NUCLEAR CHEMISTRY
... The total energy required to break up a nucleus into its constituent protons & neutrons binding energy E = mc2 The nuclear binding energy is measured in MeV (mega electron volts) which is much larger than the few eV required to hold electrons to an atom ...
... The total energy required to break up a nucleus into its constituent protons & neutrons binding energy E = mc2 The nuclear binding energy is measured in MeV (mega electron volts) which is much larger than the few eV required to hold electrons to an atom ...
atomic
... impossible to know precisely both an electron’s position and path at a given time. Led to the electron cloud theory. electron ...
... impossible to know precisely both an electron’s position and path at a given time. Led to the electron cloud theory. electron ...
Name
... • An atom is the smallest particle of an element that retains the chemical properties of that element. • The nucleus is a very small region located at the center of an atom. • The nucleus is made up of at least one positively charged particle called a proton and usually one or more neutral particles ...
... • An atom is the smallest particle of an element that retains the chemical properties of that element. • The nucleus is a very small region located at the center of an atom. • The nucleus is made up of at least one positively charged particle called a proton and usually one or more neutral particles ...
Semester 1 Exam Review Part 1
... atomic number is equal to the number of protons in the nucleus A = Atomic Number These are all equal to each P = Proton Number other E = Electron Number ...
... atomic number is equal to the number of protons in the nucleus A = Atomic Number These are all equal to each P = Proton Number other E = Electron Number ...
Radioactivityunit6
... • The process of causing a large nucleus (A > 120) to split into multiple smaller nuclei, releasing energy in the process. • It can start when the large nuclei absorbs a neutron, causing it to become unstable to the point that it falls apart. • This is the reaction that we use in nuclear power plant ...
... • The process of causing a large nucleus (A > 120) to split into multiple smaller nuclei, releasing energy in the process. • It can start when the large nuclei absorbs a neutron, causing it to become unstable to the point that it falls apart. • This is the reaction that we use in nuclear power plant ...
Test Review Answers File
... 19. Phosphorus has an atomic number of 15 and a mass number of 31. How many protons, neutrons, and electrons does a neutral atom of phosphorus contain? a. Protons = 15 b. Neutrons = 16 c. Electrons = 15 20. Which part of the atom was discovered as a result of the Gold Foil experiment? ...
... 19. Phosphorus has an atomic number of 15 and a mass number of 31. How many protons, neutrons, and electrons does a neutral atom of phosphorus contain? a. Protons = 15 b. Neutrons = 16 c. Electrons = 15 20. Which part of the atom was discovered as a result of the Gold Foil experiment? ...
Uconn Physics Spring 2007 Exam
... 6. A particular double slit interference experiment causes red light with a wavelength of 700 nm to form a second order bright spot at an angle of 20 degrees. What is the space between the slits? b) An object 3.00 cm high is placed 20.0 cm from a convex mirror, with a focal length of 8.00 cm. Find t ...
... 6. A particular double slit interference experiment causes red light with a wavelength of 700 nm to form a second order bright spot at an angle of 20 degrees. What is the space between the slits? b) An object 3.00 cm high is placed 20.0 cm from a convex mirror, with a focal length of 8.00 cm. Find t ...
1 - cloudfront.net
... 13. Why did J. J. Thomson reason that electrons must be a part of the atoms of all elements? 14. Know characteristics regarding the nucleus of an atom. 15. Describe the model of the atom as a result of Rutherford’s discovery of the nucleus. 16. The smallest particle of an element that retains the pr ...
... 13. Why did J. J. Thomson reason that electrons must be a part of the atoms of all elements? 14. Know characteristics regarding the nucleus of an atom. 15. Describe the model of the atom as a result of Rutherford’s discovery of the nucleus. 16. The smallest particle of an element that retains the pr ...
The study of biology can help you better understand human
... 10. Atomic mass is measured in ____________________________11. How do the isotopes of an element differ?_________________________________ How are they alike? ______________________________________________ 12. The number 37 in the name chlorine-37 represents __________________ 13. What does each numb ...
... 10. Atomic mass is measured in ____________________________11. How do the isotopes of an element differ?_________________________________ How are they alike? ______________________________________________ 12. The number 37 in the name chlorine-37 represents __________________ 13. What does each numb ...
four forces in atom ppt
... Comparatively, electrons have 1/1836 the mass of either the proton or neutron ...
... Comparatively, electrons have 1/1836 the mass of either the proton or neutron ...
The Atom - Taylorsville
... Five main points of Dalton's atomic theory 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. ...
... Five main points of Dalton's atomic theory 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. ...
Alpha Decay
... The total number of neutrons and protons in a nucleus is called its mass number A. Neutrons and protons, when considered collectively, are called nucleons. ...
... The total number of neutrons and protons in a nucleus is called its mass number A. Neutrons and protons, when considered collectively, are called nucleons. ...
Atoms: The Building Blocks of Matter
... ▫ atomic mass units ▫ masses so small, came up with scale that is relative ▫ amu = mass of 1/12 mass of carbon-12 atom • Average atomic masses ▫ # on periodic table ▫ Decimals averages ...
... ▫ atomic mass units ▫ masses so small, came up with scale that is relative ▫ amu = mass of 1/12 mass of carbon-12 atom • Average atomic masses ▫ # on periodic table ▫ Decimals averages ...
Cycle 4 Topic 1 C1 Atomic Structure Cycle Sheet
... I can use the atomic model to describe atoms. I can calculate the numbers of protons, neutrons and electrons in an atom or ion, given its atomic number and mass number. I can represent the electronic structures of the first twenty elements of the periodic table in both forms I can visualise and repr ...
... I can use the atomic model to describe atoms. I can calculate the numbers of protons, neutrons and electrons in an atom or ion, given its atomic number and mass number. I can represent the electronic structures of the first twenty elements of the periodic table in both forms I can visualise and repr ...
Chapter 25 Radioactivity
... Isotopes: atoms of the same element with different numbers of neutrons i.e 1214C and 1213C each has 12 protons which is the Atomic number. The mass number varies. It is the sum of the protons and neutrons. ...
... Isotopes: atoms of the same element with different numbers of neutrons i.e 1214C and 1213C each has 12 protons which is the Atomic number. The mass number varies. It is the sum of the protons and neutrons. ...
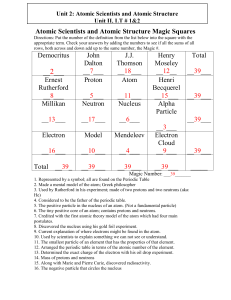
Atomic Scientists and Atomic Structure Magic Squares
... 9. Current explanation of where electrons might be found in the atom. 10. Used by scientists to explain something we can not see or understand. 11. The smallest particle of an element that has the properties of that element. 12. Arranged the periodic table in terms of the atomic number of the elemen ...
... 9. Current explanation of where electrons might be found in the atom. 10. Used by scientists to explain something we can not see or understand. 11. The smallest particle of an element that has the properties of that element. 12. Arranged the periodic table in terms of the atomic number of the elemen ...
An atom - Ms. Buicke maths and science
... Because of this atoms are neutral. The charge of the proton cancels out the charge on the electron. ...
... Because of this atoms are neutral. The charge of the proton cancels out the charge on the electron. ...
Document
... A simple model of the atom What is inside atoms? In a simple model of atomic structure the atoms are thought to be made from three types of sub-atomic particle: protons, neutrons and electrons. Protons and neutrons form the dense nucleus of atoms. Electrons are much more diffuse and move around the ...
... A simple model of the atom What is inside atoms? In a simple model of atomic structure the atoms are thought to be made from three types of sub-atomic particle: protons, neutrons and electrons. Protons and neutrons form the dense nucleus of atoms. Electrons are much more diffuse and move around the ...
Atom
... the “planetary model” 1926: Erwin Schrodinger analyzed electron orbits from a geometric point using quantum physics, ...
... the “planetary model” 1926: Erwin Schrodinger analyzed electron orbits from a geometric point using quantum physics, ...
Chemistry Notes
... • The sum of the number of neutrons and protons in an atomic nucleus. 12.01 is the mass of 1 mole of Carbon. ...
... • The sum of the number of neutrons and protons in an atomic nucleus. 12.01 is the mass of 1 mole of Carbon. ...
Bohr Model
... Bohr model will show how many protons and neutrons are in the nucleus. It will also show how many electrons are surrounding the nucleus. Follow the directions below to create Bohr models of the elements listed. ...
... Bohr model will show how many protons and neutrons are in the nucleus. It will also show how many electrons are surrounding the nucleus. Follow the directions below to create Bohr models of the elements listed. ...
File - Dr. Wall`s Science
... • Put experiment in desk drawer, where there was no light • Image still showed up on paper, due to energy from minerals • This energy is nuclear radiation ...
... • Put experiment in desk drawer, where there was no light • Image still showed up on paper, due to energy from minerals • This energy is nuclear radiation ...
atoms_minerals_2001
... the "electron cloud" orbits the nucleus; electrons are confined to "shells" electron (-) = negligible mass the structure of an atom invokes the analogy of a miniature solar system where the sun represents the nucleus, and the rest of the solar system represents the elctron cloud. The planetary ...
... the "electron cloud" orbits the nucleus; electrons are confined to "shells" electron (-) = negligible mass the structure of an atom invokes the analogy of a miniature solar system where the sun represents the nucleus, and the rest of the solar system represents the elctron cloud. The planetary ...