Worksheet - Rudds Classroom
... 17. The atomic mass of oxygen is 16. How many neutrons does oxygen have in its nucleus? a. l6 b. 4 c. 8 d. 32 18. A uranium atom can have an atomic mass of 235 or 238. Each atom is, therefore, a. a different isotope c. a different element b. negatively charged d. stable 19. Atoms that emit particles ...
... 17. The atomic mass of oxygen is 16. How many neutrons does oxygen have in its nucleus? a. l6 b. 4 c. 8 d. 32 18. A uranium atom can have an atomic mass of 235 or 238. Each atom is, therefore, a. a different isotope c. a different element b. negatively charged d. stable 19. Atoms that emit particles ...
Additional Problems - AppServ Open Project 2.4.9
... radioactive decays. (c) 218Po decays into 214Pb with a half-life of 3.10 min, and 214Pb decays into 214Bi with a half-life of 26.8 min. On the same axes, plot graphs of N1(t) for 218Po and N2(t) for 214Pb. Let N10 = 1 000 nuclei, and choose values of t from 0 to 36 min in 2-min intervals. The curve ...
... radioactive decays. (c) 218Po decays into 214Pb with a half-life of 3.10 min, and 214Pb decays into 214Bi with a half-life of 26.8 min. On the same axes, plot graphs of N1(t) for 218Po and N2(t) for 214Pb. Let N10 = 1 000 nuclei, and choose values of t from 0 to 36 min in 2-min intervals. The curve ...
Nuclear Chemistry - Duplin County Schools
... • Gamma rays are electromagnetic waves with the highest frequencies and the shortest wavelengths in the electromagnetic spectrum. ...
... • Gamma rays are electromagnetic waves with the highest frequencies and the shortest wavelengths in the electromagnetic spectrum. ...
History of Atomic Structure
... proton and a electron, proton remains in nucleus, electron (β particle) propelled out of nucleus at high speeds. ! Mass # is zero ! 100x more penetrating that alpha radiation ! Able to pass through clothing and damage skin ...
... proton and a electron, proton remains in nucleus, electron (β particle) propelled out of nucleus at high speeds. ! Mass # is zero ! 100x more penetrating that alpha radiation ! Able to pass through clothing and damage skin ...
Nuclear Reactions - Kelso High School
... Each element in the periodic table has a different atomic number and is identified by that number. It is possible to have different versions of the same element, called isotopes. An isotope of a atom has the same number of protons but a different number of neutrons, i.e. the same atomic number but a ...
... Each element in the periodic table has a different atomic number and is identified by that number. It is possible to have different versions of the same element, called isotopes. An isotope of a atom has the same number of protons but a different number of neutrons, i.e. the same atomic number but a ...
Nuclear Chemistry - Moorpark College
... from alpha particles due to their negative charge. The neutrino and antineutrino, ν, were first postulated in the 1930's and finally detected in the 1950's. Even today little is known of the neutrinos properties. Neutrinos accompany positron emission and electron capture; antineutrinos are associate ...
... from alpha particles due to their negative charge. The neutrino and antineutrino, ν, were first postulated in the 1930's and finally detected in the 1950's. Even today little is known of the neutrinos properties. Neutrinos accompany positron emission and electron capture; antineutrinos are associate ...
Topic 7_2__Radioactive decay
... Studies showed that there were three types of radioactive particles. If a radioactive substance is placed in a lead chamber and its emitted particles passed through a magnetic field, as shown, the three different types of radioactivity can be distinguished. Alpha particles () are two protons and ...
... Studies showed that there were three types of radioactive particles. If a radioactive substance is placed in a lead chamber and its emitted particles passed through a magnetic field, as shown, the three different types of radioactivity can be distinguished. Alpha particles () are two protons and ...
2 α
... body’s metabolism and regulates other systems in your body. When iodine is ingested, most of it accumulates in the thyroid gland. Iodine-131 is a radioactive isotope of iodine that doctors use to treat diseases associated with this gland. Grave’s disease, an illness caused by an overactive thyroid, ...
... body’s metabolism and regulates other systems in your body. When iodine is ingested, most of it accumulates in the thyroid gland. Iodine-131 is a radioactive isotope of iodine that doctors use to treat diseases associated with this gland. Grave’s disease, an illness caused by an overactive thyroid, ...
radiation!!! - Mr Schmitt
... When these nuclei lose energy and break apart, decay occurs ▪ Radioactive decay releases energy from the nucleus as radiation ▪ Radioactive atoms release energy until they become stable, often ending up as different atoms ▪ For example: uranium-238 (parent nucleus) decays in several stages until i ...
... When these nuclei lose energy and break apart, decay occurs ▪ Radioactive decay releases energy from the nucleus as radiation ▪ Radioactive atoms release energy until they become stable, often ending up as different atoms ▪ For example: uranium-238 (parent nucleus) decays in several stages until i ...
View Transcript
... all the black spots, those are the stable isotopes and the orange-red spots are the radioactive isotopes. Now, if you go way up into the upper right-hand corner of the graphic, you’ll see that there are a bunch of nuclei that become really unstable. So 84 and above, they’re all unstable. And if you ...
... all the black spots, those are the stable isotopes and the orange-red spots are the radioactive isotopes. Now, if you go way up into the upper right-hand corner of the graphic, you’ll see that there are a bunch of nuclei that become really unstable. So 84 and above, they’re all unstable. And if you ...
radioisotopes and radiotherapy - video
... 10% Cosmic Rays from Sun and outer space, 35% medical and industrial use. 13. Ionising radiation can kill cells or change the way they function. 14. High doses of radiation from beta particles. 15. Low dose of gamma radiation. 16. In nuclear reactors (or cyclotrons, discussed later in video). In a n ...
... 10% Cosmic Rays from Sun and outer space, 35% medical and industrial use. 13. Ionising radiation can kill cells or change the way they function. 14. High doses of radiation from beta particles. 15. Low dose of gamma radiation. 16. In nuclear reactors (or cyclotrons, discussed later in video). In a n ...
Balancing a Nuclear Equation
... Nuclear Chemistry • Nuclear chemistry • study of atomic nucleus structure and the changes they undergo ...
... Nuclear Chemistry • Nuclear chemistry • study of atomic nucleus structure and the changes they undergo ...
25.1 Nuclear Radiation
... alpha (α ), beta (β ), and gamma (γ ) radiation. Although all forms of radiation are somewhat harmful, gamma rays are particularly dangerous because they penetrate body tissues.) Explain that radioactivity reflects the tendency of atomic nuclei to achieve stability. Ask, What makes a nucleus unstabl ...
... alpha (α ), beta (β ), and gamma (γ ) radiation. Although all forms of radiation are somewhat harmful, gamma rays are particularly dangerous because they penetrate body tissues.) Explain that radioactivity reflects the tendency of atomic nuclei to achieve stability. Ask, What makes a nucleus unstabl ...
Appendix A Glossary of Nuclear Terms
... nuclide: Any species of atom that exists for a measurable length of time. A nuclide can be distinguished by its atomic mass, atomic number, and energy state. parent: A radionuclide that decays to another nuclide. photon: A packet of electromagnetic energy. Photons have momentum and energy, but no re ...
... nuclide: Any species of atom that exists for a measurable length of time. A nuclide can be distinguished by its atomic mass, atomic number, and energy state. parent: A radionuclide that decays to another nuclide. photon: A packet of electromagnetic energy. Photons have momentum and energy, but no re ...
Basics of nuclear physics
... Weak force acts between particles called leptons (electron, neutrino and their antiparticles are leptons). The transformation of nucleons, mediated by weak force, is always accompanied by emission of two leptons, which are particle-antiparticle pair. ...
... Weak force acts between particles called leptons (electron, neutrino and their antiparticles are leptons). The transformation of nucleons, mediated by weak force, is always accompanied by emission of two leptons, which are particle-antiparticle pair. ...
Nuclear Chemistry
... in balancing the reaction we always made sure that the number of atoms of each element on both sides of the eqn. was the same and the charge was equal as well. In a nuclear process the identity of an atom may change. In chemical rxns., only the outer electrons of the atoms are disturbed. Remember th ...
... in balancing the reaction we always made sure that the number of atoms of each element on both sides of the eqn. was the same and the charge was equal as well. In a nuclear process the identity of an atom may change. In chemical rxns., only the outer electrons of the atoms are disturbed. Remember th ...
Examination 1
... in balancing the reaction we always made sure that the number of atoms of each element on both sides of the eqn. was the same and the charge was equal as well. In a nuclear process the identity of an atom may change. In chemical rxns., only the outer electrons of the atoms are disturbed. Remember th ...
... in balancing the reaction we always made sure that the number of atoms of each element on both sides of the eqn. was the same and the charge was equal as well. In a nuclear process the identity of an atom may change. In chemical rxns., only the outer electrons of the atoms are disturbed. Remember th ...
Notes: Nuclear Chemistry
... a. For light elements (1-20), proton to neutron ratio should be 1:1 for stable nuclei (non-radioactive). a. For heavier elements (above 20), proton to neutron ratio increases gradually to 1:1.5 for stable nuclei. Radioactivity = the spontaneous emission of penetrating rays/particles (radiation) by a ...
... a. For light elements (1-20), proton to neutron ratio should be 1:1 for stable nuclei (non-radioactive). a. For heavier elements (above 20), proton to neutron ratio increases gradually to 1:1.5 for stable nuclei. Radioactivity = the spontaneous emission of penetrating rays/particles (radiation) by a ...
Basics of Nuclear Physics and Fission
... Beta decay, which the emission of an electron or a positron (a particle identical to an electron except that it has a positive electrical charge). Electron capture, which is the capture by the nucleus of an electron from among the ones whirling around it. In effect, the electron combines with a prot ...
... Beta decay, which the emission of an electron or a positron (a particle identical to an electron except that it has a positive electrical charge). Electron capture, which is the capture by the nucleus of an electron from among the ones whirling around it. In effect, the electron combines with a prot ...
have shown no evidence
... • Stability is favoured by even numbers of protons and neutrons • Not usually equal numbers • Plotting neutron number (A) against proton number (Z) for all known nuclei, shows area of stability • For very light elements N ≈ Z gives stable elements • 1:1 up to 4020Ca • Ratio gradually rises (A>Z) unt ...
... • Stability is favoured by even numbers of protons and neutrons • Not usually equal numbers • Plotting neutron number (A) against proton number (Z) for all known nuclei, shows area of stability • For very light elements N ≈ Z gives stable elements • 1:1 up to 4020Ca • Ratio gradually rises (A>Z) unt ...
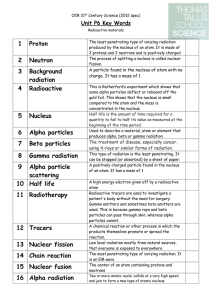
3 Background radiation
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
Chemistry: Matter and Change
... that led to understanding radiation. • Identify alpha, beta, and gamma radiations in terms of composition and key properties. ...
... that led to understanding radiation. • Identify alpha, beta, and gamma radiations in terms of composition and key properties. ...
Topic 14 - Lloyd Crosby
... d. The RBE factor depends on how destructive to biological tissues a type of radiation happens to be for the same amount of energy delivered to the tissue e. RBEs for selected radiation (1) X-rays: RBE = 0.7 (2) beta: ...
... d. The RBE factor depends on how destructive to biological tissues a type of radiation happens to be for the same amount of energy delivered to the tissue e. RBEs for selected radiation (1) X-rays: RBE = 0.7 (2) beta: ...
Nuclear Chemistry
... atom of a different element is formed. This decomposition of the nucleus is referred to as radioactive decay. During radioactive decay an unstable nucleus spontaneosly decomposes to form a different nucleus, giving off radiation in the form of atomic partices or high energy rays. This decay occurs ...
... atom of a different element is formed. This decomposition of the nucleus is referred to as radioactive decay. During radioactive decay an unstable nucleus spontaneosly decomposes to form a different nucleus, giving off radiation in the form of atomic partices or high energy rays. This decay occurs ...
Radioactive decay
Radioactive decay, also known as nuclear decay or radioactivity, is the process by which a nucleus of an unstable atom loses energy by emitting radiation. A material that spontaneously emits such radiation — which includes alpha particles, beta particles, gamma rays and conversion electrons — is considered radioactive.Radioactive decay is a stochastic (i.e. random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. The chance that a given atom will decay never changes, that is, it does not matter how long the atom has existed. For a large collection of atoms however, the decay rate for that collection can be calculated from their measured decay constants or half-lives. This is the basis of radiometric dating. The half-lives of radioactive atoms have no known limits for shortness or length of duration, and range over 55 orders of magnitude in time.There are many types of radioactive decay (see table below). A decay, or loss of energy from the nucleus, results when an atom with one type of nucleus, called the parent radionuclide (or parent radioisotope), transforms into an atom with a nucleus in a different state, or with a nucleus containing a different number of protons and neutrons. The product is called the daughter nuclide. In some decays, the parent and the daughter nuclides are different chemical elements, and thus the decay process results in the creation of an atom of a different element. This is known as a nuclear transmutation.The first decay processes to be discovered were alpha decay, beta decay, and gamma decay. Alpha decay occurs when the nucleus ejects an alpha particle (helium nucleus). This is the most common process of emitting nucleons, but in rarer types of decays, nuclei can eject protons, or in the case of cluster decay specific nuclei of other elements. Beta decay occurs when the nucleus emits an electron or positron and a neutrino, in a process that changes a proton to a neutron or the other way about. The nucleus may capture an orbiting electron, causing a proton to convert into a neutron in a process called electron capture. All of these processes result in a well-defined nuclear transmutation.By contrast, there are radioactive decay processes that do not result in a nuclear transmutation. The energy of an excited nucleus may be emitted as a gamma ray in a process called gamma decay, or be used to eject an orbital electron by its interaction with the excited nucleus, in a process called internal conversion. Highly excited neutron-rich nuclei, formed as the product of other types of decay, occasionally lose energy by way of neutron emission, resulting in a change of an element from one isotope to another. Another type of radioactive decay results in products that are not defined, but appear in a range of ""pieces"" of the original nucleus. This decay, called spontaneous fission, happens when a large unstable nucleus spontaneously splits into two (and occasionally three) smaller daughter nuclei, and generally leads to the emission of gamma rays, neutrons, or other particles from those products.For a summary table showing the number of stable and radioactive nuclides in each category, see radionuclide. There exist twenty-nine chemical elements on Earth that are radioactive. They are those that contain thirty-four radionuclides that date before the time of formation of the solar system, and are known as primordial nuclides. Well-known examples are uranium and thorium, but also included are naturally occurring long-lived radioisotopes such as potassium-40. Another fifty or so shorter-lived radionuclides, such as radium and radon, found on Earth, are the products of decay chains that began with the primordial nuclides, and ongoing cosmogenic processes, such as the production of carbon-14 from nitrogen-14 by cosmic rays. Radionuclides may also be produced artificially in particle accelerators or nuclear reactors, resulting in 650 of these with half-lives of over an hour, and several thousand more with even shorter half-lives. See this list of nuclides for a list of these, sorted by half life.