穨 Ams1a
... By introducing a short- lived radioisotope into the pipes. The level of radioactivity on the ground surface can then be monitored. A sudden increase of surface radioactivity indicates that there is a leakage of underground pipes. iv) Efficiency of lubricating oils Engine wear can be measured by usin ...
... By introducing a short- lived radioisotope into the pipes. The level of radioactivity on the ground surface can then be monitored. A sudden increase of surface radioactivity indicates that there is a leakage of underground pipes. iv) Efficiency of lubricating oils Engine wear can be measured by usin ...
Practice Test/Quiz Questions – Chapter 1, 2, 3 Chemical Foundations
... What causes the deflection of an alpha particle by the nucleus of a gold atom? a. The widespread distribution of mass in the atom b. The low mass of the nucleus c. The positive charge of the alpha particle and the negative charge of the nucleus d. The negative charge of the alpha particle and the po ...
... What causes the deflection of an alpha particle by the nucleus of a gold atom? a. The widespread distribution of mass in the atom b. The low mass of the nucleus c. The positive charge of the alpha particle and the negative charge of the nucleus d. The negative charge of the alpha particle and the po ...
Learning Targets Chapter 4
... charge, location and relative mass of protons (p+) , neutrons (n0) and electrons (e-) in an atom. I can calculate the number of protons (p+) , neutrons (n0) and electrons (e-) in an atom using the atomic number, mass number and overall charge of the atom or a periodic table provided. I can describe ...
... charge, location and relative mass of protons (p+) , neutrons (n0) and electrons (e-) in an atom. I can calculate the number of protons (p+) , neutrons (n0) and electrons (e-) in an atom using the atomic number, mass number and overall charge of the atom or a periodic table provided. I can describe ...
Terms to Know
... Positrons : The positron is the antiparticle of the electron. It has the same mass and the same quantity of electric charge as does the electron, but its electric charge is positive rather than negative. Radioactivity : Radioactivity is the emission of radiation by unstable nuclei. That radiation ma ...
... Positrons : The positron is the antiparticle of the electron. It has the same mass and the same quantity of electric charge as does the electron, but its electric charge is positive rather than negative. Radioactivity : Radioactivity is the emission of radiation by unstable nuclei. That radiation ma ...
SMART Notebook
... - But, the little bit of carbon-13 does pull up the number a bit, so we get 12.01u. - The number of neutrons strongly affects the stability of the nucleus. This is why some isotopes are more common than others. - Because different isotopes have different masses, they can be separated from each other ...
... - But, the little bit of carbon-13 does pull up the number a bit, so we get 12.01u. - The number of neutrons strongly affects the stability of the nucleus. This is why some isotopes are more common than others. - Because different isotopes have different masses, they can be separated from each other ...
Radioactivity Unit - hrsbstaff.ednet.ns.ca
... c. 239Np 239U + _____ ? 5. In #3 and #4 above, which reactions involved the emission of a neutrino? Which involved the emission of an antineutrino? 6. Write the equation for the electron capture of chromium 51. 7. The mass of a Beryllium 7 nucleus is 1.1652 10-26 kg. a. What is the difference be ...
... c. 239Np 239U + _____ ? 5. In #3 and #4 above, which reactions involved the emission of a neutrino? Which involved the emission of an antineutrino? 6. Write the equation for the electron capture of chromium 51. 7. The mass of a Beryllium 7 nucleus is 1.1652 10-26 kg. a. What is the difference be ...
Chapter 5
... Unlike charges attract and like charges repel Charges may be transferred by contact or induction The closer two object are, the greater the force of attraction ...
... Unlike charges attract and like charges repel Charges may be transferred by contact or induction The closer two object are, the greater the force of attraction ...
e - Central Lyon CSD
... a. Electrons (e) – negatively charged particle found outside of the nucleus -has a mass of 0 -has a charge of 1-moves around the outside of the nucleus ...
... a. Electrons (e) – negatively charged particle found outside of the nucleus -has a mass of 0 -has a charge of 1-moves around the outside of the nucleus ...
Atomic Nuclei - RAJEEV Classes
... number increases by 1 but mass number remains same. c)The emission of a γ-particle does not change the mass number or the atomic number of the radioactive nucleus. The γ-particle emission by a radioactive nucleus lowers its energy state. ...
... number increases by 1 but mass number remains same. c)The emission of a γ-particle does not change the mass number or the atomic number of the radioactive nucleus. The γ-particle emission by a radioactive nucleus lowers its energy state. ...
Section 4.2 The Structure of an Atom
... blocks” of matter, and some may know that atoms contain subatomic particles. ...
... blocks” of matter, and some may know that atoms contain subatomic particles. ...
Neutron - Piscataway High School
... Atomic mass unit: one-twelfth the mass of a carbon atom having 6 protons and 6 neutrons Atomic number: the number of protons in the nucleus of an atom Electron: a negatively charged subatomic particle Isotope: atoms with the same number of protons, but different number of neutrons in the nucleus of ...
... Atomic mass unit: one-twelfth the mass of a carbon atom having 6 protons and 6 neutrons Atomic number: the number of protons in the nucleus of an atom Electron: a negatively charged subatomic particle Isotope: atoms with the same number of protons, but different number of neutrons in the nucleus of ...
Atom - Sites
... atoms join together chemically. •Combinations of two or more different elements are called compounds. •All compounds are molecules but not all molecules are compounds. (ex. H2O vs. O2) •Molecules can also join together to form larger molecules. •Many, many repeating small molecules joined together f ...
... atoms join together chemically. •Combinations of two or more different elements are called compounds. •All compounds are molecules but not all molecules are compounds. (ex. H2O vs. O2) •Molecules can also join together to form larger molecules. •Many, many repeating small molecules joined together f ...
Glencoe Chapter 4 Structure of the Atom for the Wiki
... Based on atomic theory but no experiment evidence at the time • The ratio of the masses of one element that combine with a constant mass of another element can be expressed in small whole numbers. ...
... Based on atomic theory but no experiment evidence at the time • The ratio of the masses of one element that combine with a constant mass of another element can be expressed in small whole numbers. ...
File
... Isotopes are often unstable – they have more neutrons than the element “wants” The isotopes are naturally occurring & decompose at different rates depending on the type of element. A nucleus that is unstable can become stable by undergoing a nuclear reaction (or change) Properties: Alters ph ...
... Isotopes are often unstable – they have more neutrons than the element “wants” The isotopes are naturally occurring & decompose at different rates depending on the type of element. A nucleus that is unstable can become stable by undergoing a nuclear reaction (or change) Properties: Alters ph ...
Nucleus - schoolphysics
... Light nuclei such as hydrogen and carbon usually contain as many protons as neutrons but the nuclei of heavy elements such as iron, radium or uranium contain many more neutrons than protons. You can find a list of the structure of many nuclei in Nuclear physics/Elements. One big problems with this m ...
... Light nuclei such as hydrogen and carbon usually contain as many protons as neutrons but the nuclei of heavy elements such as iron, radium or uranium contain many more neutrons than protons. You can find a list of the structure of many nuclei in Nuclear physics/Elements. One big problems with this m ...
All substances are made from atoms
... smallest particle which exists of an element. All of the atoms of any one element (say oxygen) are identical. Oxygen gas is made from trillions of identical oxygen atoms. There are just over one hundred elements in the periodic table, so there are just over one hundred types of atoms in the universe ...
... smallest particle which exists of an element. All of the atoms of any one element (say oxygen) are identical. Oxygen gas is made from trillions of identical oxygen atoms. There are just over one hundred elements in the periodic table, so there are just over one hundred types of atoms in the universe ...
Concept Reviews Answer sheet Section: matter and Energy 1. a
... 3. Coffee at 38°C has more kinetic energy than coffee at 34°C. Although there is less tea than coffee, the temperature of the tea is greater, so the tea has more average kinetic energy than the coffee. 4. The temperature, and therefore the kinetic energy of the particles in 0.5 L of coffee and 0.25 ...
... 3. Coffee at 38°C has more kinetic energy than coffee at 34°C. Although there is less tea than coffee, the temperature of the tea is greater, so the tea has more average kinetic energy than the coffee. 4. The temperature, and therefore the kinetic energy of the particles in 0.5 L of coffee and 0.25 ...
making a bohr model - hrsbstaff.ednet.ns.ca
... and negatively charged electrons in specific orbits, also called energy levels or shells, around the nucleus. Because of its simplicity and general ability to explain chemical change, the Bohr model is still used today. In a Bohr model, the nucleus is drawn as a circle with the number of protons & n ...
... and negatively charged electrons in specific orbits, also called energy levels or shells, around the nucleus. Because of its simplicity and general ability to explain chemical change, the Bohr model is still used today. In a Bohr model, the nucleus is drawn as a circle with the number of protons & n ...
Chapter 16 – Nuclear Energy
... • Radioactive atoms: Atoms that decay and emit particles and energy from their nuclei. • Radiation: alpha particles, beta particles and gamma rays given off in the decaying of unstable nuclei. ...
... • Radioactive atoms: Atoms that decay and emit particles and energy from their nuclei. • Radiation: alpha particles, beta particles and gamma rays given off in the decaying of unstable nuclei. ...
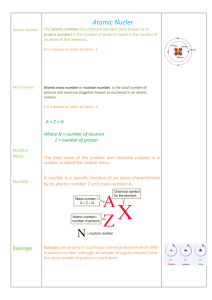
Atomic Notation
... Z – the atomic number -The mass number is the number of protons and neutrons in that element. The number is rounded to the nearest whole number (no decimals in standard notation) -The atomic number is the number of protons -All atoms of the same element have the same number of protons (or atomic num ...
... Z – the atomic number -The mass number is the number of protons and neutrons in that element. The number is rounded to the nearest whole number (no decimals in standard notation) -The atomic number is the number of protons -All atoms of the same element have the same number of protons (or atomic num ...
PHY140Y 33 Nuclear Properties - University of Toronto, Particle
... Lord Rutherford in 1909-1911 showed that the atom had to have a small, hard core that carried the bulk of the mass of the atom. Since atoms where neutral, the so-called nucleus of the atom had to have the same number of protons as the atom had electrons. This was the beginning of nuclear physics as ...
... Lord Rutherford in 1909-1911 showed that the atom had to have a small, hard core that carried the bulk of the mass of the atom. Since atoms where neutral, the so-called nucleus of the atom had to have the same number of protons as the atom had electrons. This was the beginning of nuclear physics as ...
Earth`s Chemistry
... Atomic number = equals the number of protons in the atom. An uncharged atom has the same amount of protons & electrons so there’s no charge. It’s said to be neutral Example --- Oxygen has an atomic number of 8 so it has 8 protons & 8 ...
... Atomic number = equals the number of protons in the atom. An uncharged atom has the same amount of protons & electrons so there’s no charge. It’s said to be neutral Example --- Oxygen has an atomic number of 8 so it has 8 protons & 8 ...
Content Domain III: Chemistry—Atomic Theory and
... unstable, which means that the nucleus emits a particle or radiation with a large amount of energy to help it become more stable. This process is called radioactive decay. When the nucleus in a radioactive material breaks down, it can produce three types of nuclear radiation—alpha, beta, and gamma. ...
... unstable, which means that the nucleus emits a particle or radiation with a large amount of energy to help it become more stable. This process is called radioactive decay. When the nucleus in a radioactive material breaks down, it can produce three types of nuclear radiation—alpha, beta, and gamma. ...