Unit 2 Atomic Structure 2015
... Energy Levels • Bohr discovered that different orbitals required different amounts of energy for an ...
... Energy Levels • Bohr discovered that different orbitals required different amounts of energy for an ...
Chapter 2 - The Chemical Context of Life
... A covalent bond is the sharing of a pair of valence electrons by two atoms Covalent bonds can form between atoms of the same element or atoms of different elements In a covalent bond, the shared electrons count as part of each atom’s valence shell ...
... A covalent bond is the sharing of a pair of valence electrons by two atoms Covalent bonds can form between atoms of the same element or atoms of different elements In a covalent bond, the shared electrons count as part of each atom’s valence shell ...
Summaries of Review Topics for AP Chemistry
... (1) Element is a pure substance that cannot be broken down into other substances by chemical or physical means; it contains only one kind of atom. Elements are represented by the chemical symbols. The symbols for most elements consist of the first one or two letters of the name of the element. Ex: H ...
... (1) Element is a pure substance that cannot be broken down into other substances by chemical or physical means; it contains only one kind of atom. Elements are represented by the chemical symbols. The symbols for most elements consist of the first one or two letters of the name of the element. Ex: H ...
What are atoms?
... Alt solids, liquids, gases, and plasmas are made up of these tiny particles. Atoms are matter. One atom take up space—very, very, very little space. An atom also has mass—very, very, very little mass. How small is the atom? Atoms are so small that in just one drop of water, there are about six sexti ...
... Alt solids, liquids, gases, and plasmas are made up of these tiny particles. Atoms are matter. One atom take up space—very, very, very little space. An atom also has mass—very, very, very little mass. How small is the atom? Atoms are so small that in just one drop of water, there are about six sexti ...
Chapter_04_Structure_of_the_atom
... • How is the atomic number used in determining the identity of an atom? • What is an isotope? • Why are atomic masses not whole numbers? • Given its mass number and atomic number, how do you calculate the number of electrons, protons, and neutrons in an atom? ...
... • How is the atomic number used in determining the identity of an atom? • What is an isotope? • Why are atomic masses not whole numbers? • Given its mass number and atomic number, how do you calculate the number of electrons, protons, and neutrons in an atom? ...
Atomic Structure Atomic Structure
... Atoms are composed of protons, neutrons, and electrons, each of which is uniquely essential to the structure and function of the atom. The core of the atom is the nucleus, which consists of protons and neutrons. Most of the mass of an atom (about 99.9 percent) is in the nucleus, even though the nucl ...
... Atoms are composed of protons, neutrons, and electrons, each of which is uniquely essential to the structure and function of the atom. The core of the atom is the nucleus, which consists of protons and neutrons. Most of the mass of an atom (about 99.9 percent) is in the nucleus, even though the nucl ...
Chapter 4
... • Atoms are indivisible by chemical processes. – All atoms present at beginning are present at the end. – Atoms are not created or destroyed, just rearranged in chemical reactions. – Atoms of one element cannot change into atoms of another element. • Cannot turn lead into gold by a chemical reaction ...
... • Atoms are indivisible by chemical processes. – All atoms present at beginning are present at the end. – Atoms are not created or destroyed, just rearranged in chemical reactions. – Atoms of one element cannot change into atoms of another element. • Cannot turn lead into gold by a chemical reaction ...
Chapter 2 Notes
... Solution (a) The number of protons (22) is the atomic number of the element, which means this element is titanium (Ti). The mass number of this isotope is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the s ...
... Solution (a) The number of protons (22) is the atomic number of the element, which means this element is titanium (Ti). The mass number of this isotope is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the s ...
Atoms - ChemGod.com
... Law of Definite Proportions Joseph Proust made a series of mixtures of different elements and discovered that the ratio of the masses that reacted was always the same. For example if mixing hydrogen and oxygen to get water, he found that if you started with 16.0 g of oxygen, you needed 2.0 g of hyd ...
... Law of Definite Proportions Joseph Proust made a series of mixtures of different elements and discovered that the ratio of the masses that reacted was always the same. For example if mixing hydrogen and oxygen to get water, he found that if you started with 16.0 g of oxygen, you needed 2.0 g of hyd ...
Chem Test 2 - TeacherWeb
... ____ 21. Who was the man who lived from 460B.C.–370B.C. and was among the first to suggest the idea of atoms? a. Atomos c. Democritus b. Dalton d. Thomson ____ 22. Which of the following was NOT among Democritus’s ideas? a. Matter consists of tiny particles called atoms. b. Atoms are indivisible. c. ...
... ____ 21. Who was the man who lived from 460B.C.–370B.C. and was among the first to suggest the idea of atoms? a. Atomos c. Democritus b. Dalton d. Thomson ____ 22. Which of the following was NOT among Democritus’s ideas? a. Matter consists of tiny particles called atoms. b. Atoms are indivisible. c. ...
Of Atoms, Molecules & Ions I Sing
... Law of Definite Proportions Joseph Proust made a series of mixtures of different elements and discovered that the ratio of the masses that reacted was always the same. For example if mixing hydrogen and oxygen to get water, he found that if you started with 16.0 g of oxygen, you needed 2.0 g of hyd ...
... Law of Definite Proportions Joseph Proust made a series of mixtures of different elements and discovered that the ratio of the masses that reacted was always the same. For example if mixing hydrogen and oxygen to get water, he found that if you started with 16.0 g of oxygen, you needed 2.0 g of hyd ...
Unit 2: Atoms, Moles and The Periodic Table Notes (answers)
... atomos, which means indivisible. A Greek philosopher Democritus in 5th-century BC first suggested this concept). The Dalton’s Atomic Theory can be summarized as: ...
... atomos, which means indivisible. A Greek philosopher Democritus in 5th-century BC first suggested this concept). The Dalton’s Atomic Theory can be summarized as: ...
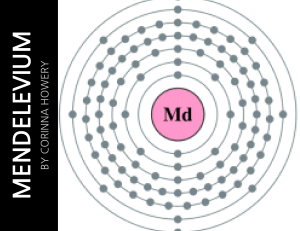
Mendelevium
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
All chemical equations must be balanced, that is, they must have the
... These reactions usually involve an element reacting with an ionic compound. The element can only replace the part of the compound that forms the same type of ion that the element forms : a metallic element will replace the positive part of the compound and a non-metal will replace the negative part. ...
... These reactions usually involve an element reacting with an ionic compound. The element can only replace the part of the compound that forms the same type of ion that the element forms : a metallic element will replace the positive part of the compound and a non-metal will replace the negative part. ...
chemistry intro and lesson 1
... You will have to make personal choices in your life. Should I use this chemical? What are the harmful effects it has on people and the environment? Are there other chemicals that are just as effective but safer to use? In society, you will influence public decisions in your community, your province ...
... You will have to make personal choices in your life. Should I use this chemical? What are the harmful effects it has on people and the environment? Are there other chemicals that are just as effective but safer to use? In society, you will influence public decisions in your community, your province ...
Chapter 23 (Section 3) Pregnancy, Birth, and
... *a. Extensions of PERIOD 6 (lanthanides) & PERIOD 7 (actinides) are below the periodic table *3. An element’s PROPERTIES can be predicted based on its LOCATION on the periodic table *a. to the right, left or bordering the ZIGZAG line indicates the “classification” of the ELEMENT as a METAL, NON-META ...
... *a. Extensions of PERIOD 6 (lanthanides) & PERIOD 7 (actinides) are below the periodic table *3. An element’s PROPERTIES can be predicted based on its LOCATION on the periodic table *a. to the right, left or bordering the ZIGZAG line indicates the “classification” of the ELEMENT as a METAL, NON-META ...
Chapter 6 Chemical Composition
... • Atoms have very tiny masses so scientists made a unit to avoid using very small numbers. • 1 atomic mass unit (amu) is 1/12 of the mass of a 12C atom • 1 amu = 1.66 10-24 g How many amu’s does a 12C atom weigh? How many grams does a 12C atom weigh? ...
... • Atoms have very tiny masses so scientists made a unit to avoid using very small numbers. • 1 atomic mass unit (amu) is 1/12 of the mass of a 12C atom • 1 amu = 1.66 10-24 g How many amu’s does a 12C atom weigh? How many grams does a 12C atom weigh? ...
Matter - cloudfront.net
... b. Is boiled water still water? 2. Can be reversible, or irreversible. 3. Chemical change - a change where a new form of matter is formed. – Rust, burn, decompose, ferment ...
... b. Is boiled water still water? 2. Can be reversible, or irreversible. 3. Chemical change - a change where a new form of matter is formed. – Rust, burn, decompose, ferment ...
Unit PowerPoint
... However, not all of the elements fit into the periodic table in order of increasing atomic mass. Mendeleev arranged tellurium and iodine and cobalt and nickel out of order by atomic mass so that they could be placed in the groups with which they shared similar chemical properties. (Mendeleev believe ...
... However, not all of the elements fit into the periodic table in order of increasing atomic mass. Mendeleev arranged tellurium and iodine and cobalt and nickel out of order by atomic mass so that they could be placed in the groups with which they shared similar chemical properties. (Mendeleev believe ...
Chapter 6 ppt
... • Protons, neutrons, and electrons make up atoms. • All atoms of a given element have the same number of protons in the nucleus. • Isotopes of an element differ by the number of neutrons in the nucleus. • Atomic mass is an average of the masses of all of the naturally occurring isotopes of an elemen ...
... • Protons, neutrons, and electrons make up atoms. • All atoms of a given element have the same number of protons in the nucleus. • Isotopes of an element differ by the number of neutrons in the nucleus. • Atomic mass is an average of the masses of all of the naturally occurring isotopes of an elemen ...
Building Atoms Unit Interactive Science Notebook III
... small dense nucleus of positive charge around which the negatively charged electrons circulate. His was the "solar system" model of an atom. Two years later the Danish physicist Niels Bohr (who had worked under Thomson at one time) developed Rutherford's model even further. He proposed that electron ...
... small dense nucleus of positive charge around which the negatively charged electrons circulate. His was the "solar system" model of an atom. Two years later the Danish physicist Niels Bohr (who had worked under Thomson at one time) developed Rutherford's model even further. He proposed that electron ...
Chem101 - Lecture 2 Elements Elements
... written in Exercise 2.44 and the factor-unit method to determine the following (The factorunit method is discussed in Study Skills 2.1.): a. The mass in grams of one bromine atom b. The number ot grams of carbon in 2.75 mol of carbon c. The total mass in grams of one-half Avogadro’s number of silver ...
... written in Exercise 2.44 and the factor-unit method to determine the following (The factorunit method is discussed in Study Skills 2.1.): a. The mass in grams of one bromine atom b. The number ot grams of carbon in 2.75 mol of carbon c. The total mass in grams of one-half Avogadro’s number of silver ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.