Stars Answers - Science Skool!
... 8. What happens to massive stars after the red super giant stage? Outer layers are thrown into space which scatters gas and dust into space and distributes the elements throughout space. The core left behind forms a neutron star or black hole if sufficient mass is left behind 9. Why do scientists be ...
... 8. What happens to massive stars after the red super giant stage? Outer layers are thrown into space which scatters gas and dust into space and distributes the elements throughout space. The core left behind forms a neutron star or black hole if sufficient mass is left behind 9. Why do scientists be ...
大爆炸---宇宙的起源 - 中正大學化學系
... body thermal energy coming from all parts of the sky. The radiation is isotropic to roughly one part in 100,000. As the universe expanded, adiabatic cooling caused the plasma to lose energy until it became favorable for electrons to combine with protons, forming hydrogen atoms. This recombination ev ...
... body thermal energy coming from all parts of the sky. The radiation is isotropic to roughly one part in 100,000. As the universe expanded, adiabatic cooling caused the plasma to lose energy until it became favorable for electrons to combine with protons, forming hydrogen atoms. This recombination ev ...
Presentation - Science in the News
... What did we start with and how do we get what we have now? The Big Bang and Stellar ...
... What did we start with and how do we get what we have now? The Big Bang and Stellar ...
Unit_Phys_2_nuclear_fusion__fission
... form a larger one. b) Nuclear fusion is the process by which energy is released in stars. c) Stars form when enough dust and gas from space is pulled together by gravitational attraction. Smaller masses may also form and be attracted by a larger mass to become planets. d) During the ‘main sequence’ ...
... form a larger one. b) Nuclear fusion is the process by which energy is released in stars. c) Stars form when enough dust and gas from space is pulled together by gravitational attraction. Smaller masses may also form and be attracted by a larger mass to become planets. d) During the ‘main sequence’ ...
Groupmeeting_shshiu_20090803_nuclearx
... The rapid proton capture process consists of consecutive proton captures onto nuclei to produce heavier elements. The possible sites suggested for the rp-process are binary systems. One star is a compact object, the other one is low mass black hole or neutron star. The rp-process is constrained by a ...
... The rapid proton capture process consists of consecutive proton captures onto nuclei to produce heavier elements. The possible sites suggested for the rp-process are binary systems. One star is a compact object, the other one is low mass black hole or neutron star. The rp-process is constrained by a ...
Lecture 16 - Yet More Evolution of Stars
... • Solar mass star produce elements up to Carbon and Oxygen – these are ejected into planetary nebula and then recycled into new stars and planets • Supernova produce all of the heavier elements – Elements up to Iron can be produced by fusion – Elements heavier than iron are produced by the neutrons ...
... • Solar mass star produce elements up to Carbon and Oxygen – these are ejected into planetary nebula and then recycled into new stars and planets • Supernova produce all of the heavier elements – Elements up to Iron can be produced by fusion – Elements heavier than iron are produced by the neutrons ...
Ejecta from neutron star mergers and the role of
... IKP-Theorie, TU-Darmstadt, Darmstadt, Germany ...
... IKP-Theorie, TU-Darmstadt, Darmstadt, Germany ...
sep04 neutrinos - Charles J Horowitz
... ½… The mass of the neutrons should be of the same order as the electron mass and in any event not larger than 0.01 proton masses. The continuous beta spectrum would then be explained by the assumption that in beta decay a neutron is emitted in addition to the electron such that the sum of the energi ...
... ½… The mass of the neutrons should be of the same order as the electron mass and in any event not larger than 0.01 proton masses. The continuous beta spectrum would then be explained by the assumption that in beta decay a neutron is emitted in addition to the electron such that the sum of the energi ...
Facts - GreenSpirit
... Eventually, there is so much iron that the inner core contracts, the centre melts down. Under the enormous pressure, the elements dissolve, neutrinos flood out leaving no energy at the centre of the star. ...
... Eventually, there is so much iron that the inner core contracts, the centre melts down. Under the enormous pressure, the elements dissolve, neutrinos flood out leaving no energy at the centre of the star. ...
Document
... Milky Way. The star, known in modern times as Sanduleak 69202, was a blue supergiant 25 times more massive than the Sun. Such explosions distribute all the common elements such as Oxygen, Carbon, Nitrogen, Calcium and Iron into interstellar space where they enrich clouds of Hydrogen and Helium that ...
... Milky Way. The star, known in modern times as Sanduleak 69202, was a blue supergiant 25 times more massive than the Sun. Such explosions distribute all the common elements such as Oxygen, Carbon, Nitrogen, Calcium and Iron into interstellar space where they enrich clouds of Hydrogen and Helium that ...
Star Arsenic, Star Selenium The Big Bang produced lots of hydrogen
... The Big Bang produced lots of hydrogen and helium and a smidgen of lithium. All heavier elements found on the periodic table have been produced by stars over the last 13.7 billion years. Astronomers analyze starlight to determine the chemical makeup of stars, the origin of the elements, the ages of ...
... The Big Bang produced lots of hydrogen and helium and a smidgen of lithium. All heavier elements found on the periodic table have been produced by stars over the last 13.7 billion years. Astronomers analyze starlight to determine the chemical makeup of stars, the origin of the elements, the ages of ...
Nineteenth lecture
... HOME. (Ever wonder why all the planets except for Pluto rotate in the same direction around the sun, in a flat plane?) ...
... HOME. (Ever wonder why all the planets except for Pluto rotate in the same direction around the sun, in a flat plane?) ...
Where Did the Elements Come From?
... • Most atoms in living things come from six elements: carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur • It is believed all 93 elements were created in the centers of stars billions of years ago. ...
... • Most atoms in living things come from six elements: carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur • It is believed all 93 elements were created in the centers of stars billions of years ago. ...
Lecture 1: Nucleosynthesis, solar composition, chondrites, volatility
... The rest of the elements are produced by 2 pathways: s-process (slow): addition of neutrons to nuclei one at a time (only stable elements can be made) r-process (rapid): addition of neutron at a rapid rate so as to bridge areas of nuclear instability (only in supernovas and accounts for about half o ...
... The rest of the elements are produced by 2 pathways: s-process (slow): addition of neutrons to nuclei one at a time (only stable elements can be made) r-process (rapid): addition of neutron at a rapid rate so as to bridge areas of nuclear instability (only in supernovas and accounts for about half o ...
Unit 2: The Sun and Other Stars
... by the star (which we call a supernova). While scientists still don’t completely understand the process, the collision of neutron stars and supernova explosions appear to be the “creators” of the heavier elements. ...
... by the star (which we call a supernova). While scientists still don’t completely understand the process, the collision of neutron stars and supernova explosions appear to be the “creators” of the heavier elements. ...
Yes, we are all star dust. Even Gary!
... • Small Stellar Nucleosynthesis (fusion in Solar-type Stars) = more Helium … plus Beryllium, Boron and Carbon … As carbon life-forms, we are indeed star dust! • Massive Stellar Nuc. (fusion in massive stars) = Oxygen to Iron • Supernovae (exploding massive stars) = all other naturally occurring elem ...
... • Small Stellar Nucleosynthesis (fusion in Solar-type Stars) = more Helium … plus Beryllium, Boron and Carbon … As carbon life-forms, we are indeed star dust! • Massive Stellar Nuc. (fusion in massive stars) = Oxygen to Iron • Supernovae (exploding massive stars) = all other naturally occurring elem ...
The Universe Inside of You: Where do the atoms in your body come
... We know the amount of stuff in the Solar System because we have data from meteorites and the sun's photosphere (outer shell). The sun has 99% of the mass of the solar system – so it is important to understand its composition. ...
... We know the amount of stuff in the Solar System because we have data from meteorites and the sun's photosphere (outer shell). The sun has 99% of the mass of the solar system – so it is important to understand its composition. ...
Origin of the Elements Essay
... and faster rates. Since the heaviest element created in a star by nuclear fusion reactions is iron, a large iron core eventually forms at the center of everything. At this point, gravity becomes overwhelming, the core collapses, and an explosion occurs, during which outer layers of gas and heavy ele ...
... and faster rates. Since the heaviest element created in a star by nuclear fusion reactions is iron, a large iron core eventually forms at the center of everything. At this point, gravity becomes overwhelming, the core collapses, and an explosion occurs, during which outer layers of gas and heavy ele ...
Star Factories: Nuclear Fusion and the Creation of the Elements
... What is it about the environment inside stars that makes nuclear fusion possible? High temperature; and high density. ...
... What is it about the environment inside stars that makes nuclear fusion possible? High temperature; and high density. ...
Life2
... trace (.000,000,001) of lithium. No elements produced with an atomic number higher than lithium !!!!!! Heterogeneous universe from a homogenous one ? Quantum fluctuations in early universe produced “framework” of galaxy formation. Attracted gas and dark matter that coalesced to form first galaxies a ...
... trace (.000,000,001) of lithium. No elements produced with an atomic number higher than lithium !!!!!! Heterogeneous universe from a homogenous one ? Quantum fluctuations in early universe produced “framework” of galaxy formation. Attracted gas and dark matter that coalesced to form first galaxies a ...
The Evolution of the Universe: from Cosmic Soup to Earth
... Big Bang: The initial point from which the universe began developing approximately 13.7 billion years ago. Cosmic Microwave Background: the leftover energy that can be detected from the initial Big Bang. Element: a pure chemical substance which is determined by the number of protons in its nu ...
... Big Bang: The initial point from which the universe began developing approximately 13.7 billion years ago. Cosmic Microwave Background: the leftover energy that can be detected from the initial Big Bang. Element: a pure chemical substance which is determined by the number of protons in its nu ...
Elements from Stardust
... temperature in the sun, nuclei of atoms are squeezed together and they collide. Nuclear fusion combines smaller nuclei into larger nuclei—making bigger and heavier atoms. ...
... temperature in the sun, nuclei of atoms are squeezed together and they collide. Nuclear fusion combines smaller nuclei into larger nuclei—making bigger and heavier atoms. ...
SMP Quiz Session 1
... You have two atoms, one has 6 protons, 6 neutrons, and 6 electrons and the other has 6 protons, 8 neutrons, and 6 electrons. Which of the following best describes the difference between them? The ...
... You have two atoms, one has 6 protons, 6 neutrons, and 6 electrons and the other has 6 protons, 8 neutrons, and 6 electrons. Which of the following best describes the difference between them? The ...
Nucleosynthesis
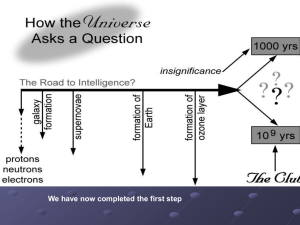
Nucleosynthesis is the process that creates new atomic nuclei from pre-existing nucleons, primarily protons and neutrons. The first nuclei were formed about three minutes after the Big Bang, through the process called Big Bang nucleosynthesis. It was then that hydrogen and helium formed to become the content of the first stars, and this primeval process is responsible for the present hydrogen/helium ratio of the cosmos.With the formation of stars, heavier nuclei were created from hydrogen and helium by stellar nucleosynthesis, a process that continues today. Some of these elements, particularly those lighter than iron, continue to be delivered to the interstellar medium when low mass stars eject their outer envelope before they collapse to form white dwarfs. The remains of their ejected mass form the planetary nebulae observable throughout our galaxy.Supernova nucleosynthesis within exploding stars by fusing carbon and oxygen is responsible for the abundances of elements between magnesium (atomic number 12) and nickel (atomic number 28). Supernova nucleosynthesis is also thought to be responsible for the creation of rarer elements heavier than iron and nickel, in the last few seconds of a type II supernova event. The synthesis of these heavier elements absorbs energy (endothermic) as they are created, from the energy produced during the supernova explosion. Some of those elements are created from the absorption of multiple neutrons (the R process) in the period of a few seconds during the explosion. The elements formed in supernovas include the heaviest elements known, such as the long-lived elements uranium and thorium.Cosmic ray spallation, caused when cosmic rays impact the interstellar medium and fragment larger atomic species, is a significant source of the lighter nuclei, particularly 3He, 9Be and 10,11B, that are not created by stellar nucleosynthesis.In addition to the fusion processes responsible for the growing abundances of elements in the universe, a few minor natural processes continue to produce very small numbers of new nuclides on Earth. These nuclides contribute little to their abundances, but may account for the presence of specific new nuclei. These nuclides are produced via radiogenesis (decay) of long-lived, heavy, primordial radionuclides such as uranium and thorium. Cosmic ray bombardment of elements on Earth also contribute to the presence of rare, short-lived atomic species called cosmogenic nuclides.