Pdf - Text of NPTEL IIT Video Lectures
... the isotopes of a particular element possessing radio activity. Now, isotopes are nuclei having the same number of protons, but different number of neutrons. So, we will discuss this in detail a little later isotopes of an element have seem characteristic atomic number, so that means it have same nu ...
... the isotopes of a particular element possessing radio activity. Now, isotopes are nuclei having the same number of protons, but different number of neutrons. So, we will discuss this in detail a little later isotopes of an element have seem characteristic atomic number, so that means it have same nu ...
1 + - crypt
... radiations including an animation of absorption and a couple of decay equations to fill in on screen. Alpha Decay - PhET - Watch alpha particles escape from a Polonium nucleus, causing radioactive alpha decay. See how random decay times relate to the half life. Beta Decay - PhET - Watch beta decay o ...
... radiations including an animation of absorption and a couple of decay equations to fill in on screen. Alpha Decay - PhET - Watch alpha particles escape from a Polonium nucleus, causing radioactive alpha decay. See how random decay times relate to the half life. Beta Decay - PhET - Watch beta decay o ...
Episode 534 - Teaching Advanced Physics
... the discoverers of nuclear fission – but that was yet to come) wrote to Ernest Rutherford: RaE is the worst of all. We can only obtain a fairly broad band. We formerly thought that it was as narrow as the other bands [as found in other emitters], but that is not true. It looks as if secondary … effe ...
... the discoverers of nuclear fission – but that was yet to come) wrote to Ernest Rutherford: RaE is the worst of all. We can only obtain a fairly broad band. We formerly thought that it was as narrow as the other bands [as found in other emitters], but that is not true. It looks as if secondary … effe ...
Atomic and Nuclear Terms
... ► Radioactive Dating – Method of estimating the age of an object based on the object’s half-life and the amount of isotope present. Carbon dating is limited to the dating of organic (once living) materials. ► (Artificial) Radioactivity – The (induced) spontaneous breakdown of atomic nucleus with emi ...
... ► Radioactive Dating – Method of estimating the age of an object based on the object’s half-life and the amount of isotope present. Carbon dating is limited to the dating of organic (once living) materials. ► (Artificial) Radioactivity – The (induced) spontaneous breakdown of atomic nucleus with emi ...
Structure of the nucleus • It is now known that the nucleus consists of
... It is possible to have different versions of the same element, called isotopes. ...
... It is possible to have different versions of the same element, called isotopes. ...
The Band of Stability
... I can define radioactive decay. I can explain why an unstable nuclide will undergo radioactive decay. I can describe three different types of radioactive decay in terms of balanced nuclear equations. Radioactive Decay Unstable Nuclides 1. An unstable nuclide is also known as a _____________________ ...
... I can define radioactive decay. I can explain why an unstable nuclide will undergo radioactive decay. I can describe three different types of radioactive decay in terms of balanced nuclear equations. Radioactive Decay Unstable Nuclides 1. An unstable nuclide is also known as a _____________________ ...
radioactivity and radioactive decay - rct study guide
... other words, if a nuclide has this property it is said to be radioactive. (The term radionuclide has been coined to refer to these radioactive nuclides.) The emission of a particle or electromagnetic radiation in order to reach a more stable configuration produces a change or transformation. Followi ...
... other words, if a nuclide has this property it is said to be radioactive. (The term radionuclide has been coined to refer to these radioactive nuclides.) The emission of a particle or electromagnetic radiation in order to reach a more stable configuration produces a change or transformation. Followi ...
Document
... • The angular momentum of the electron in an atom is quantized, leading to quantized energy levels in hydrogen-like atoms: En = –13.6 Z2/n2 eV • Photons are emitted and absorbed by atoms at the same wavelength ! identification of elements in the “atmosphere” of stars, discovery of helium. • The ener ...
... • The angular momentum of the electron in an atom is quantized, leading to quantized energy levels in hydrogen-like atoms: En = –13.6 Z2/n2 eV • Photons are emitted and absorbed by atoms at the same wavelength ! identification of elements in the “atmosphere” of stars, discovery of helium. • The ener ...
CH_8_nucleus_new
... Since the neutrons absorbed by 238U are wasted, and since 99.3% of natural U is 238U, too many disappear for a chain reaction to occur in a solid lump of natural U. Slow neutrons are more likely to induce fission in 235U than fast ones. To get around this problem of fast moving neutrons, they are sl ...
... Since the neutrons absorbed by 238U are wasted, and since 99.3% of natural U is 238U, too many disappear for a chain reaction to occur in a solid lump of natural U. Slow neutrons are more likely to induce fission in 235U than fast ones. To get around this problem of fast moving neutrons, they are sl ...
Unit 3 Study Guide: Atomic Structure and Nuclear
... Section 4.3 How Atoms Differ For each statement below, write true or false. _______________ 1. The number of neutrons in an atom is referred to as its atomic number. _______________ 2. The periodic table is arranged by increasing atomic number. _______________ 3. Atomic number is equal to the number ...
... Section 4.3 How Atoms Differ For each statement below, write true or false. _______________ 1. The number of neutrons in an atom is referred to as its atomic number. _______________ 2. The periodic table is arranged by increasing atomic number. _______________ 3. Atomic number is equal to the number ...
Nuclear Physics and Radioactivity2
... Stable nuclei tend to have equal numbers of protons and neutrons for nuclei with Z = to about 30 or 40. If there are too many or too few neutrons relative to the number of protons, the nuclei tends to be unstable. For nuclei with Z greater than 30 or 40, stable nuclei have more neutrons than proton ...
... Stable nuclei tend to have equal numbers of protons and neutrons for nuclei with Z = to about 30 or 40. If there are too many or too few neutrons relative to the number of protons, the nuclei tends to be unstable. For nuclei with Z greater than 30 or 40, stable nuclei have more neutrons than proton ...
Chapter 1
... 9.2 Writing a Balanced Nuclear Equation • Nuclear equation - used to represent nuclear change • In a nuclear equation, you do not balance the elements, instead... – the total mass on each side of the reaction arrow must be identical – the sum of the atomic numbers on each side of the reaction arrow ...
... 9.2 Writing a Balanced Nuclear Equation • Nuclear equation - used to represent nuclear change • In a nuclear equation, you do not balance the elements, instead... – the total mass on each side of the reaction arrow must be identical – the sum of the atomic numbers on each side of the reaction arrow ...
File
... a. Alpha and beta particles penetrate equally. b. Alpha particles penetrate deeper than beta particles. c. Alpha particles and gamma radiation penetrate equally. d. Gamma radiation penetrates deeper than alpha particles. ...
... a. Alpha and beta particles penetrate equally. b. Alpha particles penetrate deeper than beta particles. c. Alpha particles and gamma radiation penetrate equally. d. Gamma radiation penetrates deeper than alpha particles. ...
Power Point - Old Saybrook Public Schools
... Electrons (e) occupy “cloud” outside of nucleus They DO NOT orbit around the nucleus like the planets do around the sun… Impossible to know where an electron is at any ...
... Electrons (e) occupy “cloud” outside of nucleus They DO NOT orbit around the nucleus like the planets do around the sun… Impossible to know where an electron is at any ...
Period 10 Activity Solutions: Nuclear Reactions
... Next, we use the equipment to produce a more realistic model of radioactive decay. Your instructor will explain how to change the settings on the board. a) After charging the capacitor, flip the switch to the right to allow the capacitor to discharge through the resistor. Start the timer and measure ...
... Next, we use the equipment to produce a more realistic model of radioactive decay. Your instructor will explain how to change the settings on the board. a) After charging the capacitor, flip the switch to the right to allow the capacitor to discharge through the resistor. Start the timer and measure ...
The Strong Nuclear Force and the Stability of the Nucleus
... neutrons to be stable. Why? It is the strong nuclear force that holds the nucleons together, but this is a very short range force. The repulsive electric force between the protons is a longer range force. So in a large nucleus all the protons repel each other, but each nucleon ...
... neutrons to be stable. Why? It is the strong nuclear force that holds the nucleons together, but this is a very short range force. The repulsive electric force between the protons is a longer range force. So in a large nucleus all the protons repel each other, but each nucleon ...
nuclear physics - review
... neutrons to be stable. Why? It is the strong nuclear force that holds the nucleons together, but this is a very short range force. The repulsive electric force between the protons is a longer range force. So in a large nucleus all the protons repel each other, but each nucleon ...
... neutrons to be stable. Why? It is the strong nuclear force that holds the nucleons together, but this is a very short range force. The repulsive electric force between the protons is a longer range force. So in a large nucleus all the protons repel each other, but each nucleon ...
Atomic Concepts and Nuclear Chemistry Regents Review Page 1 A
... 39. Which notation of a radioisotope is correctly paired with the notation of its emission particle? B) ...
... 39. Which notation of a radioisotope is correctly paired with the notation of its emission particle? B) ...
lecture notes - University of Chicago
... In terms of our everyday experience, gravity has an obviousness that can’t be claimed by any of the other fundamental forces in nature. Stuff falls. You don’t fly away into space. However, gravity has almost no importance on the subatomic scale, where the remaining three forces (the electromagn ...
... In terms of our everyday experience, gravity has an obviousness that can’t be claimed by any of the other fundamental forces in nature. Stuff falls. You don’t fly away into space. However, gravity has almost no importance on the subatomic scale, where the remaining three forces (the electromagn ...
Document
... • Beta (") – an electron (+ or – charge) is emitted • Gamma (#) – a nucleus falls from one energy level to another and emits a gamma ray photon Friday, April 3, 2009 ...
... • Beta (") – an electron (+ or – charge) is emitted • Gamma (#) – a nucleus falls from one energy level to another and emits a gamma ray photon Friday, April 3, 2009 ...
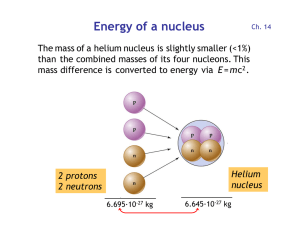
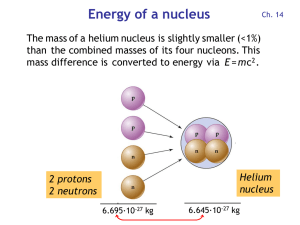
Energy of a nucleus
... Red dots = stable nuclei. The gray region contains unstable nuclei, created in the laboratory. Stable nuclei have about equal neutron and proton numbers N and Z (dashed). At high Z, there are more neutrons than protons, because protons are charged and repel each other. ...
... Red dots = stable nuclei. The gray region contains unstable nuclei, created in the laboratory. Stable nuclei have about equal neutron and proton numbers N and Z (dashed). At high Z, there are more neutrons than protons, because protons are charged and repel each other. ...
Energy per nucleon
... red dots.The gray region contains unstable isotopes which are radioactive. Different isotopes of the same element ...
... red dots.The gray region contains unstable isotopes which are radioactive. Different isotopes of the same element ...
Radioactive decay
Radioactive decay, also known as nuclear decay or radioactivity, is the process by which a nucleus of an unstable atom loses energy by emitting radiation. A material that spontaneously emits such radiation — which includes alpha particles, beta particles, gamma rays and conversion electrons — is considered radioactive.Radioactive decay is a stochastic (i.e. random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. The chance that a given atom will decay never changes, that is, it does not matter how long the atom has existed. For a large collection of atoms however, the decay rate for that collection can be calculated from their measured decay constants or half-lives. This is the basis of radiometric dating. The half-lives of radioactive atoms have no known limits for shortness or length of duration, and range over 55 orders of magnitude in time.There are many types of radioactive decay (see table below). A decay, or loss of energy from the nucleus, results when an atom with one type of nucleus, called the parent radionuclide (or parent radioisotope), transforms into an atom with a nucleus in a different state, or with a nucleus containing a different number of protons and neutrons. The product is called the daughter nuclide. In some decays, the parent and the daughter nuclides are different chemical elements, and thus the decay process results in the creation of an atom of a different element. This is known as a nuclear transmutation.The first decay processes to be discovered were alpha decay, beta decay, and gamma decay. Alpha decay occurs when the nucleus ejects an alpha particle (helium nucleus). This is the most common process of emitting nucleons, but in rarer types of decays, nuclei can eject protons, or in the case of cluster decay specific nuclei of other elements. Beta decay occurs when the nucleus emits an electron or positron and a neutrino, in a process that changes a proton to a neutron or the other way about. The nucleus may capture an orbiting electron, causing a proton to convert into a neutron in a process called electron capture. All of these processes result in a well-defined nuclear transmutation.By contrast, there are radioactive decay processes that do not result in a nuclear transmutation. The energy of an excited nucleus may be emitted as a gamma ray in a process called gamma decay, or be used to eject an orbital electron by its interaction with the excited nucleus, in a process called internal conversion. Highly excited neutron-rich nuclei, formed as the product of other types of decay, occasionally lose energy by way of neutron emission, resulting in a change of an element from one isotope to another. Another type of radioactive decay results in products that are not defined, but appear in a range of ""pieces"" of the original nucleus. This decay, called spontaneous fission, happens when a large unstable nucleus spontaneously splits into two (and occasionally three) smaller daughter nuclei, and generally leads to the emission of gamma rays, neutrons, or other particles from those products.For a summary table showing the number of stable and radioactive nuclides in each category, see radionuclide. There exist twenty-nine chemical elements on Earth that are radioactive. They are those that contain thirty-four radionuclides that date before the time of formation of the solar system, and are known as primordial nuclides. Well-known examples are uranium and thorium, but also included are naturally occurring long-lived radioisotopes such as potassium-40. Another fifty or so shorter-lived radionuclides, such as radium and radon, found on Earth, are the products of decay chains that began with the primordial nuclides, and ongoing cosmogenic processes, such as the production of carbon-14 from nitrogen-14 by cosmic rays. Radionuclides may also be produced artificially in particle accelerators or nuclear reactors, resulting in 650 of these with half-lives of over an hour, and several thousand more with even shorter half-lives. See this list of nuclides for a list of these, sorted by half life.