Units 1
... Chemistry is the branch of science that studies the composition and properties of matter and the changes that matter undergoes ...
... Chemistry is the branch of science that studies the composition and properties of matter and the changes that matter undergoes ...
AP Biology
... 11. Describe the structure of an amino acid. What is the alpha carbon? Do any of the amino acids contain alpha carbons that are not asymmetric? 12. What is an R group? Discuss the various properties that the R group (side chains) possess. Give an example of each type of amino acid. 13. Describe the ...
... 11. Describe the structure of an amino acid. What is the alpha carbon? Do any of the amino acids contain alpha carbons that are not asymmetric? 12. What is an R group? Discuss the various properties that the R group (side chains) possess. Give an example of each type of amino acid. 13. Describe the ...
study guide section 3-1 carbon compounds
... 2. ______The different shapes and functions of different proteins are determined by a. the R groups of the amino acids they contain. b. the amino groups of the amino acids they contain. c. the carboxyl groups of the amino acids they contain. d. whether or not they contain any amino acids. 3. ______ ...
... 2. ______The different shapes and functions of different proteins are determined by a. the R groups of the amino acids they contain. b. the amino groups of the amino acids they contain. c. the carboxyl groups of the amino acids they contain. d. whether or not they contain any amino acids. 3. ______ ...
Anaerobic Respiration
... Some organisms, such as yeast and some bacteria, do not require oxygen and can survive on a less efficient way of getting energy Other organisms that generally require oxygen sometimes don’t have enough for all their cells to do aerobic respiration so they can use a less effiecent way of breaking do ...
... Some organisms, such as yeast and some bacteria, do not require oxygen and can survive on a less efficient way of getting energy Other organisms that generally require oxygen sometimes don’t have enough for all their cells to do aerobic respiration so they can use a less effiecent way of breaking do ...
Ion exchange chromatography
... Chromatography is separation method based on different retention of analytes on the stationary phase. Different chemical of physical phenomena can be used to retain an analyte. Depending on the phenomenon responsible for separation several types of chromatography are considered. Types of chromatogr ...
... Chromatography is separation method based on different retention of analytes on the stationary phase. Different chemical of physical phenomena can be used to retain an analyte. Depending on the phenomenon responsible for separation several types of chromatography are considered. Types of chromatogr ...
$doc.title
... From the balanced equation you can predict the quantity of tris(ethylenediamine)nickel(II) chloride that would be expected if the reaction went to completion. This quantity is called the theoretical yield and may be expressed in either grams or moles. Since most chemical reactions do not go to compl ...
... From the balanced equation you can predict the quantity of tris(ethylenediamine)nickel(II) chloride that would be expected if the reaction went to completion. This quantity is called the theoretical yield and may be expressed in either grams or moles. Since most chemical reactions do not go to compl ...
Lecture 6
... any random biological process e. Tree of form similarities parallel to tree of biological process similarities f. DNA replication traps random error in time, for natural selection to work on Origin of LCA a. Problem of improbability before DNA, random mutation traps history b. Two models, not mutual ...
... any random biological process e. Tree of form similarities parallel to tree of biological process similarities f. DNA replication traps random error in time, for natural selection to work on Origin of LCA a. Problem of improbability before DNA, random mutation traps history b. Two models, not mutual ...
UNIT 1 - MATTER AND CHEMICAL BONDING
... f) ferrous iodide l) cobalt(III) sulphate 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper b) phosphorus + oxygen diphosphorus pentoxide c) calcium carbonate ca ...
... f) ferrous iodide l) cobalt(III) sulphate 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper b) phosphorus + oxygen diphosphorus pentoxide c) calcium carbonate ca ...
AP Chemistry - School Webmasters
... Welcome to AP Chemistry. In order to ensure the best start for everyone next fall, I have prepared a summer assignment that reviews basic chemistry concepts some of which you may have forgotten you learned. For those topics you need help with there are a multitude of tremendous chemistry resources a ...
... Welcome to AP Chemistry. In order to ensure the best start for everyone next fall, I have prepared a summer assignment that reviews basic chemistry concepts some of which you may have forgotten you learned. For those topics you need help with there are a multitude of tremendous chemistry resources a ...
SEMESTER 1 EXAM Prblms/Short Ans
... 16.Write the formulas for the following binary compounds formed between the following elements: (#1 p. 223) a. Sodium and sulfur _______________________ d. aluminum and nitrogen __________________ Name the binary ionic compound as indicated by the following formulas: (#2 p. 223) b. AgCl ___________ ...
... 16.Write the formulas for the following binary compounds formed between the following elements: (#1 p. 223) a. Sodium and sulfur _______________________ d. aluminum and nitrogen __________________ Name the binary ionic compound as indicated by the following formulas: (#2 p. 223) b. AgCl ___________ ...
Chapt 2
... Which of the amino acids described below has the most potential for forming ionic bonds: 1. polar, non-charged 2. polar, charged 3. nonpolar 4. glycine 5. proline Hydrogen bonds can occur between which of the following molecules: 1. The sulfhydral in cysteine and the hydroxyl of serine 2. Two parts ...
... Which of the amino acids described below has the most potential for forming ionic bonds: 1. polar, non-charged 2. polar, charged 3. nonpolar 4. glycine 5. proline Hydrogen bonds can occur between which of the following molecules: 1. The sulfhydral in cysteine and the hydroxyl of serine 2. Two parts ...
Molecules of life 2.4 - Madison County Schools
... A. Proteins make up greater than 50% of an organisms dry weight (referred to as biomass). B. Proteins are not used for energy unless there are no lipids or carbohydrates available. Proteins and enzymes are the “work horses” of a cell. They carry out numerous functions within cells. Proteins basicall ...
... A. Proteins make up greater than 50% of an organisms dry weight (referred to as biomass). B. Proteins are not used for energy unless there are no lipids or carbohydrates available. Proteins and enzymes are the “work horses” of a cell. They carry out numerous functions within cells. Proteins basicall ...
Exam1
... 4. In an aqueous solution, protein conformation is determined by two major factors. One is the formation of the maximum number of hydrogen bonds. The other is the: A) formation of the maximum number of hydrophilic interactions. B) maximization of ionic interactions. C) minimization of entropy by the ...
... 4. In an aqueous solution, protein conformation is determined by two major factors. One is the formation of the maximum number of hydrogen bonds. The other is the: A) formation of the maximum number of hydrophilic interactions. B) maximization of ionic interactions. C) minimization of entropy by the ...
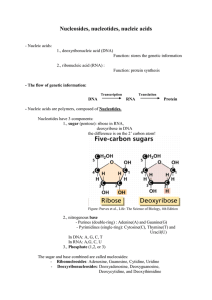
Nucleosides, nucleotides, nucleic acids
... The nucleotide sequence (=base sequence) carries the genetic information, this information will be translated into amino-acid sequence during protein synthesis. - Types and structure of RNA: - messenger RNA = mRNA: carries the information from the DNA to the site of protein synthesis. Single strande ...
... The nucleotide sequence (=base sequence) carries the genetic information, this information will be translated into amino-acid sequence during protein synthesis. - Types and structure of RNA: - messenger RNA = mRNA: carries the information from the DNA to the site of protein synthesis. Single strande ...
honors final key
... 11. Identify the following symbol. a. yield b. Δ change in heat c. Mn On catalyst d. + and e. (s) solid f. (g) gas g. (aq) aqueous, in solution, dissolved in water h. (l) liquid ...
... 11. Identify the following symbol. a. yield b. Δ change in heat c. Mn On catalyst d. + and e. (s) solid f. (g) gas g. (aq) aqueous, in solution, dissolved in water h. (l) liquid ...
ChemicalBondingPowerpoint
... H2O) probably dominated Earth’s early atmosphere, but H2, NH3, and CH4 were also present in sufficient amounts to form H2CO and HCN. ...
... H2O) probably dominated Earth’s early atmosphere, but H2, NH3, and CH4 were also present in sufficient amounts to form H2CO and HCN. ...
Proteins
... Held together by hydrogen bonds between hydrogen from the oxygen from the carbonyl group C=O and hydrogen from the amino group N – H that is four peptide bonds away Hair is an example ...
... Held together by hydrogen bonds between hydrogen from the oxygen from the carbonyl group C=O and hydrogen from the amino group N – H that is four peptide bonds away Hair is an example ...
Biomolecules PPT
... What is another everyday example you can think of that have monomers and polymers? ...
... What is another everyday example you can think of that have monomers and polymers? ...
Chemistry in Biology - Welcome to teachers.olatheschools.com!
... The number and the order in which the amino acids are joined define the protein’s primary structure. After an amino acid chain is formed, it folds into a unique three-dimensional shape, which is the protein’s secondary structure, such as a helix or a ...
... The number and the order in which the amino acids are joined define the protein’s primary structure. After an amino acid chain is formed, it folds into a unique three-dimensional shape, which is the protein’s secondary structure, such as a helix or a ...
Name: Date: Concept Check Questions Chapter 9 Cellular
... 1. During the redox reaction in glycolysis (step 6 in figure 9.9 in the orange book), which molecule acts as the oxidizing agent? The reducing agent? 9.3 The citric acid cycle completes the energy-yielding oxidation of organic molecules ...
... 1. During the redox reaction in glycolysis (step 6 in figure 9.9 in the orange book), which molecule acts as the oxidizing agent? The reducing agent? 9.3 The citric acid cycle completes the energy-yielding oxidation of organic molecules ...
practice midterm
... Multiple Choice 1) The role of an enzyme in an enzyme-catalyzed reaction is to A) bind a transition state intermediate, such that it can be converted back to a substrate B) ensure that all substrate is converted to product C) ensure that product is more stable than substrate D) increase the rate at ...
... Multiple Choice 1) The role of an enzyme in an enzyme-catalyzed reaction is to A) bind a transition state intermediate, such that it can be converted back to a substrate B) ensure that all substrate is converted to product C) ensure that product is more stable than substrate D) increase the rate at ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.