • Fission means to break apart. Nuclear fission occurs when a nucleus splits apart into different fragments. • This generally occurs with atoms that have a mass number heavier than 60. • The nuclei do not always split the same way. Scientists have found 200 different products from the fissi ...
Nuclear chemistry – the study of nuclear reactions and their uses in
... All spontaneous nuclear reactions are exothermic. The masses of nuclei are always less than the masses of the individual nucleons of which they are composed. i. Helium-4 (2protons, 2 neutrons, 2 electrons) has a mass of 4.00150 amu ii. The mass of 2 protons and 2 neutrons is 4.03188 amu. 1. The mass ...
... All spontaneous nuclear reactions are exothermic. The masses of nuclei are always less than the masses of the individual nucleons of which they are composed. i. Helium-4 (2protons, 2 neutrons, 2 electrons) has a mass of 4.00150 amu ii. The mass of 2 protons and 2 neutrons is 4.03188 amu. 1. The mass ...
Radioactivity
... Isotopes of an element contain the same number of protons and the same number of electrons. So isotopes have the same chemical properties chemical reactions involve the electrons in an atom. However they have different physical properties because their mass is different. Some isotopes exist naturall ...
... Isotopes of an element contain the same number of protons and the same number of electrons. So isotopes have the same chemical properties chemical reactions involve the electrons in an atom. However they have different physical properties because their mass is different. Some isotopes exist naturall ...
Nuclear Chemistry
... Go down two on periodic table Atomic number decreases by 2 Mass number decreases by 4 Beta: Go up one on periodic table Atomic number increases by 1 Mass number stays the same ...
... Go down two on periodic table Atomic number decreases by 2 Mass number decreases by 4 Beta: Go up one on periodic table Atomic number increases by 1 Mass number stays the same ...
6.2 Atomic Nucleus Stability and Isotopes
... Neutrons and protons are particles called baryons. A baryon is made up of 3 particles called quarks. There are six kinds of quarks, each having a fractional charge in relation to the charge of a proton. The most common baryons are neutrons and protons. Protons are made up of two up and one down quar ...
... Neutrons and protons are particles called baryons. A baryon is made up of 3 particles called quarks. There are six kinds of quarks, each having a fractional charge in relation to the charge of a proton. The most common baryons are neutrons and protons. Protons are made up of two up and one down quar ...
Objective 2 Average Atomic Mass
... The discovery of the (11) ___________________ in 1895 by Wilhelm Roentgen opened a whole new field of research. Among those who worked in this new field were Pierre and Marie Curie. The Curies discovered that some forms of matter give off (12) ________________, a combination of particles and energy. ...
... The discovery of the (11) ___________________ in 1895 by Wilhelm Roentgen opened a whole new field of research. Among those who worked in this new field were Pierre and Marie Curie. The Curies discovered that some forms of matter give off (12) ________________, a combination of particles and energy. ...
Remediation_unit 2_standard
... The discovery of the (11) ___________________ in 1895 by Wilhelm Roentgen opened a whole new field of research. Among those who worked in this new field were Pierre and Marie Curie. The Curies discovered that some forms of matter give off (12) ________________, a combination of particles and energy. ...
... The discovery of the (11) ___________________ in 1895 by Wilhelm Roentgen opened a whole new field of research. Among those who worked in this new field were Pierre and Marie Curie. The Curies discovered that some forms of matter give off (12) ________________, a combination of particles and energy. ...
NUCLEAR CHEMISTRY
... • Radioactive Isotopes: unstable atoms, due to a nucleus with too many or too few neutrons relative to the number of protons. • No amount of neutrons can hold a nucleus together once it has more that 82 protons. All of the elements with an atomic number greater than 82 have only unstable isotopes. • ...
... • Radioactive Isotopes: unstable atoms, due to a nucleus with too many or too few neutrons relative to the number of protons. • No amount of neutrons can hold a nucleus together once it has more that 82 protons. All of the elements with an atomic number greater than 82 have only unstable isotopes. • ...
21J 2011 The Polywell Nuclear Reactor Website July 4, 2011
... with a large sample of pitchblende, they employed a series of chemical separation techniques, always discarding the separated fraction, which did not emit the disproportionately high radiation. Eventually, they isolated a new radioactive element, which they called polonium in honor of Marie's home c ...
... with a large sample of pitchblende, they employed a series of chemical separation techniques, always discarding the separated fraction, which did not emit the disproportionately high radiation. Eventually, they isolated a new radioactive element, which they called polonium in honor of Marie's home c ...
Document
... B) of perspective and parallax. C) of energy dissipation. D) the electric charge decreases. E) the electric charge increases. 10) The half life of carbon 14 is 5730 years. If a 1-gram sample of old carbon is 1/8 as radioactive as 1-gram of a current sample, then the age of the old sample is about A) ...
... B) of perspective and parallax. C) of energy dissipation. D) the electric charge decreases. E) the electric charge increases. 10) The half life of carbon 14 is 5730 years. If a 1-gram sample of old carbon is 1/8 as radioactive as 1-gram of a current sample, then the age of the old sample is about A) ...
4550-15Lecture29 - Cornell Geological Sciences
... • Cosmic rays are high energy nuclei (mainly of H and He) from space. When they collide with nuclei in the atmosphere or the surface of the Earth, they induce nuclear reactions. The resulting particles also have high energies and can induce further reactions. The one of greatest interest is 14N(n,p) ...
... • Cosmic rays are high energy nuclei (mainly of H and He) from space. When they collide with nuclei in the atmosphere or the surface of the Earth, they induce nuclear reactions. The resulting particles also have high energies and can induce further reactions. The one of greatest interest is 14N(n,p) ...
Lecture 33 - Cornell Geological Sciences
... • Cosmic rays are high energy nuclei (mainly of H and He) from space. When they collide with nuclei in the atmosphere or the surface of the Earth, they induce nuclear reactions. The resulting particles also have high energies and can induce further reactions. The one of greatest interest is 14N(n,p) ...
... • Cosmic rays are high energy nuclei (mainly of H and He) from space. When they collide with nuclei in the atmosphere or the surface of the Earth, they induce nuclear reactions. The resulting particles also have high energies and can induce further reactions. The one of greatest interest is 14N(n,p) ...
Atomic Nuclei - RAJEEV Classes
... number increases by 1 but mass number remains same. c)The emission of a γ-particle does not change the mass number or the atomic number of the radioactive nucleus. The γ-particle emission by a radioactive nucleus lowers its energy state. ...
... number increases by 1 but mass number remains same. c)The emission of a γ-particle does not change the mass number or the atomic number of the radioactive nucleus. The γ-particle emission by a radioactive nucleus lowers its energy state. ...
Scientists` Consensus Ideas Atomic Structure and Nuclear Interactions
... Nuclear Interactions 13. Interactions involving the particles of a nucleus (protons and neutrons) are called nuclear reactions or nuclear interactions. Nuclear reactions release enormous quantities of energy compared to chemical reactions. 14. Some elements change into other elements as a result of ...
... Nuclear Interactions 13. Interactions involving the particles of a nucleus (protons and neutrons) are called nuclear reactions or nuclear interactions. Nuclear reactions release enormous quantities of energy compared to chemical reactions. 14. Some elements change into other elements as a result of ...
Radioactive decay of nucleus
... Weak nuclear force – involves the transformation of a neutron into a proton and electron. It is less powerful than strong nuclear force. 3. Beta-positive (β+) decay: nuclear decay involving emission of a positron. Positron is an antimatter to the electron. It has exactly same properties except oppos ...
... Weak nuclear force – involves the transformation of a neutron into a proton and electron. It is less powerful than strong nuclear force. 3. Beta-positive (β+) decay: nuclear decay involving emission of a positron. Positron is an antimatter to the electron. It has exactly same properties except oppos ...
Geologic Dating! - rgreenbergscience
... • The principle of Fossil succession states that fossil organisms succeed one another in a definite and determinable order. Therefore, any time period can be recognized by its fossil content. • Index Fossils – fossils of organisms that lived during a relatively short, well-defined time period. – Amm ...
... • The principle of Fossil succession states that fossil organisms succeed one another in a definite and determinable order. Therefore, any time period can be recognized by its fossil content. • Index Fossils – fossils of organisms that lived during a relatively short, well-defined time period. – Amm ...
Section 19.1 Radioactivity
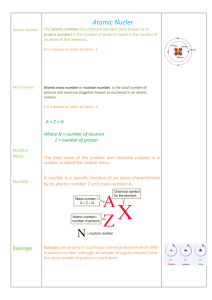
... Radioactivity A Review of Atomic Terms • nucleons – particles found in the nucleus of an atom – neutrons – protons • atomic number (Z) – number of protons in the nucleus • mass number (A) – sum of the number of protons and neutrons • isotopes – atoms with identical atomic numbers but different mass ...
... Radioactivity A Review of Atomic Terms • nucleons – particles found in the nucleus of an atom – neutrons – protons • atomic number (Z) – number of protons in the nucleus • mass number (A) – sum of the number of protons and neutrons • isotopes – atoms with identical atomic numbers but different mass ...
Glossary of Technical Terms - Institute for Energy and Environmental
... Science for Democratic Action - http://ieer.org/wp protons in each nucleus. Theexact atomic weight differs fractionally from that whole number, because neutrons are slightly heavier than protons and the mass of the nucleus is also affected by the binding energy. atomic number (symbolized Z): The num ...
... Science for Democratic Action - http://ieer.org/wp protons in each nucleus. Theexact atomic weight differs fractionally from that whole number, because neutrons are slightly heavier than protons and the mass of the nucleus is also affected by the binding energy. atomic number (symbolized Z): The num ...
Chapter 7 Worksheet
... Describe the process of the induced uranium-235 nuclear fission reaction that occurs in both fission-style nuclear weapons and in Canadian nuclear power plants. Be sure to include the production of the unstable uranium-236 isotope in your explanation, as well as all the reactants and products of the ...
... Describe the process of the induced uranium-235 nuclear fission reaction that occurs in both fission-style nuclear weapons and in Canadian nuclear power plants. Be sure to include the production of the unstable uranium-236 isotope in your explanation, as well as all the reactants and products of the ...
Chemistry: Nuclear Reactions Guided Inquiry + n → + + 3 n +
... Nuclear reactions are reactions that affect the nucleus of an atom. In nature, unstable nuclei undergo nuclear reactions to form more stable nuclei. Stable nuclei can also undergo nuclear reactions if ...
... Nuclear reactions are reactions that affect the nucleus of an atom. In nature, unstable nuclei undergo nuclear reactions to form more stable nuclei. Stable nuclei can also undergo nuclear reactions if ...
Nuclear Chemistry PowerPoint presentation
... Energy may be supplied by accelerating these particles in the magnetic or electric field of a particle accelerator. Radioactive isotopes of all the natural elements have been produced. ...
... Energy may be supplied by accelerating these particles in the magnetic or electric field of a particle accelerator. Radioactive isotopes of all the natural elements have been produced. ...
Radioactivity presentation script
... range of the residual strong force means that nucleons are attracted only to their nearest neighbors. But the protons are repelled by all of the other protons in the nucleus. This explains why larger nuclei need more neutrons than protons - they spread out the protons further and provide extra attra ...
... range of the residual strong force means that nucleons are attracted only to their nearest neighbors. But the protons are repelled by all of the other protons in the nucleus. This explains why larger nuclei need more neutrons than protons - they spread out the protons further and provide extra attra ...
Radioactive decay
Radioactive decay, also known as nuclear decay or radioactivity, is the process by which a nucleus of an unstable atom loses energy by emitting radiation. A material that spontaneously emits such radiation — which includes alpha particles, beta particles, gamma rays and conversion electrons — is considered radioactive.Radioactive decay is a stochastic (i.e. random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. The chance that a given atom will decay never changes, that is, it does not matter how long the atom has existed. For a large collection of atoms however, the decay rate for that collection can be calculated from their measured decay constants or half-lives. This is the basis of radiometric dating. The half-lives of radioactive atoms have no known limits for shortness or length of duration, and range over 55 orders of magnitude in time.There are many types of radioactive decay (see table below). A decay, or loss of energy from the nucleus, results when an atom with one type of nucleus, called the parent radionuclide (or parent radioisotope), transforms into an atom with a nucleus in a different state, or with a nucleus containing a different number of protons and neutrons. The product is called the daughter nuclide. In some decays, the parent and the daughter nuclides are different chemical elements, and thus the decay process results in the creation of an atom of a different element. This is known as a nuclear transmutation.The first decay processes to be discovered were alpha decay, beta decay, and gamma decay. Alpha decay occurs when the nucleus ejects an alpha particle (helium nucleus). This is the most common process of emitting nucleons, but in rarer types of decays, nuclei can eject protons, or in the case of cluster decay specific nuclei of other elements. Beta decay occurs when the nucleus emits an electron or positron and a neutrino, in a process that changes a proton to a neutron or the other way about. The nucleus may capture an orbiting electron, causing a proton to convert into a neutron in a process called electron capture. All of these processes result in a well-defined nuclear transmutation.By contrast, there are radioactive decay processes that do not result in a nuclear transmutation. The energy of an excited nucleus may be emitted as a gamma ray in a process called gamma decay, or be used to eject an orbital electron by its interaction with the excited nucleus, in a process called internal conversion. Highly excited neutron-rich nuclei, formed as the product of other types of decay, occasionally lose energy by way of neutron emission, resulting in a change of an element from one isotope to another. Another type of radioactive decay results in products that are not defined, but appear in a range of ""pieces"" of the original nucleus. This decay, called spontaneous fission, happens when a large unstable nucleus spontaneously splits into two (and occasionally three) smaller daughter nuclei, and generally leads to the emission of gamma rays, neutrons, or other particles from those products.For a summary table showing the number of stable and radioactive nuclides in each category, see radionuclide. There exist twenty-nine chemical elements on Earth that are radioactive. They are those that contain thirty-four radionuclides that date before the time of formation of the solar system, and are known as primordial nuclides. Well-known examples are uranium and thorium, but also included are naturally occurring long-lived radioisotopes such as potassium-40. Another fifty or so shorter-lived radionuclides, such as radium and radon, found on Earth, are the products of decay chains that began with the primordial nuclides, and ongoing cosmogenic processes, such as the production of carbon-14 from nitrogen-14 by cosmic rays. Radionuclides may also be produced artificially in particle accelerators or nuclear reactors, resulting in 650 of these with half-lives of over an hour, and several thousand more with even shorter half-lives. See this list of nuclides for a list of these, sorted by half life.