Power Point over chemistry
... It means that our climate stays much more stable than it would if there were less water on Earth. TAKS Need to Know ...
... It means that our climate stays much more stable than it would if there were less water on Earth. TAKS Need to Know ...
Dalton`s Atomic Theory
... smaller particles, created, or destroyed. • All atoms of the same element are alike and have the same mass. Atoms of different elements are different and have different masses. ...
... smaller particles, created, or destroyed. • All atoms of the same element are alike and have the same mass. Atoms of different elements are different and have different masses. ...
Chapter 2 Atoms and Elements Modern Atomic theory
... Isotopes Isotopes- Atoms of the same element with same number of protons but different number of neutrons e.g for all carbon atoms Z= 6 (C-12 and C-13) The observed mass is a weighted average of the weights of all the naturally occurring atoms Natural abundance- The percentage of one isotope in an e ...
... Isotopes Isotopes- Atoms of the same element with same number of protons but different number of neutrons e.g for all carbon atoms Z= 6 (C-12 and C-13) The observed mass is a weighted average of the weights of all the naturally occurring atoms Natural abundance- The percentage of one isotope in an e ...
Atomic Timeline There are small, negatively charged particles inside
... Atomic Timeline The table below contains a number of statements connected to major discoveries in the development of the atomic theory. ...
... Atomic Timeline The table below contains a number of statements connected to major discoveries in the development of the atomic theory. ...
Atomic Theory of Matter
... atoms of two or more elements chemically combined in fixed proportions. The relative numbers of any two kinds of atoms in a compound occur in simple ratios. Water, for example, consists of hydrogen and oxygen in a 2 to 1 ratio. ...
... atoms of two or more elements chemically combined in fixed proportions. The relative numbers of any two kinds of atoms in a compound occur in simple ratios. Water, for example, consists of hydrogen and oxygen in a 2 to 1 ratio. ...
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
... 3. Determine the number of neutrons, protons and electrons from the proton number and the nucleon number and vice versa. 4. Construct the atomic structure. ...
... 3. Determine the number of neutrons, protons and electrons from the proton number and the nucleon number and vice versa. 4. Construct the atomic structure. ...
what`s ahead - Al Akhawayn University
... because electrons contribute only a very small fraction of an atom’s mass they probably were responsible for an equally small fraction of the atom’s size. He proposed that the atom consisted of a uniform positive sphere of matter in which the electrons were embedded like raisins in a pudding or seed ...
... because electrons contribute only a very small fraction of an atom’s mass they probably were responsible for an equally small fraction of the atom’s size. He proposed that the atom consisted of a uniform positive sphere of matter in which the electrons were embedded like raisins in a pudding or seed ...
BASIC CHEMICAL CONCEPTS
... fractional distillation of a large quantity, repeated many times). The components obtained by these methods have very similar properties except in their ability to absorb or emit high-energy radiation. For example, water can be separated into components having densities (at 25 °C) of 0⋅997 and 1⋅104 ...
... fractional distillation of a large quantity, repeated many times). The components obtained by these methods have very similar properties except in their ability to absorb or emit high-energy radiation. For example, water can be separated into components having densities (at 25 °C) of 0⋅997 and 1⋅104 ...
Atoms, Molecules, and Ions
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
2 - TestBankTop
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
regents chemistry midterm - irondequoit 2014_entire exam w key
... ***Use only the elements from the above table (a) to answer questions b), c), d), & e) b) The element with the highest first ionization energy? [1pt] c) The element that will form a negative ion in a compound? [1pt] d) The element with the greatest number of unpaired electrons in its Lewis dot (elec ...
... ***Use only the elements from the above table (a) to answer questions b), c), d), & e) b) The element with the highest first ionization energy? [1pt] c) The element that will form a negative ion in a compound? [1pt] d) The element with the greatest number of unpaired electrons in its Lewis dot (elec ...
Matter and Atoms
... An Electron Cloud Drawings of an atom often show electrons circling the nucleus like planets orbiting the Sun. Scientists have conducted experiments that show the movement of electrons is more complex than this. The modern idea of an atom is called the electron-cloud model. An electron cloud is the ...
... An Electron Cloud Drawings of an atom often show electrons circling the nucleus like planets orbiting the Sun. Scientists have conducted experiments that show the movement of electrons is more complex than this. The modern idea of an atom is called the electron-cloud model. An electron cloud is the ...
Atomic Structure
... atomic structure structure of an atom chemistry - each atom consists of a very small nucleus composed of protons and neutrons which is encircled by moving electrons some of the important properties depend on, chem4kids com atoms structure - chem4kids com this tutorial introduces atomic structure in ...
... atomic structure structure of an atom chemistry - each atom consists of a very small nucleus composed of protons and neutrons which is encircled by moving electrons some of the important properties depend on, chem4kids com atoms structure - chem4kids com this tutorial introduces atomic structure in ...
minerals - Tulane University
... crystals but is deflected by the atoms that make up the crystals. The image produced and collected on film, can be used to determine the struture. The method is know as X-ray diffraction. Crystal structure depends on the conditions under which the mineral forms. Polymorphs are minerals with the same ...
... crystals but is deflected by the atoms that make up the crystals. The image produced and collected on film, can be used to determine the struture. The method is know as X-ray diffraction. Crystal structure depends on the conditions under which the mineral forms. Polymorphs are minerals with the same ...
File
... The three states of matter are solid, liquid, and gas. The way that you convert from one state to another is by increasing or decreasing temperature. Temperature is a measure of the amount of heat energy something has. If something has a high temperature is has a lot of energy and the particles move ...
... The three states of matter are solid, liquid, and gas. The way that you convert from one state to another is by increasing or decreasing temperature. Temperature is a measure of the amount of heat energy something has. If something has a high temperature is has a lot of energy and the particles move ...
Atoms and the Periodic Table Atoms and the Periodic Table
... Metals bend as they are pressed into thin, flat sheets during the coin-making process. All metals share some similarities, but each metal has its own unique chemical and physical properties. The unique building shown on the opposite page is the Guggenheim Museum in Bilboa, Spain. This art museum is ...
... Metals bend as they are pressed into thin, flat sheets during the coin-making process. All metals share some similarities, but each metal has its own unique chemical and physical properties. The unique building shown on the opposite page is the Guggenheim Museum in Bilboa, Spain. This art museum is ...
FREE Sample Here
... 41. The atomic mass of naturally occurring fluorine, which exists in nature as a single isotope, is listed as 18.9984 u. This means that a. all fluorine atoms found in nature have a mass which is 18.9984/12.000 times as great as that of a 12C atom. b. all fluorine atoms found in nature have a mass w ...
... 41. The atomic mass of naturally occurring fluorine, which exists in nature as a single isotope, is listed as 18.9984 u. This means that a. all fluorine atoms found in nature have a mass which is 18.9984/12.000 times as great as that of a 12C atom. b. all fluorine atoms found in nature have a mass w ...
FREE Sample Here
... 41. The atomic mass of naturally occurring fluorine, which exists in nature as a single isotope, is listed as 18.9984 u. This means that a. all fluorine atoms found in nature have a mass which is 18.9984/12.000 times as great as that of a 12C atom. b. all fluorine atoms found in nature have a mass w ...
... 41. The atomic mass of naturally occurring fluorine, which exists in nature as a single isotope, is listed as 18.9984 u. This means that a. all fluorine atoms found in nature have a mass which is 18.9984/12.000 times as great as that of a 12C atom. b. all fluorine atoms found in nature have a mass w ...
Dear Students, Welcome to AP Chemistry, a little early. We will have
... 3. Molecules: larger units in which two or more atoms are joined together a. The way in which matter hehaves depends on the atoms present and the manner in which they are comhined ...
... 3. Molecules: larger units in which two or more atoms are joined together a. The way in which matter hehaves depends on the atoms present and the manner in which they are comhined ...
FoundationsofChemistryppt
... • A chemical property is observed only as a material undergoes chemical change and changes identity. • Signs of possible chemical change include bubbles, energy change, and change in odor or color. • Chemical equations show the reactants and products of a chemical reaction and that mass is conserve ...
... • A chemical property is observed only as a material undergoes chemical change and changes identity. • Signs of possible chemical change include bubbles, energy change, and change in odor or color. • Chemical equations show the reactants and products of a chemical reaction and that mass is conserve ...
Introductory Chemistry: A Foundation FOURTH EDITION by Steven
... – the volume is about 1/10 trillionth the volume of the atom The nucleus is essentially the entire mass of the atom The nucleus is positively charged – the amount of positive charge of the nucleus balances the negative charge of the electrons The electrons move around in the empty space of the ...
... – the volume is about 1/10 trillionth the volume of the atom The nucleus is essentially the entire mass of the atom The nucleus is positively charged – the amount of positive charge of the nucleus balances the negative charge of the electrons The electrons move around in the empty space of the ...
Introductory Chemistry, 2nd Edition Nivaldo Tro
... 1. Each element is composed of tiny, indestructible particles called atoms. 2. All atoms of an element are identical in mass, size, and chemical properties. Atoms of different element are different. 3. Compounds are atoms joined together in small whole number ratios. 4. Chemical reactions rearrange ...
... 1. Each element is composed of tiny, indestructible particles called atoms. 2. All atoms of an element are identical in mass, size, and chemical properties. Atoms of different element are different. 3. Compounds are atoms joined together in small whole number ratios. 4. Chemical reactions rearrange ...
File
... more dense than water. Specific gravity has no units. It is simply a number. This is because the units cancel out when the densities of the two substances are compared. ...
... more dense than water. Specific gravity has no units. It is simply a number. This is because the units cancel out when the densities of the two substances are compared. ...
Chapter 12
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
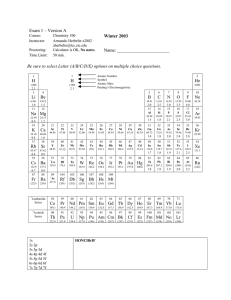
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.