ENZYMES: PROPERTIES OF B
... a molecule and the type of environment in which a molecule is found (gaseous, aqueous, a lipid bilayer, etc.). An important feature of molecular modeling programs is the ability to change aspects of the molecule being studied. For example, it is possible to add, remove, and alter specific atoms and ...
... a molecule and the type of environment in which a molecule is found (gaseous, aqueous, a lipid bilayer, etc.). An important feature of molecular modeling programs is the ability to change aspects of the molecule being studied. For example, it is possible to add, remove, and alter specific atoms and ...
Bio1A - Lec 9 slides File
... • Competitive inhibitors bind to the active site of an enzyme, competing with the substrate • Noncompetitive inhibitors bind to another part of an enzyme, causing the enzyme to change shape and making the active site less effective Allosteric Affect – Examples of inhibitors include toxins, poisons ...
... • Competitive inhibitors bind to the active site of an enzyme, competing with the substrate • Noncompetitive inhibitors bind to another part of an enzyme, causing the enzyme to change shape and making the active site less effective Allosteric Affect – Examples of inhibitors include toxins, poisons ...
Cellular Respiration - MF011 General Biology 2 (May 2011 Semester)
... Living cells require energy from outside sources Some animals, such as the giant panda, obtain energy by eating plants, and some animals feed on other organisms that eat plants ...
... Living cells require energy from outside sources Some animals, such as the giant panda, obtain energy by eating plants, and some animals feed on other organisms that eat plants ...
pyruvate dehydrogenase complex
... • TCA cycle (a.k.a Krebs cycle or citric acid cycle) plays several roles in metabolism • It is the final pathway where oxidative metabolism of CHO’s, aa’s & fatty acids converge, their C skeletons being converted to CO2 & H2O. This oxidation provides energy for production of majority of ATP. • The c ...
... • TCA cycle (a.k.a Krebs cycle or citric acid cycle) plays several roles in metabolism • It is the final pathway where oxidative metabolism of CHO’s, aa’s & fatty acids converge, their C skeletons being converted to CO2 & H2O. This oxidation provides energy for production of majority of ATP. • The c ...
Engineering of cyclodextrin glycosyltransferase reaction and
... need for a solvent recovery process [15]. The availability of K- and Q-cyclodextrins is thus rather limited at present; consequently, there is a great demand for a process that could produce these cyclodextrins economically. Also the processes used for L-cyclodextrin production are not ideal, since ...
... need for a solvent recovery process [15]. The availability of K- and Q-cyclodextrins is thus rather limited at present; consequently, there is a great demand for a process that could produce these cyclodextrins economically. Also the processes used for L-cyclodextrin production are not ideal, since ...
CH 2 -CH 2 -CH 2 -CH 2
... Of course to completely degrade palmitoyl CoA would require 7 rounds of beta oxidation: ...
... Of course to completely degrade palmitoyl CoA would require 7 rounds of beta oxidation: ...
Some Structural and Kinetic Aspects of L
... Domain A has a classical (α/β)8 topology, domain B is with an irregular β barrel and domain C can be characterized with α/β organization. If there is an additional N-terminal domain, it is usually formed by helix-turn-helix motif (Mattevi et al., 1996; Valentini et al., 2000). The active site of pyr ...
... Domain A has a classical (α/β)8 topology, domain B is with an irregular β barrel and domain C can be characterized with α/β organization. If there is an additional N-terminal domain, it is usually formed by helix-turn-helix motif (Mattevi et al., 1996; Valentini et al., 2000). The active site of pyr ...
7 rounds of beta oxidation
... Fatty acids (FA) from the diet or from the degradation of triglycerides stored in adipose cells are broken down further to smaller molecules to completely metabolize them and therefore release energy. This process of catabolism of FA includes three major parts: ...
... Fatty acids (FA) from the diet or from the degradation of triglycerides stored in adipose cells are broken down further to smaller molecules to completely metabolize them and therefore release energy. This process of catabolism of FA includes three major parts: ...
Document
... • Enzymes have extraordinary catalytic power, often far greater than those non-biological catalysts. • Enzymes often have a high degree of specificity for their substrates. • Enzymes are often regulatory. • Enzymes usually work under very mild conditions of temperature and pH. • The substance acted ...
... • Enzymes have extraordinary catalytic power, often far greater than those non-biological catalysts. • Enzymes often have a high degree of specificity for their substrates. • Enzymes are often regulatory. • Enzymes usually work under very mild conditions of temperature and pH. • The substance acted ...
Requires Rubisco
... At 5 x 10 -5 atm, CO2 fixation rate = photorespiration rate. However, plants living in hot climates need to conserve water, which requires them to use low CO2 concentration (water is used in rubisco reaction!) The disadvantage of C4 plants is extra ATP used and a more complex pathway http://methanog ...
... At 5 x 10 -5 atm, CO2 fixation rate = photorespiration rate. However, plants living in hot climates need to conserve water, which requires them to use low CO2 concentration (water is used in rubisco reaction!) The disadvantage of C4 plants is extra ATP used and a more complex pathway http://methanog ...
Understanding nature`s catalytic toolkit
... cost to this orientation. By pre-arranging the active site, this entropic cost is paid for when the enzyme folds or the substrate binds [18–20], rather than during catalysis, and thus it is beneficial to the enzyme. The individual contribution of a single residue that functions only to orientate ano ...
... cost to this orientation. By pre-arranging the active site, this entropic cost is paid for when the enzyme folds or the substrate binds [18–20], rather than during catalysis, and thus it is beneficial to the enzyme. The individual contribution of a single residue that functions only to orientate ano ...
Enzymes Enzymes are characterized by
... Commonly named by adding the suffix -ase to the name of the substrate or a phrase describing the catalytic reaction. Urease - catalyzes the hyrolysis of urea Alcohol dehydrogenase - catalyzes the oxidation of alcohols to aldehydes ...
... Commonly named by adding the suffix -ase to the name of the substrate or a phrase describing the catalytic reaction. Urease - catalyzes the hyrolysis of urea Alcohol dehydrogenase - catalyzes the oxidation of alcohols to aldehydes ...
8)Discuss the roles of cofactors and coenzymes in enzyme activity.
... General features of enzymes: 1) Most of the enzymes are proteins, however, recent work has shown that there are RNA molecules which show catalytic activity (RNA enzymes) 2) Enzymes increase the rate of reactions but do not influence the equilibrium. 3) Enzymes exhibit high degree of specificity for ...
... General features of enzymes: 1) Most of the enzymes are proteins, however, recent work has shown that there are RNA molecules which show catalytic activity (RNA enzymes) 2) Enzymes increase the rate of reactions but do not influence the equilibrium. 3) Enzymes exhibit high degree of specificity for ...
Adenosine triphosphate Adenosine triphosphate Adenosine
... molecules of the reduced coenzyme NADH, and one molecule of the reduced coenzyme FADH2. Both of these latter molecules are recycled to their oxidized states (NAD+ and FAD, respectively) via the electron transport chain, which generates additional ATP by oxidative phosphorylation. The oxidation of an ...
... molecules of the reduced coenzyme NADH, and one molecule of the reduced coenzyme FADH2. Both of these latter molecules are recycled to their oxidized states (NAD+ and FAD, respectively) via the electron transport chain, which generates additional ATP by oxidative phosphorylation. The oxidation of an ...
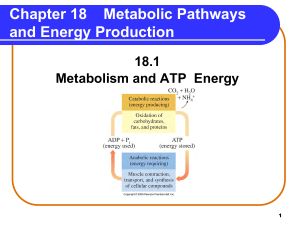
Metabolic Pathways and Energy Production
... Electron Transport and ATP In electron transport, sufficient energy is provided from • NADH (Complex I) oxidation for 3 ATPs. NADH + 3 ADP + 3Pi NAD+ + 3 ATP • FADH2 (Complex II) oxidation for 2 ATPs. FADH2 + 2 ADP + 2Pi FAD + 2 ATP ...
... Electron Transport and ATP In electron transport, sufficient energy is provided from • NADH (Complex I) oxidation for 3 ATPs. NADH + 3 ADP + 3Pi NAD+ + 3 ATP • FADH2 (Complex II) oxidation for 2 ATPs. FADH2 + 2 ADP + 2Pi FAD + 2 ATP ...
world journal of pharmaceutical research
... As shown in Figure 3, in the glycolysis step, glucose is broken down to two molecules of pyruvate via a series of enzymatic reactions that occur in the cytoplasm (anaerobic). The breakdown of glucose releases enough energy to immediately give a net gain of two ATP molecules by substrate-level ATP sy ...
... As shown in Figure 3, in the glycolysis step, glucose is broken down to two molecules of pyruvate via a series of enzymatic reactions that occur in the cytoplasm (anaerobic). The breakdown of glucose releases enough energy to immediately give a net gain of two ATP molecules by substrate-level ATP sy ...
Answers - U of L Class Index
... Glutamate forms when the amino group from any of the amino acids is transferred to _-ketoglutarate. In the process of oxidation deamination, glutamate converts that amino group to ammonium ion. ...
... Glutamate forms when the amino group from any of the amino acids is transferred to _-ketoglutarate. In the process of oxidation deamination, glutamate converts that amino group to ammonium ion. ...
Luciferase

Luciferase is a generic term for the class of oxidative enzymes used in bioluminescence and is distinct from a photoprotein. The name is derived from Lucifer, the root of which means 'light-bearer' (lucem ferre). One example is the firefly luciferase (EC 1.13.12.7) from the firefly Photinus pyralis. ""Firefly luciferase"" as a laboratory reagent often refers to P. pyralis luciferase although recombinant luciferases from several other species of fireflies are also commercially available.