Nuclear Physics - fission, fusion, and the stars
... to undergo fission- although the frequency of oscillations inside the nucleus is ~ 1021 per second. This means a tunneling probability ~ 10-38 – a very small number. Actually all nuclei except Fe decay, but Kaiser Wilhelm Institute (Berlin) in 1938 only a few do it fast enough to be seen, except for ...
... to undergo fission- although the frequency of oscillations inside the nucleus is ~ 1021 per second. This means a tunneling probability ~ 10-38 – a very small number. Actually all nuclei except Fe decay, but Kaiser Wilhelm Institute (Berlin) in 1938 only a few do it fast enough to be seen, except for ...
File - Physical Science
... Nuclear Fusion and Nucleosynthesis Stars are giant nuclear reactors. In the center of stars, atoms are taken apart by tremendous atomic collisions that alter the atomic structure and release an enormous amount of energy. This makes stars hot and bright. Nuclear fusion is an atomic reaction that fuel ...
... Nuclear Fusion and Nucleosynthesis Stars are giant nuclear reactors. In the center of stars, atoms are taken apart by tremendous atomic collisions that alter the atomic structure and release an enormous amount of energy. This makes stars hot and bright. Nuclear fusion is an atomic reaction that fuel ...
PRESENTATION: Evolution of the elements through the lifecycles of
... -Picture striking a rubber ball on the ground with a hammer. It will compress, but eventually expand and throw off the hammer, if the blow (the solar mass) is not strong enough to compress it into a black hole. -The Iron atoms are compressed to the point that the electrons are collapsing into the pr ...
... -Picture striking a rubber ball on the ground with a hammer. It will compress, but eventually expand and throw off the hammer, if the blow (the solar mass) is not strong enough to compress it into a black hole. -The Iron atoms are compressed to the point that the electrons are collapsing into the pr ...
Elements and Isotopes - University of California, Berkeley
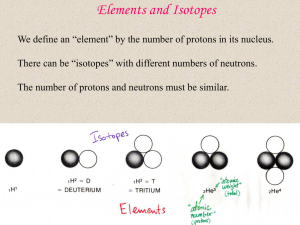
... the elements without them § the strong nuclear force holds nuclei together even though protons repel each other § it works like velcro : only unlike particles can stick Result : the elements have almost equal numbers of protons & neutrons ...
... the elements without them § the strong nuclear force holds nuclei together even though protons repel each other § it works like velcro : only unlike particles can stick Result : the elements have almost equal numbers of protons & neutrons ...
Chapter 4 Vocabulary
... the moment in time when the universe started to expand out of an extremely hot, dense state ...
... the moment in time when the universe started to expand out of an extremely hot, dense state ...
The Origin of the Elements - Indiana University Astronomy
... Large stars also fuse hydrogen into helium, and helium into carbon But their larger masses lead to higher temperatures, which allow fusion of carbon into magnesium, etc. ...
... Large stars also fuse hydrogen into helium, and helium into carbon But their larger masses lead to higher temperatures, which allow fusion of carbon into magnesium, etc. ...
Stellar Nucleosynthesis
... • For massive (more than ten solar masses, > 10 MSun) stars, direct nuclear burning continues with the production of oxygen, neon, magnesium, silicon and so on, cumulating in the synthesis of iron, the heaviest element possible through direct nuclear burning. • The other heavy elements, from yttrium ...
... • For massive (more than ten solar masses, > 10 MSun) stars, direct nuclear burning continues with the production of oxygen, neon, magnesium, silicon and so on, cumulating in the synthesis of iron, the heaviest element possible through direct nuclear burning. • The other heavy elements, from yttrium ...
Where do elements come from?
... held together by gravity • Everything is made from the elements created in massive stars. Stars are huge factories producing the elements found in the Universe. ...
... held together by gravity • Everything is made from the elements created in massive stars. Stars are huge factories producing the elements found in the Universe. ...
stellar_explosions - UT Austin (Astronomy)
... This absorbs even more thermal (heat) energy, so the core collapses even faster! Gravity is having its way… The protons combine with electrons to give neutrons and neutrinos (which escape immediately—later we’ll see that they have been accidentally observed in ...
... This absorbs even more thermal (heat) energy, so the core collapses even faster! Gravity is having its way… The protons combine with electrons to give neutrons and neutrinos (which escape immediately—later we’ll see that they have been accidentally observed in ...
N.2 Formation of Mass
... 2. Strong Force: the force between the protons and neutrons of the nucleus of atoms. It holds the nucleus together. 3. Weak Force: An attraction between hydrogen atoms. Causes nuclear fusion between hydrogen atoms, it is the reason the sun burns. ...
... 2. Strong Force: the force between the protons and neutrons of the nucleus of atoms. It holds the nucleus together. 3. Weak Force: An attraction between hydrogen atoms. Causes nuclear fusion between hydrogen atoms, it is the reason the sun burns. ...
PHY320 Glossary of Terms - The University of Sheffield
... before the formation of stars and galaxies. The so-called "Big bang" production of 2H, 3He, 4He and 7Li. Measurements of the abundances of these nuclear species provides a valuable crosscheck on the standard model of the early evolution of the Universe. Red Giants are stars which have just left the ...
... before the formation of stars and galaxies. The so-called "Big bang" production of 2H, 3He, 4He and 7Li. Measurements of the abundances of these nuclear species provides a valuable crosscheck on the standard model of the early evolution of the Universe. Red Giants are stars which have just left the ...
This chapter has a brief overview of astronomical topics that we will
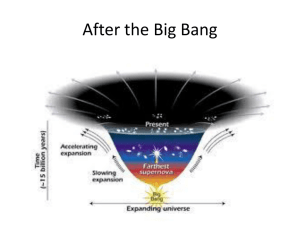
... nothing before (as there was no time) and it was not an explosion in space (since space was created here). Why do we believe this? We see the galaxies flying apart from each other, the farthest moving faster. Since we see the more distant galaxies as they were long ago, this seems to imply an explos ...
... nothing before (as there was no time) and it was not an explosion in space (since space was created here). Why do we believe this? We see the galaxies flying apart from each other, the farthest moving faster. Since we see the more distant galaxies as they were long ago, this seems to imply an explos ...
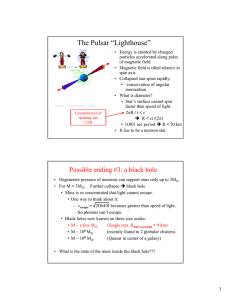
The Pulsar “Lighthouse”
... • Degenerate pressure of neutrons can support stars only up to 3M • For M > 3M: Further collapse Î black hole • Mass is so concentrated that light cannot escape. • One way to think about it: – vescape = 2GM/R becomes greater than speed of light. ...
... • Degenerate pressure of neutrons can support stars only up to 3M • For M > 3M: Further collapse Î black hole • Mass is so concentrated that light cannot escape. • One way to think about it: – vescape = 2GM/R becomes greater than speed of light. ...
Initial Evolution-The Main Sequence
... Two scenarios: stars with as mass less than 8 times the mass of the sun and stars with a mass greater than 8 times the mass of the sun. ...
... Two scenarios: stars with as mass less than 8 times the mass of the sun and stars with a mass greater than 8 times the mass of the sun. ...
We Are All Stardust: Nuclear Physics in the Cosmos
... neutron capture (r) process: neutron captures on rapid timescale (~1 s) in a hot (1 billion K), dense environment ( >1020 neutrons cm-3) The other half are produced in the slow neutron capture process ...
... neutron capture (r) process: neutron captures on rapid timescale (~1 s) in a hot (1 billion K), dense environment ( >1020 neutrons cm-3) The other half are produced in the slow neutron capture process ...
Chapter1
... How were the elements created? Were they created at the same time as the universe (in the Big Bang)? created subsequently? ...
... How were the elements created? Were they created at the same time as the universe (in the Big Bang)? created subsequently? ...
South Pasadena • Chemistry Name 8 • Nuclear Chemistry Period
... For more than 50 years, scientists have been studying the links between chemical elements and how they are created. Elements lighter than iron are formed from successive nuclear fusion reactions, beginning with hydrogen atoms. All stars form iron in their cores. ...
... For more than 50 years, scientists have been studying the links between chemical elements and how they are created. Elements lighter than iron are formed from successive nuclear fusion reactions, beginning with hydrogen atoms. All stars form iron in their cores. ...
supernova!
... For every 90 Hydrogen atoms (a total mass of 90), there are 9 Heliums (with a total mass of 9x4 = 36); and so on for the other elements. Thus Hydrogen is about 70% of the total mass, the rest (~25% or so) being mostly Helium. This justifies my earlier statements that the universe is ~2/3-3/4 Hydroge ...
... For every 90 Hydrogen atoms (a total mass of 90), there are 9 Heliums (with a total mass of 9x4 = 36); and so on for the other elements. Thus Hydrogen is about 70% of the total mass, the rest (~25% or so) being mostly Helium. This justifies my earlier statements that the universe is ~2/3-3/4 Hydroge ...
April 2013
... A bit of background Each chemical element is characterised by the number of positively charged protons within the nucleus of each of its atoms or ions. This number is the element’s atomic number. The nucleus also contains a number of uncharged neutrons, which, in the cases of most elements, is equal ...
... A bit of background Each chemical element is characterised by the number of positively charged protons within the nucleus of each of its atoms or ions. This number is the element’s atomic number. The nucleus also contains a number of uncharged neutrons, which, in the cases of most elements, is equal ...
Astrobiology 101
... The heaviest elements are created in supernovae, the fantastic death of supergiant stars. As the core of the supergiant becomes saturated with iron, its pressure and temperature increase. Eventually, the blackbody radiation from the core produces gamma rays powerful enough to break apart the iron a ...
... The heaviest elements are created in supernovae, the fantastic death of supergiant stars. As the core of the supergiant becomes saturated with iron, its pressure and temperature increase. Eventually, the blackbody radiation from the core produces gamma rays powerful enough to break apart the iron a ...
The big bang left the universe with its first atoms
... The heaviest elements are created in supernovae, the fantastic death of supergiant stars. As the core of the supergiant becomes saturated with iron, its pressure and temperature increase. Eventually, the blackbody radiation from the core produces gamma rays powerful enough to break apart the iron a ...
... The heaviest elements are created in supernovae, the fantastic death of supergiant stars. As the core of the supergiant becomes saturated with iron, its pressure and temperature increase. Eventually, the blackbody radiation from the core produces gamma rays powerful enough to break apart the iron a ...
Where Did All The Elements Come From??
... The heaviest elements are created in supernovae, the fantastic death of supergiant stars. As the core of the supergiant becomes saturated with iron, its pressure and temperature increase. Eventually, the blackbody radiation from the core produces gamma rays powerful enough to break apart the iron a ...
... The heaviest elements are created in supernovae, the fantastic death of supergiant stars. As the core of the supergiant becomes saturated with iron, its pressure and temperature increase. Eventually, the blackbody radiation from the core produces gamma rays powerful enough to break apart the iron a ...
大爆炸---宇宙的起源
... body thermal energy coming from all parts of the sky. The radiation is isotropic to roughly one part in 100,000. As the universe expanded, adiabatic cooling caused the plasma to lose energy until it became favorable for electrons to combine with protons, forming hydrogen atoms. This recombination ev ...
... body thermal energy coming from all parts of the sky. The radiation is isotropic to roughly one part in 100,000. As the universe expanded, adiabatic cooling caused the plasma to lose energy until it became favorable for electrons to combine with protons, forming hydrogen atoms. This recombination ev ...
Nucleosynthesis
Nucleosynthesis is the process that creates new atomic nuclei from pre-existing nucleons, primarily protons and neutrons. The first nuclei were formed about three minutes after the Big Bang, through the process called Big Bang nucleosynthesis. It was then that hydrogen and helium formed to become the content of the first stars, and this primeval process is responsible for the present hydrogen/helium ratio of the cosmos.With the formation of stars, heavier nuclei were created from hydrogen and helium by stellar nucleosynthesis, a process that continues today. Some of these elements, particularly those lighter than iron, continue to be delivered to the interstellar medium when low mass stars eject their outer envelope before they collapse to form white dwarfs. The remains of their ejected mass form the planetary nebulae observable throughout our galaxy.Supernova nucleosynthesis within exploding stars by fusing carbon and oxygen is responsible for the abundances of elements between magnesium (atomic number 12) and nickel (atomic number 28). Supernova nucleosynthesis is also thought to be responsible for the creation of rarer elements heavier than iron and nickel, in the last few seconds of a type II supernova event. The synthesis of these heavier elements absorbs energy (endothermic) as they are created, from the energy produced during the supernova explosion. Some of those elements are created from the absorption of multiple neutrons (the R process) in the period of a few seconds during the explosion. The elements formed in supernovas include the heaviest elements known, such as the long-lived elements uranium and thorium.Cosmic ray spallation, caused when cosmic rays impact the interstellar medium and fragment larger atomic species, is a significant source of the lighter nuclei, particularly 3He, 9Be and 10,11B, that are not created by stellar nucleosynthesis.In addition to the fusion processes responsible for the growing abundances of elements in the universe, a few minor natural processes continue to produce very small numbers of new nuclides on Earth. These nuclides contribute little to their abundances, but may account for the presence of specific new nuclei. These nuclides are produced via radiogenesis (decay) of long-lived, heavy, primordial radionuclides such as uranium and thorium. Cosmic ray bombardment of elements on Earth also contribute to the presence of rare, short-lived atomic species called cosmogenic nuclides.