Name: : :______ (2) Science 8: Ch 6.1 Development of the Atomic
... Based on his results, Rutherford determined that most of the matter in an atom is found where? Rutherford revised the atomic theory in 1911 and proposed there was a dense part of an atom found in the center. What did he call this? And because the positively charge particles in the beam were pushed ...
... Based on his results, Rutherford determined that most of the matter in an atom is found where? Rutherford revised the atomic theory in 1911 and proposed there was a dense part of an atom found in the center. What did he call this? And because the positively charge particles in the beam were pushed ...
Metals scheme
... The structure of matter: Describe the structure of the atoms of different elements. Distinguish between an element and a compound, and a pure substance and a mixture at particle level. Chemistry and society: Link the properties of different groups of substances to the way they are used in soci ...
... The structure of matter: Describe the structure of the atoms of different elements. Distinguish between an element and a compound, and a pure substance and a mixture at particle level. Chemistry and society: Link the properties of different groups of substances to the way they are used in soci ...
Chemistry Outcomes - hrsbstaff.ednet.ns.ca
... properties for a missing element of hypothetical element of the same family. Distinguish between atoms and ions (cations and anions and their formation) ...
... properties for a missing element of hypothetical element of the same family. Distinguish between atoms and ions (cations and anions and their formation) ...
Section 3
... Democritus – was a fourth century B.C. Greek philosopher who proposed that the universe was made up of tiny particles called atoms. The word atom was derived from the Greek word Atomos, “meaning indivisible or indestructible”. John Dalton’s Atomic Theory •Every element is made of tiny, unique partic ...
... Democritus – was a fourth century B.C. Greek philosopher who proposed that the universe was made up of tiny particles called atoms. The word atom was derived from the Greek word Atomos, “meaning indivisible or indestructible”. John Dalton’s Atomic Theory •Every element is made of tiny, unique partic ...
CHEM 1305 - HCC Learning Web
... -------5. How many neutrons are in the nucleus of an atom of silver-107? A) 47 B) 60 C) 107 D) 154 ------6. What is the name of the family of elements in Group IIA/ 2? A) Alkali metals B) Alkaline earth metals C) Halogens D) Noble gases -------7. Which fifth period representative element has the hig ...
... -------5. How many neutrons are in the nucleus of an atom of silver-107? A) 47 B) 60 C) 107 D) 154 ------6. What is the name of the family of elements in Group IIA/ 2? A) Alkali metals B) Alkaline earth metals C) Halogens D) Noble gases -------7. Which fifth period representative element has the hig ...
Solid - burgess
... atomic number and is read from left to right. 2. Each vertical column is called a group or family. All the elements in a family have the same number of valence electrons 3. Each horizontal row is called a period. All elements in the same period have the same ending energy level (where electrons are ...
... atomic number and is read from left to right. 2. Each vertical column is called a group or family. All the elements in a family have the same number of valence electrons 3. Each horizontal row is called a period. All elements in the same period have the same ending energy level (where electrons are ...
Year 11 Chemistry Balancing Equations
... Looking over your electron configurations, are there any elements above that have similar valence electron configurations to those of other elements? If so, list below the elements that are similar (in terms of valence electrons) and state the similarity for each of the groups. ...
... Looking over your electron configurations, are there any elements above that have similar valence electron configurations to those of other elements? If so, list below the elements that are similar (in terms of valence electrons) and state the similarity for each of the groups. ...
Atoms - Grass Range Science
... nature contains a mixtures of isotopes. • Therefore, the atomic mass is an average of the atomic masses of all the existing isotopes of a particular element. ...
... nature contains a mixtures of isotopes. • Therefore, the atomic mass is an average of the atomic masses of all the existing isotopes of a particular element. ...
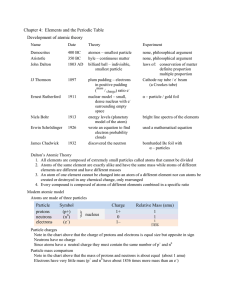
Chapter 4: Elements and the Periodic Table Development of atomic
... patterns repeated (Li, Na, and K for example) so he started a new column to match properties He noticed two important things: A few element properties did not match exactly in order of atomic mass – he moved these He noticed three missing elements and predicted their properties All three of these el ...
... patterns repeated (Li, Na, and K for example) so he started a new column to match properties He noticed two important things: A few element properties did not match exactly in order of atomic mass – he moved these He noticed three missing elements and predicted their properties All three of these el ...
- Chapter 7 - Periodic Properties of the Elements
... Zeff = Z − S where Z is the atomic number and S is a screening constant, usually close to the number of inner electrons. ...
... Zeff = Z − S where Z is the atomic number and S is a screening constant, usually close to the number of inner electrons. ...
Electron Configuration, Noble Gas Configuration
... Teach students how to write the electron configurations for main group elements, Finish with Electron Configuration Battleship; WS - Electron Configurations (or do H thru K, Ga, Br, Te, Cs, Pb, Rn); Quiz – Electron Configurations ...
... Teach students how to write the electron configurations for main group elements, Finish with Electron Configuration Battleship; WS - Electron Configurations (or do H thru K, Ga, Br, Te, Cs, Pb, Rn); Quiz – Electron Configurations ...
Chapter 3 Discovering the atom and subatomic particles (History of
... but has the same (reverse) electric charge as an electron. The number of protons each atom of a given element contains is called atomic number. Neutron (中子) is another subatomic particle in nucleus, having the similar mass as the proton but electrically neutral. It has very important role in holding ...
... but has the same (reverse) electric charge as an electron. The number of protons each atom of a given element contains is called atomic number. Neutron (中子) is another subatomic particle in nucleus, having the similar mass as the proton but electrically neutral. It has very important role in holding ...
Chemistry
... K. Used the cathode ray tube to ID negative particles which led to the plum pudding model of the atom. L. Theorized that all matter is continuous and not made of particles. M. Stated that equal volumes of different gases have equal numbers of molecules. N. Named the electron after its discovery by T ...
... K. Used the cathode ray tube to ID negative particles which led to the plum pudding model of the atom. L. Theorized that all matter is continuous and not made of particles. M. Stated that equal volumes of different gases have equal numbers of molecules. N. Named the electron after its discovery by T ...
Using your periodic table (9/30-10/6) File
... • Neils Bohr – Bohr model with orbitals for electrons around the nucleus, like a planetary system ...
... • Neils Bohr – Bohr model with orbitals for electrons around the nucleus, like a planetary system ...
Reactions I Can..
... 5. Identify key sections of the periodic table including orbital blocks, metal vs. nonmetal, alkali metals, alkaline earth metals, halogens, noble gases, lanthanide series, actinide series, transition elements, inner-transition elements, and transuranic elements. 6. Identify the phase (solid, liquid ...
... 5. Identify key sections of the periodic table including orbital blocks, metal vs. nonmetal, alkali metals, alkaline earth metals, halogens, noble gases, lanthanide series, actinide series, transition elements, inner-transition elements, and transuranic elements. 6. Identify the phase (solid, liquid ...
Atoms
... 2. List two of the problems with Mendeleev’s table. 3. Identify the key contribution of Henry Moseley to the modern periodic table 4. Identify groups and periods on the Periodic Table 5. Identify key sections of the periodic table including orbital blocks, metal vs. nonmetal, alkali metals, alkaline ...
... 2. List two of the problems with Mendeleev’s table. 3. Identify the key contribution of Henry Moseley to the modern periodic table 4. Identify groups and periods on the Periodic Table 5. Identify key sections of the periodic table including orbital blocks, metal vs. nonmetal, alkali metals, alkaline ...
Atoms, Molecules and Ions In This Chapter
... – An element is composed of identical atoms with fixed, identical properties and masses. – Compounds are formed by the combination of atoms of 2 or more different elements in a fixed whole number ratio. – A chemical reaction involves a combination, separation or rearrangement of atoms. Atoms are nei ...
... – An element is composed of identical atoms with fixed, identical properties and masses. – Compounds are formed by the combination of atoms of 2 or more different elements in a fixed whole number ratio. – A chemical reaction involves a combination, separation or rearrangement of atoms. Atoms are nei ...
Parts of the Atom
... Example: The element hydrogen has three isotopes. It exists in nature 99.41% of the time as Hydrogen-1, 0.4% of the time as Hydrogen-2 and 0.19% of the time as Hydrogen – 3. What is the average atomic mass of hydrogen? ...
... Example: The element hydrogen has three isotopes. It exists in nature 99.41% of the time as Hydrogen-1, 0.4% of the time as Hydrogen-2 and 0.19% of the time as Hydrogen – 3. What is the average atomic mass of hydrogen? ...
8th Grade Chapter 3 Study Guide
... of copper. Pennies made later have a core of zinc covered by a thin layer of copper. Nickels contain about 25% copper and 75% nickel. Classify each type of coin in terms of elements, compounds, homogeneous mixtures, and heterogeneous mixtures. 34. Give four examples of properties of nonmetals that a ...
... of copper. Pennies made later have a core of zinc covered by a thin layer of copper. Nickels contain about 25% copper and 75% nickel. Classify each type of coin in terms of elements, compounds, homogeneous mixtures, and heterogeneous mixtures. 34. Give four examples of properties of nonmetals that a ...
Periodic Table notes
... b) more mass and is about the same in size c) more mass and is smaller in size d) none of the above ...
... b) more mass and is about the same in size c) more mass and is smaller in size d) none of the above ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.