2.10 Basic Nuclear Chemistry
... III. Marie and Pierre Curie (1896) A. The Curie’s were a husband and wife team of French Physicists. B. Marie Curie was the first woman to win a Nobel in Prize in 1903 for her work on radioactivity. She later won a second Nobel Prize in 1911. She is the only female to win 2 Nobel Prizes. That is awe ...
... III. Marie and Pierre Curie (1896) A. The Curie’s were a husband and wife team of French Physicists. B. Marie Curie was the first woman to win a Nobel in Prize in 1903 for her work on radioactivity. She later won a second Nobel Prize in 1911. She is the only female to win 2 Nobel Prizes. That is awe ...
2013 Q9 - Loreto Balbriggan
... Iodine–131 decays with the emission of a beta-particle and has a half-life of 8 days. Write an equation for the beta-decay of iodine–131. Estimate the fraction of the iodine–131 that remained after 40 days. (15) Caesium–137 has a half-life of 30 years and it remains a significant contaminant in the ...
... Iodine–131 decays with the emission of a beta-particle and has a half-life of 8 days. Write an equation for the beta-decay of iodine–131. Estimate the fraction of the iodine–131 that remained after 40 days. (15) Caesium–137 has a half-life of 30 years and it remains a significant contaminant in the ...
Chapter 14 section 2
... energy. An alpha particle is a particle that is made up of two protons and two neutrons. The energy and alpha particle that are ejected are called nuclear radiation. In a smoke detector, the alpha particles make it possible for the air to conduct an electric current. As long as the electric current ...
... energy. An alpha particle is a particle that is made up of two protons and two neutrons. The energy and alpha particle that are ejected are called nuclear radiation. In a smoke detector, the alpha particles make it possible for the air to conduct an electric current. As long as the electric current ...
Radiation Questions March 4th
... Draw a graph of count rate against time. The first two points have been plotted for you. ...
... Draw a graph of count rate against time. The first two points have been plotted for you. ...
Name
... particles in the air. 2. Nuclear radiation is used to detect diseases a. A radioactive tracer is a radioactive material that is added to a substance so that its distribution can be detected later. b. Radioactive tracers are widely used in medicine. 3. Nuclear radiation therapy is used to treat cance ...
... particles in the air. 2. Nuclear radiation is used to detect diseases a. A radioactive tracer is a radioactive material that is added to a substance so that its distribution can be detected later. b. Radioactive tracers are widely used in medicine. 3. Nuclear radiation therapy is used to treat cance ...
Chapter 16 Notes - Mr. Julien`s Homepage
... 1. Elements with atomic number 20 or higher usually have one or more isotopes that have unstable nuclei. a. An unstable nucleus is radioactive which means that it spontaneously emits small particles of energy. b. Radiation— 2. Radiation can take the form of alpha () and beta () particles, positron ...
... 1. Elements with atomic number 20 or higher usually have one or more isotopes that have unstable nuclei. a. An unstable nucleus is radioactive which means that it spontaneously emits small particles of energy. b. Radiation— 2. Radiation can take the form of alpha () and beta () particles, positron ...
strong force
... nucleus plus the masses of Z electrons The atomic masses of different isotopes are different The periodic table contains an average value of the atomic mass for each element based on the natural abundance of each isotope The value listed in the periodic table is the mass in grams of 1 mole [Avogadro ...
... nucleus plus the masses of Z electrons The atomic masses of different isotopes are different The periodic table contains an average value of the atomic mass for each element based on the natural abundance of each isotope The value listed in the periodic table is the mass in grams of 1 mole [Avogadro ...
catch some rays: alpha, beta, gamma (modified for adeed)
... an electron, and a neutrino or a proton decays into a neutron, a positron, and a neutrino. Gamma decay happens when electromagnetic radiation (a photon) is given off by the nucleus as the protons and neutrons lower in energy state. In alpha decay, the nucleus emits an alpha particle, a helium nucleu ...
... an electron, and a neutrino or a proton decays into a neutron, a positron, and a neutrino. Gamma decay happens when electromagnetic radiation (a photon) is given off by the nucleus as the protons and neutrons lower in energy state. In alpha decay, the nucleus emits an alpha particle, a helium nucleu ...
Chapter 7 - Bakersfield College
... C. Radioactive decay occurs when a nucleus emits particles or high frequency em waves. D. There are five kinds of radioactive decay. 1. Alpha decay, in which an alpha particle (He) is emitted from a large, unstable nucleus. 2. Gamma decay, in which very high frequency em waves, or gamma rays, are em ...
... C. Radioactive decay occurs when a nucleus emits particles or high frequency em waves. D. There are five kinds of radioactive decay. 1. Alpha decay, in which an alpha particle (He) is emitted from a large, unstable nucleus. 2. Gamma decay, in which very high frequency em waves, or gamma rays, are em ...
atoms - Groupfusion.net
... Normally, this is the same for every atom of an element, therefore number of protons identifies the element. In a “normal”, neutral atom, number of protons = number of electrons Mass Number = the number of protons + number of neutrons = A Number of neutrons in an atom of an element can vary. These a ...
... Normally, this is the same for every atom of an element, therefore number of protons identifies the element. In a “normal”, neutral atom, number of protons = number of electrons Mass Number = the number of protons + number of neutrons = A Number of neutrons in an atom of an element can vary. These a ...
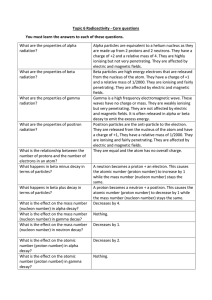
Topic 6 Radioactivity Core Questions
... The mass number (nucleon number) before with the total mass numbers (nucleon numbers) of the new isotope and released particles after and the atomic number (proton number) before with the total atomic numbers (proton numbers) of the new isotope and released particles after. When is gamma radiation e ...
... The mass number (nucleon number) before with the total mass numbers (nucleon numbers) of the new isotope and released particles after and the atomic number (proton number) before with the total atomic numbers (proton numbers) of the new isotope and released particles after. When is gamma radiation e ...
Document
... 1.35% is by beta emission, and 0.49% is by positron emission. (a) Why should we expect 40K to be radioactive? (b) Write the nuclear equations for the three modes by which 40K decays. (c) How many 40K+ ions are present in 1.00 g of KCl? (d) How long does it take for 1.00% of the 40K in a sample to un ...
... 1.35% is by beta emission, and 0.49% is by positron emission. (a) Why should we expect 40K to be radioactive? (b) Write the nuclear equations for the three modes by which 40K decays. (c) How many 40K+ ions are present in 1.00 g of KCl? (d) How long does it take for 1.00% of the 40K in a sample to un ...
Nuclear Chemistry
... radiation by an unstable nucleus is called radioactivity. • Marie & Pierre Curie further investigated Becquerel’s observations and determined that for the first time, reactions involving a change in the nucleus were being correctly observed. • All scientists were awarded the Nobel Prize for Physics ...
... radiation by an unstable nucleus is called radioactivity. • Marie & Pierre Curie further investigated Becquerel’s observations and determined that for the first time, reactions involving a change in the nucleus were being correctly observed. • All scientists were awarded the Nobel Prize for Physics ...
Chapter 21 Nuclear Chemistry - Ocean County Vocational
... • Since only 1 electron is emitted, the mass number will stay the same but the atomic number will increase by one. This is called ...
... • Since only 1 electron is emitted, the mass number will stay the same but the atomic number will increase by one. This is called ...
Grade 10S Physics T3W5 material
... 5‐ By measuring in the rocks the present rate of radioactivity or the concentration of the non radioactive products of radioactivity, the age of the rock can be determined. 6‐ a. Lower doses may cause Leukemia within 2years or other forms of cancer 15 to 20 years. b. Radiation can cause genetic a ...
... 5‐ By measuring in the rocks the present rate of radioactivity or the concentration of the non radioactive products of radioactivity, the age of the rock can be determined. 6‐ a. Lower doses may cause Leukemia within 2years or other forms of cancer 15 to 20 years. b. Radiation can cause genetic a ...
on Nuclear Physics - Good Earth School
... Radioactive decay (also known as nuclear decay or radioactivity) is the process by which the nucleus of an unstable atom loses energy by emitting radiations ...
... Radioactive decay (also known as nuclear decay or radioactivity) is the process by which the nucleus of an unstable atom loses energy by emitting radiations ...
Slide 1
... and the mass of a n0 is 1.008665 u. There are 931.50 MeV/u. Al is atomic # 13, so it has 13(1.007825 u) of p+ and e-, = 13.101725 u. Al has 14 n0 = 14(1.008665 u) = 14.12131 u Dm = (13.101725 + 14.12131) u – 26.981534 u = .241501 u x 931.50 MeV/u = 225 MeV ...
... and the mass of a n0 is 1.008665 u. There are 931.50 MeV/u. Al is atomic # 13, so it has 13(1.007825 u) of p+ and e-, = 13.101725 u. Al has 14 n0 = 14(1.008665 u) = 14.12131 u Dm = (13.101725 + 14.12131) u – 26.981534 u = .241501 u x 931.50 MeV/u = 225 MeV ...
NUCLEAR CHEMISTRY
... Energy may be supplied by accelerating these particles in the magnetic or electric field of a particle accelerator. ...
... Energy may be supplied by accelerating these particles in the magnetic or electric field of a particle accelerator. ...
Nuclear Chemistry Radioactivity
... Nuclear Fission and Nuclear Fusion • Nuclear fusion is a nuclear reaction in which a light nuclei combine to give a stabler heavy nucleus plus possibly several neutrons, and energy is released. – At these elevated temperature, a plasma resuts, that is, an electrically neutral gas of ions and electr ...
... Nuclear Fission and Nuclear Fusion • Nuclear fusion is a nuclear reaction in which a light nuclei combine to give a stabler heavy nucleus plus possibly several neutrons, and energy is released. – At these elevated temperature, a plasma resuts, that is, an electrically neutral gas of ions and electr ...
NUCLEAR CHEMISTRY
... Most naturally occurring isotopes of elements up to atomic number 19 have stable nuclei. Elements with higher atomic number (20-83) consist of a mixture isotopes, some of which may have unstable nuclei. When the nucleus of an isotope is unstable, it is radioactive, which means that it will spontaneo ...
... Most naturally occurring isotopes of elements up to atomic number 19 have stable nuclei. Elements with higher atomic number (20-83) consist of a mixture isotopes, some of which may have unstable nuclei. When the nucleus of an isotope is unstable, it is radioactive, which means that it will spontaneo ...
NUCLEAR CHEMISTRY
... the plate was developed, the image of the rock was found on the plate. Based on further experiments, he concluded that the plate had been exposed by rays given off by the uranium. ...
... the plate was developed, the image of the rock was found on the plate. Based on further experiments, he concluded that the plate had been exposed by rays given off by the uranium. ...
Alpha
... In some types of atom, the nucleus is unstable, and will decay into a more stable atom. This radioactive decay is completely spontaneous. It's not the same as what happens in a nuclear power station (where neutrons whizz around and hit uranium nuclei, causing them to split). ...
... In some types of atom, the nucleus is unstable, and will decay into a more stable atom. This radioactive decay is completely spontaneous. It's not the same as what happens in a nuclear power station (where neutrons whizz around and hit uranium nuclei, causing them to split). ...
NUCLEAR CHEMISTRY REVIEW SHEET
... a. It increases by four b. It decreases by one c. It decreases by four d. It remains the same _____ 10. When an atom undergoes radioactive decay by emitting an alpha particle, what change occurs to the atomic number of that atom? a. It increases by one b. It decreases by one c. It increases by two d ...
... a. It increases by four b. It decreases by one c. It decreases by four d. It remains the same _____ 10. When an atom undergoes radioactive decay by emitting an alpha particle, what change occurs to the atomic number of that atom? a. It increases by one b. It decreases by one c. It increases by two d ...
Chapter 28
... of a large nucleus into smaller fragments. They break apart to form smaller elements. U-235 combined with a neutron to split into Ba-144 and Kr-89 + 3 neutrons. Those neutrons can then react with other U-235 atoms, causing a “chain reaction.” Note the 235 + 1 = 89 + 144 + 3 ...
... of a large nucleus into smaller fragments. They break apart to form smaller elements. U-235 combined with a neutron to split into Ba-144 and Kr-89 + 3 neutrons. Those neutrons can then react with other U-235 atoms, causing a “chain reaction.” Note the 235 + 1 = 89 + 144 + 3 ...
Radioactive decay
Radioactive decay, also known as nuclear decay or radioactivity, is the process by which a nucleus of an unstable atom loses energy by emitting radiation. A material that spontaneously emits such radiation — which includes alpha particles, beta particles, gamma rays and conversion electrons — is considered radioactive.Radioactive decay is a stochastic (i.e. random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. The chance that a given atom will decay never changes, that is, it does not matter how long the atom has existed. For a large collection of atoms however, the decay rate for that collection can be calculated from their measured decay constants or half-lives. This is the basis of radiometric dating. The half-lives of radioactive atoms have no known limits for shortness or length of duration, and range over 55 orders of magnitude in time.There are many types of radioactive decay (see table below). A decay, or loss of energy from the nucleus, results when an atom with one type of nucleus, called the parent radionuclide (or parent radioisotope), transforms into an atom with a nucleus in a different state, or with a nucleus containing a different number of protons and neutrons. The product is called the daughter nuclide. In some decays, the parent and the daughter nuclides are different chemical elements, and thus the decay process results in the creation of an atom of a different element. This is known as a nuclear transmutation.The first decay processes to be discovered were alpha decay, beta decay, and gamma decay. Alpha decay occurs when the nucleus ejects an alpha particle (helium nucleus). This is the most common process of emitting nucleons, but in rarer types of decays, nuclei can eject protons, or in the case of cluster decay specific nuclei of other elements. Beta decay occurs when the nucleus emits an electron or positron and a neutrino, in a process that changes a proton to a neutron or the other way about. The nucleus may capture an orbiting electron, causing a proton to convert into a neutron in a process called electron capture. All of these processes result in a well-defined nuclear transmutation.By contrast, there are radioactive decay processes that do not result in a nuclear transmutation. The energy of an excited nucleus may be emitted as a gamma ray in a process called gamma decay, or be used to eject an orbital electron by its interaction with the excited nucleus, in a process called internal conversion. Highly excited neutron-rich nuclei, formed as the product of other types of decay, occasionally lose energy by way of neutron emission, resulting in a change of an element from one isotope to another. Another type of radioactive decay results in products that are not defined, but appear in a range of ""pieces"" of the original nucleus. This decay, called spontaneous fission, happens when a large unstable nucleus spontaneously splits into two (and occasionally three) smaller daughter nuclei, and generally leads to the emission of gamma rays, neutrons, or other particles from those products.For a summary table showing the number of stable and radioactive nuclides in each category, see radionuclide. There exist twenty-nine chemical elements on Earth that are radioactive. They are those that contain thirty-four radionuclides that date before the time of formation of the solar system, and are known as primordial nuclides. Well-known examples are uranium and thorium, but also included are naturally occurring long-lived radioisotopes such as potassium-40. Another fifty or so shorter-lived radionuclides, such as radium and radon, found on Earth, are the products of decay chains that began with the primordial nuclides, and ongoing cosmogenic processes, such as the production of carbon-14 from nitrogen-14 by cosmic rays. Radionuclides may also be produced artificially in particle accelerators or nuclear reactors, resulting in 650 of these with half-lives of over an hour, and several thousand more with even shorter half-lives. See this list of nuclides for a list of these, sorted by half life.