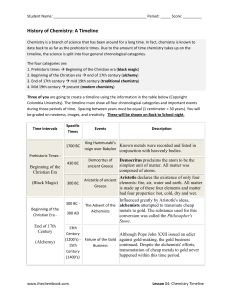
chemistry form iii - Covington Latin School
... Students enrolled in this course will receive an introduction to the following topics: measurements in chemistry; elements and compounds; matter and energy; periodic table and chemical nomenclature; chemical reactions; quantities in chemistry; stoichiometry; atomic theory; the chemical bond; gases; ...
... Students enrolled in this course will receive an introduction to the following topics: measurements in chemistry; elements and compounds; matter and energy; periodic table and chemical nomenclature; chemical reactions; quantities in chemistry; stoichiometry; atomic theory; the chemical bond; gases; ...
Unit Description - Honors Chemistry
... Explain the origin of the atomic emission spectrum of an element, using Bohr’s hydrogen spectrum (5.1) Describe the quantum mechanical model of the atom (5.2) Describe Heisenberg’s Uncertainty Principle (5.2) Distinguish between an orbit and an orbital (5.2) Distinguish among principal ene ...
... Explain the origin of the atomic emission spectrum of an element, using Bohr’s hydrogen spectrum (5.1) Describe the quantum mechanical model of the atom (5.2) Describe Heisenberg’s Uncertainty Principle (5.2) Distinguish between an orbit and an orbital (5.2) Distinguish among principal ene ...
第一章 绪论
... Another step in drug discovery involves further chemical modifications in order to improve the biological and physiochemical properties of a given candidate compound library. Chemical modifications can improve the pharmacophores of the candidate compounds, their pharmacokinetics, or indeed their rea ...
... Another step in drug discovery involves further chemical modifications in order to improve the biological and physiochemical properties of a given candidate compound library. Chemical modifications can improve the pharmacophores of the candidate compounds, their pharmacokinetics, or indeed their rea ...
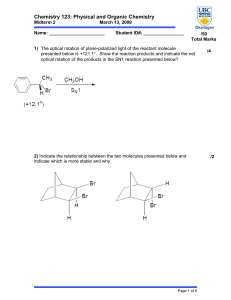
Midterm Review Sample Content Questions
... 9. Which of the following signs of a chemical reaction could potentially occur in a physical change too: color change, temperature change, gas evolution, formation of a precipitate, new substance forms. List any/all that apply and what conditions must occur for them to be considered physical by prov ...
... 9. Which of the following signs of a chemical reaction could potentially occur in a physical change too: color change, temperature change, gas evolution, formation of a precipitate, new substance forms. List any/all that apply and what conditions must occur for them to be considered physical by prov ...
Document
... In this reaction two light atomic nuclei, when they are very close to each other, fuse together to form a single heavier nucleus of a new element. The process is exothermic (release of energy). The nuclear fusions occur at only very high temperatures. When 2 hydrogen nuclei fuse together by nuclear ...
... In this reaction two light atomic nuclei, when they are very close to each other, fuse together to form a single heavier nucleus of a new element. The process is exothermic (release of energy). The nuclear fusions occur at only very high temperatures. When 2 hydrogen nuclei fuse together by nuclear ...
Atomic Theory Handout CNS 8
... had recently suggested the atom had a miniature, dense nucleus surrounded by a cloud of nearly weightless electrons. There were a few problems with the model, however. For example, according to classical physics, the electrons orbiting the nucleus should lose energy until they spiral down into the c ...
... had recently suggested the atom had a miniature, dense nucleus surrounded by a cloud of nearly weightless electrons. There were a few problems with the model, however. For example, according to classical physics, the electrons orbiting the nucleus should lose energy until they spiral down into the c ...
Topic 14 - Lloyd Crosby
... d. The RBE factor depends on how destructive to biological tissues a type of radiation happens to be for the same amount of energy delivered to the tissue e. RBEs for selected radiation (1) X-rays: RBE = 0.7 (2) beta: ...
... d. The RBE factor depends on how destructive to biological tissues a type of radiation happens to be for the same amount of energy delivered to the tissue e. RBEs for selected radiation (1) X-rays: RBE = 0.7 (2) beta: ...
Chemistry and the Environment - Teach-n-Learn-Chem
... 4. What gas is produced by burning coal and wood? What is a possible side effect of increasing the levels of this gas in the atmosphere? Carbon dioxide - greenhouse effect 5. What insecticide which saved many lives from insect borne diseases was banned because it killed birds and fish? ...
... 4. What gas is produced by burning coal and wood? What is a possible side effect of increasing the levels of this gas in the atmosphere? Carbon dioxide - greenhouse effect 5. What insecticide which saved many lives from insect borne diseases was banned because it killed birds and fish? ...
nuclear reactions
... A 2000 MW coal-fired power station releases 42,000 tons of CO2, 600 tons of SO2 (and related acid gases) and 10 tons of fly ash per day. Scrubbers can reduce the acid gases and fly ash considerably, but nothing can be done about the CO2 greenhouse gas Power in Alberta is generated near Edmonton from ...
... A 2000 MW coal-fired power station releases 42,000 tons of CO2, 600 tons of SO2 (and related acid gases) and 10 tons of fly ash per day. Scrubbers can reduce the acid gases and fly ash considerably, but nothing can be done about the CO2 greenhouse gas Power in Alberta is generated near Edmonton from ...
Biology Fall Semester Test 1 Study Guide
... In the metric system, the basic unit of length is the How many centimeters are in 2.4 km? The basic unit of mass in SI is the The three particles that make up atoms are ...
... In the metric system, the basic unit of length is the How many centimeters are in 2.4 km? The basic unit of mass in SI is the The three particles that make up atoms are ...
Unit IV Review Guide: Atomic Structure and Nuclear Reactions
... ● Understand and define the three types of radioactive decay in terms of composition and penetrating ability. ● Be able to write nuclear decay equations ...
... ● Understand and define the three types of radioactive decay in terms of composition and penetrating ability. ● Be able to write nuclear decay equations ...
Chapter 4: The Structure of the Atom &
... o Gamma radiation is made of GAMMA RAYS (high energy radiation) No mass or charge Symbol is _______________ ...
... o Gamma radiation is made of GAMMA RAYS (high energy radiation) No mass or charge Symbol is _______________ ...