The Structure of an Atom
... Dalton’s Atomic Theory • All matter is composed of extremely small particles called atoms. • All atoms of a given element are identical. • Atoms cannot be created, divided into smaller particles, or destroyed. (This part proven wrong) • Different atoms combine in simple whole number ratios to form ...
... Dalton’s Atomic Theory • All matter is composed of extremely small particles called atoms. • All atoms of a given element are identical. • Atoms cannot be created, divided into smaller particles, or destroyed. (This part proven wrong) • Different atoms combine in simple whole number ratios to form ...
Name: Period:______ Table Number:______
... 51. Each element found on the periodic table of elements has a unique single letter (Hydrogen – H), two letter (Helium – He ) or three letter (Unnilquadiam – Unq) abbreviation which is called the CHEMICAL SYMBOL of that element. P. 83, Bill Nye the Science Guy Video 52. JONS BERZELIUS created the un ...
... 51. Each element found on the periodic table of elements has a unique single letter (Hydrogen – H), two letter (Helium – He ) or three letter (Unnilquadiam – Unq) abbreviation which is called the CHEMICAL SYMBOL of that element. P. 83, Bill Nye the Science Guy Video 52. JONS BERZELIUS created the un ...
Atoms, Isotopes, and Ions - Science Take-Out
... 1. Use the information on the periodic table to make a model of a hydrogen atom. Then make a hydrogen ion by removing the electron (blue chip) from the model. Draw your ion model. Use a “+” sign for each proton, an “n” for each neutron and a “–” sign for each electron. ...
... 1. Use the information on the periodic table to make a model of a hydrogen atom. Then make a hydrogen ion by removing the electron (blue chip) from the model. Draw your ion model. Use a “+” sign for each proton, an “n” for each neutron and a “–” sign for each electron. ...
zum_ic6e_ch04_lecture_ppt_23
... • Atoms of a given element are different from those of any other element. – Carbon atoms have different chemical and physical properties than sulfur atoms. ...
... • Atoms of a given element are different from those of any other element. – Carbon atoms have different chemical and physical properties than sulfur atoms. ...
Atom-notes-Powerpoint-upload
... In any like chemical compound, atoms always combine in definite proportions. ex. molecules of water always contain 2 hydrogen atoms and 1 oxygen atom ...
... In any like chemical compound, atoms always combine in definite proportions. ex. molecules of water always contain 2 hydrogen atoms and 1 oxygen atom ...
The representative Elements: Groups 1A – 4A
... • K+ is essential for nervous system; • K2O is important component in fertilizers; • KO2 is used in confined places like submarines for removal of CO2 and production of O2: 4KO2(s) + 2CO2(g) 2K2CO3(s) + 3O2(g) ...
... • K+ is essential for nervous system; • K2O is important component in fertilizers; • KO2 is used in confined places like submarines for removal of CO2 and production of O2: 4KO2(s) + 2CO2(g) 2K2CO3(s) + 3O2(g) ...
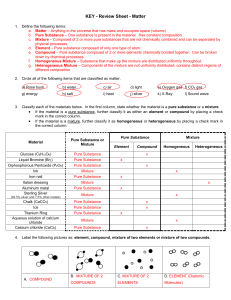
Matter Test Review Sheet
... KEY - Review Sheet - Matter 1. Define the following terms: a. Matter – Anything in the universe that has mass and occupies space (volume) b. Pure Substance – One substance is present in the material. Has constant composition. c. Mixture – Composed of 2 or more pure substances that are not chemically ...
... KEY - Review Sheet - Matter 1. Define the following terms: a. Matter – Anything in the universe that has mass and occupies space (volume) b. Pure Substance – One substance is present in the material. Has constant composition. c. Mixture – Composed of 2 or more pure substances that are not chemically ...
Chapter 2 - Phillips Scientific Methods
... Ionic Bonds: Transfer electrons • Atoms sometimes strip electrons from their bonding partners. • An example is the transfer of an electron from sodium to chlorine. • After the transfer of an electron, both atoms have charges. ...
... Ionic Bonds: Transfer electrons • Atoms sometimes strip electrons from their bonding partners. • An example is the transfer of an electron from sodium to chlorine. • After the transfer of an electron, both atoms have charges. ...
Earth’s Materials - Lower Hudson Regional Information Center
... Minerals are substances which are Naturally occurring Solid Inorganic Definite composition Definite atomic arrangement ...
... Minerals are substances which are Naturally occurring Solid Inorganic Definite composition Definite atomic arrangement ...
3.Masses of individual atoms
... • Chemistry is a quantitative science. Atoms of elements differ from one another not only in composition(number of protons ,electrons, neutrons),but also in mass, Chemical formulas of compounds tell us not only the atom ratios in which elements are present but also the mass ratios. ...
... • Chemistry is a quantitative science. Atoms of elements differ from one another not only in composition(number of protons ,electrons, neutrons),but also in mass, Chemical formulas of compounds tell us not only the atom ratios in which elements are present but also the mass ratios. ...
Elements Compounds
... Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance in each atom. The Na becomes (Na+) and the Cl becomes (Cl-), charged particles or ions. ...
... Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance in each atom. The Na becomes (Na+) and the Cl becomes (Cl-), charged particles or ions. ...
Atomic Masses: Counting Atoms by Weighing
... as we needed to know the mass of the individual jelly beans. Recall from Chapter 3 that the atoms of a given element exist as isotopes. The isotopes of carbon are 126C, 136C, and 146C. Any sample of carbon contains a mixture of these isotopes, always in the same proportions. Each of these isotopes h ...
... as we needed to know the mass of the individual jelly beans. Recall from Chapter 3 that the atoms of a given element exist as isotopes. The isotopes of carbon are 126C, 136C, and 146C. Any sample of carbon contains a mixture of these isotopes, always in the same proportions. Each of these isotopes h ...
atom - geraldinescience
... • In nature, most elements are found as mixtures of isotopes. Usually, the relative abundance of each isotope is constant. –Ex. In a banana, 93.26% is potassium-39, 6.73% is potassium-41 and 0.01% is potassium40. In another banana or in a different source of potassium, the percentage composition of ...
... • In nature, most elements are found as mixtures of isotopes. Usually, the relative abundance of each isotope is constant. –Ex. In a banana, 93.26% is potassium-39, 6.73% is potassium-41 and 0.01% is potassium40. In another banana or in a different source of potassium, the percentage composition of ...
PSN Chapter 14 Multi-format Test.tst
... 3. Solid elements described as dull, brittle, and poor electrical and thermal conductors are most likely metals. _________________________ ...
... 3. Solid elements described as dull, brittle, and poor electrical and thermal conductors are most likely metals. _________________________ ...
Mass Relationships in Chemical Reactions
... 2. Change the numbers in front of the formulas (coefficients) to make the number of atoms of each element the same on both sides of the equation. Do not change the subscripts. ...
... 2. Change the numbers in front of the formulas (coefficients) to make the number of atoms of each element the same on both sides of the equation. Do not change the subscripts. ...
chemistry in the 8th grade
... chemical change is involved in changing physical state. A chemical change only occurs when chemical bonds are broken, formed, or both. When matter is in the solid state, the particles (atoms or molecules) that make up the matter are very close together. These particles can vibrate around fixed posit ...
... chemical change is involved in changing physical state. A chemical change only occurs when chemical bonds are broken, formed, or both. When matter is in the solid state, the particles (atoms or molecules) that make up the matter are very close together. These particles can vibrate around fixed posit ...
practice exercise - Needham.K12.ma.us
... ending, the acid is given a hydro- prefix and an -ic ending: hydrocyanic acid. Only water solutions of HCN are referred to as hydrocyanic acid: The pure compound, which is a gas under normal conditions, is called hydrogen cyanide. Both hydrocyanic acid and hydrogen cyanide are extremely toxic. (b) B ...
... ending, the acid is given a hydro- prefix and an -ic ending: hydrocyanic acid. Only water solutions of HCN are referred to as hydrocyanic acid: The pure compound, which is a gas under normal conditions, is called hydrogen cyanide. Both hydrocyanic acid and hydrogen cyanide are extremely toxic. (b) B ...
Topic 2.1 Atomic Structure Notes Topic 2.1 Atomic
... charged nucleus. NOS: 1.Evidence and improvements in instrumentation – alpha particles were used in the development of the nuclear model of the atom that was first proposed by Rutherford. (1.8) 2.Paradigm shifts- the subatomic particle theory of matter represents a paradigm shift in science that occ ...
... charged nucleus. NOS: 1.Evidence and improvements in instrumentation – alpha particles were used in the development of the nuclear model of the atom that was first proposed by Rutherford. (1.8) 2.Paradigm shifts- the subatomic particle theory of matter represents a paradigm shift in science that occ ...
Atoms and Elements - Melvin R. Kantz, Ph.D.
... oxygen for every 1.00 g of carbon carbon dioxide contains 2.67 g of oxygen for every 1.00 g of carbon since there are twice as many oxygen atoms per carbon atom in carbon dioxide than in carbon monoxide, the oxygen mass ratio should be 2 mass of oxygen that combines with 1 g of carbon in carbon diox ...
... oxygen for every 1.00 g of carbon carbon dioxide contains 2.67 g of oxygen for every 1.00 g of carbon since there are twice as many oxygen atoms per carbon atom in carbon dioxide than in carbon monoxide, the oxygen mass ratio should be 2 mass of oxygen that combines with 1 g of carbon in carbon diox ...
Medical Chemistry Lecture By : Asst. Lect. Tariq-H-Almgheer
... has a diameter of 2.8 X 10'8 cm. This is so small that a length of 1 cm corresponds to 36 million uranium atoms placed side to side! Atoms are small, but the nucleus is even smaller; it occupies only a small part of the total volume of the atom. If one could magnify the size of an atom so that its n ...
... has a diameter of 2.8 X 10'8 cm. This is so small that a length of 1 cm corresponds to 36 million uranium atoms placed side to side! Atoms are small, but the nucleus is even smaller; it occupies only a small part of the total volume of the atom. If one could magnify the size of an atom so that its n ...
Year 9 Chemical Sciences Program Term 3 Course 2 2017
... Text: Oxford Science 9, Helen Silvester Chemical Sciences Content Descriptions All matter is made of atoms that are composed of protons, neutrons and electrons; natural radioactivity arises from the decay of nuclei in atoms. (ACSSU177) describing and modelling the structure of atoms in terms of th ...
... Text: Oxford Science 9, Helen Silvester Chemical Sciences Content Descriptions All matter is made of atoms that are composed of protons, neutrons and electrons; natural radioactivity arises from the decay of nuclei in atoms. (ACSSU177) describing and modelling the structure of atoms in terms of th ...
atom - WordPress.com
... • Atoms are electrically neutral because they contain equal numbers of protons and electrons. • A neutron is electrically neutral. ...
... • Atoms are electrically neutral because they contain equal numbers of protons and electrons. • A neutron is electrically neutral. ...
Atoms - RCSD
... called subatomic particle: protons, electrons & neutrons. 2)A given element can have atoms with different masses (isotopes). ...
... called subatomic particle: protons, electrons & neutrons. 2)A given element can have atoms with different masses (isotopes). ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.