EL Study Notes
... helium nuclei. Energy is released in these nuclear fusion reactions. Although the production of each helium nucleus only releases 4 x 10-12J, the Sun makes 1038 helium nuclei per second. As a result the Sun’s energy output is enormous. It has enough hydrogen for around 5 billion years when it will e ...
... helium nuclei. Energy is released in these nuclear fusion reactions. Although the production of each helium nucleus only releases 4 x 10-12J, the Sun makes 1038 helium nuclei per second. As a result the Sun’s energy output is enormous. It has enough hydrogen for around 5 billion years when it will e ...
10 Chemistry
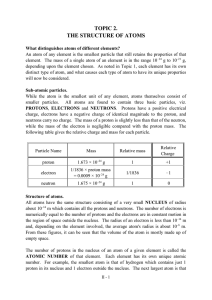
... 1. Atomic number (Z) – number of protons in the nucleus of an atom – neutral atom: number of protons = number of electrons ...
... 1. Atomic number (Z) – number of protons in the nucleus of an atom – neutral atom: number of protons = number of electrons ...
Atoms—Basic Units of Matter
... was interested in the physical properties of gases. Like Lavoisier and Proust, Dalton made careful measurements of starting materi als and products in a number of chemical reactions. To record his results accurately, he invented symbols for the known elements. As Figure 5 shows, these are more compl ...
... was interested in the physical properties of gases. Like Lavoisier and Proust, Dalton made careful measurements of starting materi als and products in a number of chemical reactions. To record his results accurately, he invented symbols for the known elements. As Figure 5 shows, these are more compl ...
B - Cloudfront.net
... charge to be quite repulsive. In fact, if it weren’t for the “strong force” holding the nucleons together, the protons would just fly out of the nucleus altogether, disgusted by each other’s positivity. ...
... charge to be quite repulsive. In fact, if it weren’t for the “strong force” holding the nucleons together, the protons would just fly out of the nucleus altogether, disgusted by each other’s positivity. ...
The Periodic Table
... Element 120 will have electron configuration [Rn] 7s26d107p6 8s2 ... it has two valence shell electrons in the 8s orbital which puts it in group two. The 'standard' ion charge for a group two element is 2+ so the formula for the oxide of element 120 should be ElO. ...
... Element 120 will have electron configuration [Rn] 7s26d107p6 8s2 ... it has two valence shell electrons in the 8s orbital which puts it in group two. The 'standard' ion charge for a group two element is 2+ so the formula for the oxide of element 120 should be ElO. ...
Unit 2 - Solon City Schools
... 3)______________________ Mass is neither created nor destroyed. 4)______________________ Value represented by # of protons + # of neutrons. 5)______________________ He founded the “mathematical” model of the atom. The electrons are located in the electron cloud. 6)______________________ The one elem ...
... 3)______________________ Mass is neither created nor destroyed. 4)______________________ Value represented by # of protons + # of neutrons. 5)______________________ He founded the “mathematical” model of the atom. The electrons are located in the electron cloud. 6)______________________ The one elem ...
Periods and Blocks of the Periodic Table
... • Mendeleev noticed that when the elements were arranged in order of increasing atomic mass, certain similarities in their chemical properties appeared at regular intervals. • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar properties were g ...
... • Mendeleev noticed that when the elements were arranged in order of increasing atomic mass, certain similarities in their chemical properties appeared at regular intervals. • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar properties were g ...
Chapter 5
... • Mendeleev noticed that when the elements were arranged in order of increasing atomic mass, certain similarities in their chemical properties appeared at regular intervals. • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar properties were g ...
... • Mendeleev noticed that when the elements were arranged in order of increasing atomic mass, certain similarities in their chemical properties appeared at regular intervals. • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar properties were g ...
Unit 2 Atomic Theories and Structures Packet
... 3)______________________ Mass is neither created nor destroyed. 4)______________________ Value represented by # of protons + # of neutrons. 5)______________________ He founded the “mathematical” model of the atom. The electrons are located in the electron cloud. 6)______________________ The one elem ...
... 3)______________________ Mass is neither created nor destroyed. 4)______________________ Value represented by # of protons + # of neutrons. 5)______________________ He founded the “mathematical” model of the atom. The electrons are located in the electron cloud. 6)______________________ The one elem ...
Document
... b) Chlorine contains 17 protons and 18.5 neutrons c) Chlorine normally exists in an excited state, and so it weighs more than 35. d) The chlorine was not pure when its atomic mass was measured. e) Chlorine, as found in nature, contains a mixture of the isotopes 35Cl and 37Cl, in such proportions as ...
... b) Chlorine contains 17 protons and 18.5 neutrons c) Chlorine normally exists in an excited state, and so it weighs more than 35. d) The chlorine was not pure when its atomic mass was measured. e) Chlorine, as found in nature, contains a mixture of the isotopes 35Cl and 37Cl, in such proportions as ...
File
... For electrons, protons and neutrons – write down the charge on each particle, the mass of each particle, and where they are found in the atom. Electron = neg. charge found outside nucleus, almost zero mass Protons = pos. charge, in nucleus, 1 amu Neutron = no charge, in nucleus, 1 amu ...
... For electrons, protons and neutrons – write down the charge on each particle, the mass of each particle, and where they are found in the atom. Electron = neg. charge found outside nucleus, almost zero mass Protons = pos. charge, in nucleus, 1 amu Neutron = no charge, in nucleus, 1 amu ...
U N I 1. laboratory tools and chemistry techniques.
... • formation of a new solid • formation of a gas • the release of energy as heat or light • an increase or decrease in the mass of material • a change in the texture of a material 2. Possible answer: The copper would always be present throughout the series of reactions. The copper could be in a diff ...
... • formation of a new solid • formation of a gas • the release of energy as heat or light • an increase or decrease in the mass of material • a change in the texture of a material 2. Possible answer: The copper would always be present throughout the series of reactions. The copper could be in a diff ...
TOPIC 2. THE STRUCTURE OF ATOMS
... must be other laws which govern the behaviour of electrons in atoms. Models to explain this will be presented later in the year as part of all first year chemistry courses, but the results of certain experimental evidence presented here is independent of those models. Experiments show that the elect ...
... must be other laws which govern the behaviour of electrons in atoms. Models to explain this will be presented later in the year as part of all first year chemistry courses, but the results of certain experimental evidence presented here is independent of those models. Experiments show that the elect ...
Chapter 2 "Elements, Atoms, and the Periodic Table"
... the water dropped, causing fish to die in large numbers. This process, called eutrophication, is considered a negative environmental impact. Today, many detergents are made without phosphorus so the detrimental effects of eutrophication are minimized. You may even see statements to that effect on de ...
... the water dropped, causing fish to die in large numbers. This process, called eutrophication, is considered a negative environmental impact. Today, many detergents are made without phosphorus so the detrimental effects of eutrophication are minimized. You may even see statements to that effect on de ...
TOPIC 2. THE STRUCTURE OF ATOMS
... nucleus, each orbit being characterised by its own associated energy and average distance from the nucleus. These orbits are grouped into ENERGY LEVELS or SHELLS and numbered 1, 2, 3,... outwards from the nucleus. The number of orbits available is strictly limited. Electrons occupying orbits closest ...
... nucleus, each orbit being characterised by its own associated energy and average distance from the nucleus. These orbits are grouped into ENERGY LEVELS or SHELLS and numbered 1, 2, 3,... outwards from the nucleus. The number of orbits available is strictly limited. Electrons occupying orbits closest ...
Structure of the Atom
... experiment is carried out using a foil of a metal other than gold? Answer: If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin f ...
... experiment is carried out using a foil of a metal other than gold? Answer: If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin f ...
Unit 2.4 Understanding the Elements Listed on the Periodic Table
... important so that we can compare experimental data from one lab to another and make sure we all are talking about the same thing. ...
... important so that we can compare experimental data from one lab to another and make sure we all are talking about the same thing. ...
1 Structure of Atom - Viva Online Learning
... Rutherford observed that most of the α-particles passed straight through the foil without any deflection from their path. However, to his surprise many particles were deflected at very large angles. As the mass of α-particles is about 8000 times that of an electron, it was evident that the force wh ...
... Rutherford observed that most of the α-particles passed straight through the foil without any deflection from their path. However, to his surprise many particles were deflected at very large angles. As the mass of α-particles is about 8000 times that of an electron, it was evident that the force wh ...
Topic 2.1 Atomic Structure Notes Topic 2.1 Atomic
... Consider an atom that has 9 protons, 9 electrons, and 10 neutrons. What is its atomic number, atomic mass, name, and symbol? Z=9 (atomic number = # protons) A=19 (atomic mass = protons + neutrons) Fluorine-19 (name and mass) ...
... Consider an atom that has 9 protons, 9 electrons, and 10 neutrons. What is its atomic number, atomic mass, name, and symbol? Z=9 (atomic number = # protons) A=19 (atomic mass = protons + neutrons) Fluorine-19 (name and mass) ...
CS3_Ch 6 - Leon County Schools
... • An atom is a building block of matter. An element is matter made of only one type of atom. A compound is a substance that contains two or more elements. • A heterogeneous mixture is not a solution because the substances that make up a heterogeneous mixture are not evenly mixed. The substances that ...
... • An atom is a building block of matter. An element is matter made of only one type of atom. A compound is a substance that contains two or more elements. • A heterogeneous mixture is not a solution because the substances that make up a heterogeneous mixture are not evenly mixed. The substances that ...
Atoms and Molecules
... the elements hadn’t been discovered yet. Although Mendeleev is credited with developing the Periodic Table, many scientists contributed to its development. The organization was done in such a way that as new elements have been discovered, they fit right where they are supposed to on the Periodic Tab ...
... the elements hadn’t been discovered yet. Although Mendeleev is credited with developing the Periodic Table, many scientists contributed to its development. The organization was done in such a way that as new elements have been discovered, they fit right where they are supposed to on the Periodic Tab ...
Ch 6 PPT - Blountstown Middle School
... • An atom is a building block of matter. An element is matter made of only one type of atom. A compound is a substance that contains two or more elements. • A heterogeneous mixture is not a solution because the substances that make up a heterogeneous mixture are not evenly mixed. The substances that ...
... • An atom is a building block of matter. An element is matter made of only one type of atom. A compound is a substance that contains two or more elements. • A heterogeneous mixture is not a solution because the substances that make up a heterogeneous mixture are not evenly mixed. The substances that ...
Lesson 1
... • An atom is a building block of matter. An element is matter made of only one type of atom. A compound is a substance that contains two or more elements. • A heterogeneous mixture is not a solution because the substances that make up a heterogeneous mixture are not evenly mixed. The substances that ...
... • An atom is a building block of matter. An element is matter made of only one type of atom. A compound is a substance that contains two or more elements. • A heterogeneous mixture is not a solution because the substances that make up a heterogeneous mixture are not evenly mixed. The substances that ...
TOPIC 2. THE STRUCTURE OF ATOMS
... first group of elements in Table 2 (the alkali metals) also have just one more electron than a noble gas atom, and they all behave as does sodium in that relatively little energy is needed to form their +1 cations Li+, Na+, K+, Rb+ and Cs+. Each of these cations has the same electron arrangement as ...
... first group of elements in Table 2 (the alkali metals) also have just one more electron than a noble gas atom, and they all behave as does sodium in that relatively little energy is needed to form their +1 cations Li+, Na+, K+, Rb+ and Cs+. Each of these cations has the same electron arrangement as ...